Abstract
Background
There is little information about very long-term outcomes of kidney allograft recipients exposed to calcineurin inhibitors.
Methods
In this single-centre retrospective study with 20-year follow-up, we analyzed data from 644 patients who underwent primary renal transplantation between 1983 and 1993. Participants were treated with a cyclosporine-based immunosuppressive scheme and had allograft function at 1 year.
Results
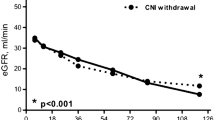
After 20 years, 15.2% patients died, 39.7% experienced allograft loss, 26.8% were alive with a functioning transplant, and 18.2% were lost to follow-up. Cardiovascular disease (30.8%), malignancy (26.6%) and infection (17.0%) were the main causes of death. Age, new-onset proteinuria > 1 g/day, major acute cardiovascular event (MACE), and malignancy were independent predictors of mortality at time-dependent multivariate analysis. Chronic rejection (63.3%), recurrent glomerulonephritis (14.0%), and nonspecific interstitial fibrosis/tubular atrophy (13.2%) were the leading cause of allograft loss. Basal disease, hepatitis C, difference between 1 year and nadir serum creatinine, new-onset proteinuria > 1 g/day, and MACE were independent predictors of transplant failure. Among patients with 20-year allograft function, we recorded the following complications: hypertension (85%), malignancy (13%), diabetes (9%), and cardiovascular disease (9%). Median serum creatinine and proteinuria were 1.4 mg/dL and 0.6 g/day, respectively.
Conclusions
Prolonged use of cyclosporine may expose to several dose-related adverse events and may contribute to the development of allograft dysfunction but it does not necessarily cause relentless, progressive transplant failure if patients are carefully and consistently monitored during the follow-up.




Similar content being viewed by others
References
Matas A, Gillingham K, Humar A, et al. 2,202 kidney transplant recipients with 10 years of graft function: What happens next? Am J Transplant. 2008;8:2410–9.
Traynor C, Jenkinson A, Williams Y, et al. Twenty-year survivors of kidney transplant. Am J Transplant. 2012;12:3289–95.
McCaughan JA, Courtney AE. The clinical course of kidney transplant recipients after 20 years of graft function. Am J Transplant. 2015;15:734–40.
Rao KV, Kasiske BL, Dahl DC, et al. Long-term results and complications of renal transplantation: the hennepin experience. Clin Transplant. 1997;119–24.
Chapman JR. Chronic calcineurin inhibitor nephrotoxicity-lest we forget. Am J Transplant. 2011;11:693–7.
European Multicentre Trial Group. Cyclosporin in cadaveric renal transplantation: 1-year follow-up of a multicentre trial. Lancet. 1983;2:986–9.
Loupy A, Haas M, Solez K, et al. The Banff 2015 kidney meeting report: current challenges in rejection classification and prospects for adopting molecular pathology. Am J Transplant. 2017;17:28–41.
Montagnino G, Tarantino A, Segoloni GP, et al. Long-term results of a randomized study comparing three immunosuppressive schedules with cyclosporine in cadaveric kidney transplantation. J Am Soc Nephrol. 2001;12:2163–9.
Opelz G, Dohler B. Association between steroid dosage and death with a functioning graft after kidney transplantation. Am J Transplant. 2013;13:2096–105.
Luan FL, Steffick DE, Ojo AO. Steroid-free immunosuppression in kidney transplantation: is it time to consider it? Kidney Int. 2009;76:825–30.
Wong G, Chapman JR. Cancers after renal transplantation. Transplant Rev (Orlando). 2008;22:141–9.
Morath C, Mueller M, Goldschmidt H, Schwenger V, Opelz G, Zeier M. Malignancy in renal transplantation. JASN. 2004;15:1582–8.
Dantal J, Hourmant M, Cantarovich D, et al. Effect of long-term immunosuppression in kidney-graft recipients on cancer incidence: randomised comparison of two cyclosporin regimens. Lancet. 1998;351:623–8.
Alfadheel TA, Soroka SD, Kiberd BA, Landry D, Moorhouse P, Tennankore KK. Frailty and mortality in dialysis: evaluation of a clinical frailty scale. Clin J Am Soc Nephrol. 2015;10:832–40.
Ljungman S, Wikstrand J, Hartford M, Berglund G. Urinary albumin excretion—a predictor of risk of cardiovascular disease: a prospective 10-year follow-up of middle-aged nondiabetic normal and hypertensive men. Am J Hypertens. 1996;9:770–8.
Agrawal V, Marinescu V, Agarwal M, McCullough PA. Cardiovascular implications of proteinuria: an indicator of chronic kidney disease. Nat Rev Cardiol. 2009;6:301–11.
Sarnak MJ, Levey AS, Schoolwerth AC, et al. Kidney disease as a risk factor for development of cardiovascular disease: a statement from the American Heart Association Councils on Kidney in Cardiovascular Disease, High Blood Pressure Research, Clinical Cardiology, and Epidemiology and Prevention. Circulation. 2003;108:2154–69.
Sellarés J, de Freitas DG, Mengel M, et al. Understanding the causes of kidney transplant failure: the dominant role of antibody-mediated rejection and nonadherence. Am J Transplant. 2012;2:388–99.
Montgomery RA, Loupy A, Segev DL. Antibody-mediated rejection: new approaches in prevention and management. Am J Transplant. 2018;18:3–17.
Solez K, Colvin RB, Racusen LC, et al. Banff’ 05 meeting report: differential diagnosis of chronic allograft injury and elimination of chronic allograft nephropathy (‘CAN’). Am J Transplant. 2007;7:518–26.
Ponticelli C, Glassock RJ. Posttransplant recurrence of primary glomerulonephritis. Clin J Am Soc Nephrol. 2010;5:2363–72.
Ponticelli C, Moroni G, Glassock RJ. Recurrence of secondary glomerular disease after renal transplantation. Clin J Am Soc Nephrol. 2011;6:1214–21.
Briganti EM, Russ GR, McNeil JJ, Atkins RC, Chadban SJ. Risk of renal allograft loss from recurrent glomerulonephritis. N Engl J Med. 2002;347:103–9.
Pippias M, Stel VS, Aresté-Fosalba N, et al. Long-term kidney transplant outcomes in primary glomerulonephritis: analysis from the ERA-EDTA Registry. Transplantation. 2016;100:1955–62.
Morales JM, Aguado JM. (2012) Hepatitis C and renal transplantation. Curr Opin Organ Transplant. 2012;17:609–15.
Ortiz F, Paavonen T, Törnroth T, et al. Predictors of renal allograft histologic damage progression. J Am Soc Nephrol. 2005;516:817–24.
Salvadori M, Rosati A, Bock A, et al. Estimated 1-year glomerular filtration rate is the best predictor of long-term graft function following renal transplant. Transplantation. 2006;81:202–6.
Roodnat JI, Mulder PG, Rischen-Vos J, et al. Proteinuria after renal transplantation affects not only graft survival but also patient survival. Transplantation. 2001;72:438–44.
Ronco C, McCullough P, Anker SD, et al. Cardio-renal syndromes: reports from the consensus conference of the acute dialysis quality initiative. Eur Heart J. 2010;31:703–11.
Damman K, van Deursen VM, Navis G, Voors AA, van Veldhuisen DJ, Hillege HL. Increased central venous pressure is associated with impaired renal function and mortality in a broad spectrum of patients with cardiovascular disease. J Am Coll Cardiol. 2009;53:582–8.
Wu C, Evans I, Joseph R, et al. Comorbid conditions in kidney transplantation: association with graft and patient survival. J Am Soc Nephrol. 2005;16:3437–44.
Venner JM, Famulski KS, Reeve J, Chang J, Halloran PF. Relationships among injury, fibrosis, and time in human kidney transplants. JCI Insight. 2016;1:e85323.
Issa N, Kukla A, Ibrahim HN. Calcineurin inhibitor nephrotoxicity: a review and perspective of the evidence. Am J Nephrol. 2013;37:602–12.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors have declared that no conflict of interest exists.
Ethical approval
All the procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional committee at which the studies were conducted and with the 1964 Helsinki Declaration and its later amendments or comparable ethical standards. At the time the study was designed, it was formally discussed in our regular Multidisciplinary Research Meeting at the Ospedale Maggiore Policlinico (Milan, Italy) and the consensus was that IRB approval was not necessary because it was retrospective and non-interventional. Therefore, nor specific IRB approval number nor specific written documents regarding the present study are actually available. All transplant recipients followed up at our institution are consented for both treatment and research purposes at the time of activation on the transplant waiting list as per local and national regulatory requirements. As a consequence, all subjects enrolled into the study were already aware that their anonymized data including viral status and other biomedical parameters would have been used for planning and/or research.
Informed consent
Informed consent was obtained from all individual participants included in the study.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
About this article
Cite this article
Moroni, G., Binda, V., Quaglini, S. et al. Causes of late transplant failure in cyclosporine-treated kidney allograft recipients. Clin Exp Nephrol 23, 1076–1086 (2019). https://doi.org/10.1007/s10157-019-01740-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10157-019-01740-7