Abstract
Background
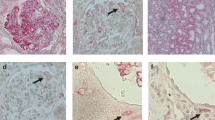
Apoptosis inhibitor of macrophage (AIM) expressed on macrophages prolongs inflammation by protecting macrophages from apoptosis. Most circulating AIM co-exists with immunoglobulin M (IgM). AIM’s pathophysiological role in relation to IgM remains unclear. Here we evaluated the glomerular expression/deposition of AIM and IgM in the kidney using immunohistochemistry and its associations with clinical manifestations in 43 patients with biopsy-confirmed kidney diseases.
Methods
Kidney biopsy tissue from all patients was immunostained for AIM and IgM. Staining patterns and percent stained areas within the glomeruli were determined. Cells expressing AIM were identified by co-staining with macrophage and endothelial cell surface markers. Correlations between staining results and clinical parameters were evaluated using univariate and multivariate analyses.
Results
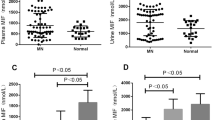
AIM was deposited in various areas, such as mesangial and capillary area. A part of AIM expression was localized to CD68-positive macrophages in the glomerulus. Amount of glomerular expression was positively correlated with urinary protein in patients with severe proteinuria (urinary protein ≥0.5 g/day) and kidney dysfunction [estimated glomerular filtration ratio (eGFR) <60 ml/min/1.73 m2]. Urinary protein was higher in patients exhibiting overlapping glomerular expression of AIM and IgM. Annual eGFR decline rate negatively correlated with AIM-positive area. AIM-positive area and initial serum creatinine were independently associated with decreased kidney function.
Conclusion
AIM expression in the kidney was associated with urinary protein and decline in kidney function. Co-expression with IgM appeared to exacerbate AIM’s deleterious effects on kidney function. Combined glomerular AIM and IgM expression is a candidate prognostic index for kidney disease.




Similar content being viewed by others
References
Miyazaki T, Hirokami Y, Matsuhashi N, Takatsuka H, Naito M. Increased susceptibility of thymocytes to apoptosis in mice lacking AIM, a novel murine macrophage-derived soluble factor belonging to the scavenger receptor cysteine-rich domain superfamily. J Exp Med. 1999;189:413–22.
Mera K, Uto H, Mawatari S, Ido A, Yoshimine Y, Nosaki T, Oda K, Tabu K, Kumagai K, Tamai T, Moriuchi A, Oketani M, Shimada Y, Hidaka M, Eguchi S, Tsubouchi H. Serum levels of apoptosis inhibitor of macrophage are associated with hepatic fibrosis in patients with chronic hepatitis C. BMC Gastroenterol. 2014;14:27.
Yamazaki T, Mori M, Arai S, Tateishi R, Abe M, Ban M, Nishijima A, Maeda M, Asano T, Kai T, Izumino K, Takahashi J, Aoyama K, Harada S, Takebayashi T, Gunji T, Ohnishi S, Seto S, Yoshida Y, Hiasa Y, Koike K, Yamamura K, Inoue K, Miyazaki T. Circulating AIM as an indicator of liver damage and hepatocellular carcinoma in humans. PLoS One. 2014;9:e109123.
Kojima J, Araya J, Hara H, Ito S, Takasaka N, Kobayashi K, Fujii S, Tsurushige C, Numata T, Ishikawa T, Shimizu K, Kawaishi M, Saito K, Kamiya N, Hirano J, Odaka M, Morikawa T, Hano H, Arai S, Miyazaki T, Kaneko Y, Nakayama K, Kuwano K. Apoptosis inhibitor of macrophage (AIM) expression in alveolar macrophages in COPD. Respir Res. 2013;14:30.
Uramatsu T, Nishio T, Obata T, Sato Y, Furusu A, Koji T, Miyazaki T, Kohno S. Involvement of apoptosis inhibitor of macrophages in a rat hypertension model with nephrosclerosis: possible mechanisms of action of olmesartan and azelnidipine. Biol Pharm Bull. 2013;36:1271–7.
Hamada M, Nakamura M, Tran MT, Moriguchi T, Hong C, Ohsumi T, Dinh TT, Kusakabe M, Hattori M, Katsumata T, Arai S, Nakashima K, Kudo T, Kuroda E, Wu CH, Kao PH, Sakai M, Shimano H, Miyazaki T, Tontonoz P, Takahashi S. MafB promotes atherosclerosis by inhibiting foam-cell apoptosis. Nat Commun. 2014;5:3147.
Miyazaki T, Kurokawa J, Arai S. AIMing at metabolic syndrome. Towards the development of novel therapies for metabolic disease via apoptosis inhibitor of macrophage (AIM). Circ J. 2011;75:2522–31.
Arai S, Miyazaki T. Impacts of the apoptosis inhibitor of macrophage (AIM) on obesity-associated inflammatory diseases. Semin Immunopathol. 2014;36:3–12.
Arai S, Maehara N, Iwamura Y, Honda S, Nakashima K, Kai T, Ogishi M, Morita K, Kurokawa J, Mori M, Motoi Y, Miyake K, Matsuhashi N, Yamamura K, Ohara O, Shibuya A, Wakeland EK, Li QZ, Miyazaki T. Obesity-associated autoantibody production requires AIM to retain IgM immune complex on follicular dendritic cells. Cell Rep. 2015;3:1187–98.
Kai T, Yamazaki T, Arai S, Miyazaki T. Stabilization and augmentation of circulating AIM in mice by synthesized IgM-Fc. PLoS One. 2014;9:e97037.
Strassheim D, Renner B, Panzer S, Fuquay R, Kulik L, Ljubanović D, Holers VM, Thurman JM. IgM contributes to glomerular injury in FSGS. J Am Soc Nephrol. 2013;24:393–406.
Panzer SE, Laskowski J, Renner B, Kulik L, Ljubanovic D, Huber KM, Zhong W, Pickering MC, Holers VM, Thurman JM. IgM exacerbates glomerular disease progression in complement-induced glomerulopathy. Kidney Int. 2015;88:528–37.
Mubarak M. IgM nephropathy; time to act. J Nephropathol. 2014;3:22–5.
Cohen AH, Border WA, Glassock RJ. Nephrotic syndrome with glomerular mesangial IgM deposits. Lab Invest. 1978;38:610–9.
Bhasin HK, Abuelo JG, Nayak R, Esparza AR. Mesangial proliferative glomerulonephritis. Lab Invest. 1978;39:21–9.
Mubarak M, Kazi JI. IgM nephropathy revisited. Nephrourol Mon. 2012;4:603–8.
Myllymaki J, Saha H, Mustonen J, Helin H, Pasternack A. IgM nephropathy: clinical picture and long-term prognosis. Am J Kidney Dis. 2003;41:343–50.
Yokoyama H, Wada T, Furuichi K. Chemokines in renal fibrosis. Contrib Nephrol. 2003;139:66–89.
Iwata Y, Boström EA, Menke J, Rabacal WA, Morel L, Wada T, Kelley VR. Aberrant macrophages mediate defective kidney repair that triggers nephritis in lupus-susceptible mice. J Immunol. 2012;188:4568–80.
Sakai N, Wada T, Furuichi K, Iwata Y, Yoshimoto K, Kitagawa K, Kokubo S, Kobayashi M, Hara A, Yamahana J, Okumura T, Takasawa K, Takeda S, Yoshimura M, Kida H, Yokoyama H. Involvement of extracellular signal-regulated kinase and p38 in human diabetic nephropathy. Am J Kidney Dis. 2005;45:54–65.
Furuichi K, Gao JL, Horuk R, Wada T, Kaneko S, Murphy PM. Chemokine receptor CCR1 regulates inflammatory cell infiltration after renal ischemia-reperfusion injury. J Immunol. 2008;181:8670–6.
Wada T, Yokoyama H, Furuichi K, Kobayashi KI, Harada K, Naruto M, Su SB, Akiyama M, Mukaida N, Matsushima K. Intervention of crescentic glomerulonephritis by antibodies to monocyte chemotactic and activating factor (MCAF/MCP-1). FASEB J. 1996;10:1418–25.
Kitagawa K, Wada T, Furuichi K, Hashimoto H, Ishiwata Y, Asano M, Takeya M, Kuziel WA, Matsushima K, Mukaida N, Yokoyama H. Blockade of CCR2 ameliorates progressive fibrosis in kidney. Am J Pathol. 2004;165:237–46.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors have declared that no conflict of interest exists.
About this article
Cite this article
Oshima, M., Iwata, Y., Furuichi, K. et al. Association of apoptosis inhibitor of macrophage (AIM) expression with urinary protein and kidney dysfunction. Clin Exp Nephrol 21, 35–42 (2017). https://doi.org/10.1007/s10157-016-1240-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10157-016-1240-5