Abstract
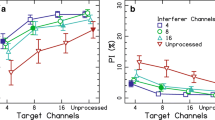
A cochlear implant (CI) electrode in a “cochlear dead region” will excite neighboring neural populations. In previous research that simulated such dead regions, stimulus information in the simulated dead region was either added to the immediately adjacent frequency regions or dropped entirely. There was little difference in speech perception ability between the two conditions. This may imply that there may be little benefit of ensuring that stimulus information on an electrode in a suspected cochlear dead region is transmitted. Alternatively, performance may be enhanced by a broader frequency redistribution, rather than adding stimuli from the dead region to the edges. In the current experiments, cochlear dead regions were introduced by excluding selected CI electrodes or vocoder noise-bands. Participants were assessed for speech understanding as well as spectral and temporal sensitivities as a function of the size of simulated dead regions. In one set of tests, the normal input frequency range of the sound processor was distributed among the active electrodes in bands with approximately logarithmic spacing (“redistributed” maps); in the remaining tests, information in simulated dead regions was dropped (“dropped” maps). Word recognition and Schroeder-phase discrimination performance, which require both spectral and temporal sensitivities, decreased as the size of simulated dead regions increased, but the redistributed and dropped remappings showed similar performance in these two tasks. Psychoacoustic experiments showed that the near match in word scores may reflect a tradeoff between spectral and temporal sensitivity: spectral-ripple discrimination was substantially degraded in the redistributed condition relative to the dropped condition while performance in a temporal modulation detection task degraded in the dropped condition but remained constant in the redistributed condition.












Similar content being viewed by others
References
Başkent D, Shannon RV (2003) Speech recognition under conditions of frequency-place compression and expansion. J Acoust Soc Am 113:2064–2076
Başkent D, Shannon RV (2004) Frequency-place compression and expansion in cochlear implant listeners. J Acoust Soc Am 116:3130–3140
Başkent D, Shannon RV (2005) Interactions between cochlear implant electrode insertion depth and frequency-place mapping. J Acoust Soc Am 117:1405–1416
Başkent D, Shannon RV (2006) Frequency transposition around dead regions simulated with a noiseband vocoder. J Acoust Soc Am 119:1156–1163
Bierer JA (2010) Probing the electrode-neuron interface with focused cochlear implant stimulation. Trends Amplif 14:84–95
Bierer JA, Faulkner KF, Tremblay KL (2011) Identifying cochlear implant channels with poor electrode-neuron interfaces: electrically evoked auditory brain stem responses measured with the partial tripolar configuration. Ear Hear 32(4):436–444
Bingabr M, Espinoza-Varas B, Loizou PC (2008) Simulating the effect of spread of excitation in cochlear implants. Hear Res 241:73–79
Crew JD, Galvin JJ 3rd, Fu QJ (2012) Channel interaction limits melodic pitch perception in simulated cochlear implants. J Acoust Soc Am 132(5):EL429–EL435
Dorman MF, Loizou PC, Rainey D (1997) Simulating the effect of cochlear implant electrode insertion depth on speech understating. J Acoust Soc Am 102:2993–2996
Drennan WR, Longnion JK, Ruffin C, Rubinstein JT (2008) Discrimination of Schroeder-phase harmonic complexes by normal-hearing and cochlear-implant listeners. J Assoc Res Otolaryngol 9:138–149
Drennan WR, Won JH, Nie KB, Jameyson E, Rubinstein JT (2010) Sensitivity of psychophysical measures to signal processor modifications in cochlear implant users. Hear Res 262:1–8
Faulkner A (2006) Adaptation to distorted frequency-to-place maps: implications of simulations in normal listeners for cochlear implants and electroacoustic stimulation. Audiol Neurootol 11(suppl 1):21–26
Fishman K, Shannon RV, Slattery WH (1997) Speech recognition as a function of the number of electrodes used in the SPEAK cochlear implant speech processor. J Speech Hear Res 40:1201–1215
Friesen LM, Shannon RV, Başkent D, Wang X (2001) Speech recognition in noise as a function of the number of spectral channels: comparison of acoustic hearing and cochlear implants. J Acoust Soc Am 110:1150–1163
Fu QJ, Galvin J III (2003) The effects of short-term training for spectrally mismatched noise-band speech. J Acoust Soc Am 113:1065–1072
Fu QJ, Shannon RV (1999) Recognition of spectrally degraded and frequency-shifted vowels in acoustic and electric hearing. J Acoust Soc Am 105:1889–1900
Fu QJ, Shannon RV (2000) Effect of stimulation rate on phoneme recognition by Nucleus-22 cochlear implant listeners. J Acoust Soc Am 107:589–597
Garadat SN, Zwolan TA, Pfingst BE (2012) Across-site patterns of modulation detection: relation to speech recognition. J Acoust Soc Am 131:4030–4041
Greenwood DD (1990) A cochlear frequency-position function for several species—29 years later. J Acoust Soc Am 87:2592–2605
Henry BA, Turner CW (2003) The resolution of complex spectral patterns by cochlear implant and normal-hearing listeners. J Acoust Soc Am 113:2861–2873
Jones GL, Won JH, Drennan WR, Rubinstein JT (2013) Relationship between channel interaction and spectral-ripple discrimination in cochlear implant users. J Acoust Soc Am 133:425–433
Kasturi K, Loizou PC, Dorman M, Spahr T (2002) The intelligibility of speech with “holes” in the spectrum. J Acoust Soc Am 112:1102–1111
Kawano A, Seldon H, Clark G (1996) Computer-aided three-dimensional reconstruction in human cochlear maps: measurement of the lengths of organ of Corti, outer wall, inner wall and Rosenthal’s canal. Ann Otol Rhinol Laryngol 105:701–709
Laneau J, Moonen M, Wouters J (2006) Factors affecting the use of noise-band vocoders as acoustic models for pitch perception in cochlear implants. J Acoust Soc Am 119(1):491–506
Levitt H (1971) Transformed up-down methods in psychoacoustics. J Acoust Soc Am 49:467–477
Long CJ, Holden TA, McClelland GH, Parkinson WS, Shelton C, Kelsall DC, Smith ZM (2014) Examining the electro-neural interface of cochlear implant users using psychophysics, CT scans, and speech understanding. J Assoc Res Otolaryngol 15:293–304
Makary CA, Shin J, Kujawa SG, Liberman MC, Merchant SN (2011) Age-related primary cochlear neuronal degeneration in human temporal bones. J Assoc Res Otolaryngol 12:711–717
Moore BCJ (2004) Dead regions in the cochlea: conceptual foundations, diagnosis, and clinical applications. Ear Hear 25:98–116
Nelson DA, Donaldson GS, Kreft H (2008) Forward-masked spatial tuning curves in cochlear implant users. J Acoust Soc Am 123:1522–1543
Nie K, Barco A, Zeng FG (2006) Spectral and temporal cues in cochlear implant speech perception. Ear Hear 27:208–217
Noble JH, Labadie RF, Gifford RH, Dawant BM (2013) Image-guidance enables new methods for customizing cochlear implant stimulation strategies. IEEE Trans Neural Syst Rehabil Eng 21:820–829
Peterson GE, Lehiste I (1962) Revised CNC lists for auditory tests. J Speech Hear Disord 27:62–70
Schucknecht HF (1964) Further observations on the pathology of presbycusis. Arch Otolaryngol 80:369–382
Shannon RV, Zeng FG, Wygonski J (1998) Speech recognition with altered spectral distribution of envelope cues. J Acoust Soc Am 104:2467–2476
Shannon RV, Galvin JJ, Başkent D (2002) Holes in hearing. J Assoc Res Otolaryngol 3:185–199
Shannon RV, Cruz RJ, Galvin JJ (2011) Effect of stimulation rate on cochlear implant users’ phoneme, word and sentence recognition in quiet and in noise. Audiol Neurootol 16:113–123
Smith MW, Faulkner A (2006) Perceptual adaptation by normally hearing listeners to a simulated “hole” in hearing. J Acoust Soc Am 120:4019–4030
Sridhar D, Stakhovskaya O, Leake PA (2006) A frequency-position map for the human spiral ganglion. Audiol Neurootol 11:16–20
Strickland EA, Viemeister NF (1997) The effects of frequency region and bandwidth on the temporal modulation transfer function. J Acoust Soc Am 102(3):1799–1810
Weber BP, Lai WK, Dillier N, von Wallenberg EL, Killian MJP, Pesch J, Battmer RD, Lenarz T (2007) Performance and preference for ACE stimulation rates obtained with nucleus RP 8 and freedom system. Ear Hear 28:46s–48s
Won JH, Drennan WR, Rubinstein JT (2007) Spectral-ripple resolution correlates with speech reception in noise in cochlear implant users. J Assoc Res Otolaryngol 8:384–392
Won JH, Drennan WR, Kang RS, Rubinstein JT. (2010) Psychoacoustic abilities associated with music perception in cochlear implant users. Ear Hear 31(6):796–805
Won JH, Drennan WR, Nie K, Jameyson EM, Rubinstein JT (2011a) Acoustic temporal modulation detection and speech perception in cochlear implant listeners. J Acoust Soc Am 130:376–388
Won JH, Clinard CG, Kwon SY, Dasika VK, Nie K, Drennan WR, Tremblay KL, Rubinstein JT (2011b) Relationship between behavioral and physiologic spectral-ripple discrimination. J Assoc Res Otolaryngol 12:375–393
Won JH, Jones GL, Drennan WR, Jameyson EM, Rubinstein JT (2011c) Evidence of across-channel processing for spectral-ripple discrimination in cochlear implant listeners. J Acoust Soc Am 130(4):2088–2097
Won JH, Humphrey EL, Yeager KR, Martinez AA, Robinson CH, Mills KE, Johnstone PM, Moon IJ, Woo JH (2014) Relationship among the physiologic channel interactions, spectral-ripple discrimination, and vowel identification in cochlear implant users. J Acoust Soc Am 136(5):2714–2725
Xu L, Thompson CS, Pfingst BE (2005) Relative contributions of spectral and temporal cues for phoneme recognition. J Acoust Soc Am 117:3255–3267
Acknowledgments
We are grateful for the dedicated efforts of our cochlear implant and normal-hearing subjects. This research was supported by NIH-NIDCD Grants F31-DC009755, F32-DC011431, R01-DC007525, and P30-DC04661 and the Advanced Bionics Corporation. Il Joon Moon was supported by the Samsung Medical Center.
Conflict of Interest Disclosure
The authors have no conflict of interests regarding this work.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Won, J.H., Jones, G.L., Moon, I.J. et al. Spectral and Temporal Analysis of Simulated Dead Regions in Cochlear Implants. JARO 16, 285–307 (2015). https://doi.org/10.1007/s10162-014-0502-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10162-014-0502-8