Abstract
Background
The purpose was to compare the effectiveness and safety of calcineurin inhibitors (CNIs) withdrawal and continued therapies in kidney transplant recipients.
Methods
We searched the PubMed, MEDLINE, Springer, Elsevier Science Direct, Cochrane Library and Google Scholar up to May 2014. Risk ratio (RR) or weighted mean difference (WMD) and their 95 % confidence intervals (CIs) were calculated in fixed-effects model or random-effects model when appropriate. Besides, sensitivity analysis was performed based on the addition of sirolimus in initial immunosuppression protocols.
Results
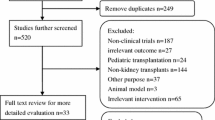
Total seven studies with 1071 kidney transplant recipients received CNIs withdrawal therapy (experimental group) and 792 kidney transplant recipients received CNIs continued therapy (control group) were included in the meta-analysis. The overall estimates of acute rejection rate (RR = 1.64, 95 % CI: 1.19–2.27, P = 0.003), mean measured glomerular filtration rate (WMD = 9.50, 95 % CI = 2.96–16.03, P = 0.004), thrombocytopenia (RR = 3.39, 95 % CI: 2.27–5.05, P < 0.00001) and hypertension (RR = 0.56, 95 % CI: 0.40–0.78, P = 0.0006) showed that there were significant differences between the CNIs withdrawal and continued therapies in kidney transplant recipients, while no significant differences were found between groups in survival rate, graft survival rate, diabetes, hypercholesterolemia, hypertriglyceridemia and malignancies. In addition, two studies, in which sirolimus was not used in initial immunosuppression protocol, were excluded in sensitivity analysis and the results were still consistent with the overall analysis.
Conclusions
CNIs withdrawal therapy in kidney transplant recipients could significantly decrease risk of hypertension and improve glomerular filtration rate, accompanying with increased risk of acute rejection and thrombocytopenia, compared with the CNIs continued therapy.



Similar content being viewed by others
Abbreviations
- CNIs:
-
Calcineurin inhibitors
- RCT:
-
Randomized controlled trials
- RR:
-
Risk ratio
- WMD:
-
Weighted mean difference
References
Lozano R, Naghavi M, Foreman K, et al. Global and regional mortality from 235 causes of death for 20 age groups in 1990 and 2010: a systematic analysis for the Global Burden of Disease Study 2010. Lancet. 2012;380(9859):2095–128.
Cortazar F, Diaz-Wong R, Roth D, Isakova T. Corticosteroid and calcineurin inhibitor sparing regimens in kidney transplantation. Nephrol Dial Transplant. 2013;28(11):2708–16.
Bellingham JM, Anderson ME, D’Alessandro AM. Successful kidney transplantation from a donation after cardiac death donor with an ileal conduit. Transplant Proc. 2009;41(10):4420–1.
Eknoyan G, Levin NW. K/DOQI clinical practice guidelines for chronic kidney disease: evaluation, classification, and stratification. Am J Kidney Dis. 2002;39(2 Suppl 1):S1–266.
de Francisco AL, Pinera C. Challenges and future of renal replacement therapy. Hemodial Int. 2006;10(Suppl 1):S19–23.
Roodnat JI, Hilbrands LB, Hene RJ, et al. 15-year follow-up of a multicenter, randomized, calcineurin inhibitor withdrawal study in kidney transplantation. Transplantation. 2014;98(1):47–53.
Krejci K, Tichy T, Bachleda P, Zadrazil J. Calcineurin inhibitor-induced renal allograft nephrotoxicity. Biomed Pap Med Fac Univ Palacky Olomouc Czech Repub. 2010;154(4):297–306.
Tanabe T, Morita K, Fujita H, et al. Long-term clinicopathological impact of calcineurin inhibitor cessation without specific cytoreductive induction in kidney transplantation. Clin Transplant. 2013;27(Suppl 26):9–13.
Chapman JR, Nankivell BJ. Nephrotoxicity of ciclosporin A: short-term gain, long-term pain? Nephrol Dial Transplant. 2006;21(8):2060–3.
Naesens M, Lerut E, Damme BV, Vanrenterghem Y, Kuypers DRJ. Tacrolimus exposure and evolution of renal allograft histology in the first year after transplantation. Am J Transplant. 2007;7(9):2114–23.
Chen GD, Liu XC, Shi L, et al. Calcineurin inhibitor conversion to rapamycin can improve graft function in living donor kidney transplantation with older donors. Transplant Proc. 2013;45(4):1648–50.
Zeier M, Van Der Giet M. Calcineurin inhibitor sparing regimens using m-target of rapamycin inhibitors: an opportunity to improve cardiovascular risk following kidney transplantation? Transpl Int. 2011;24(1):30–42.
Gonwa TA, Hricik DE, Brinker K, Grinyo JM, Schena FP, Sirolimus Renal Function Study G. Improved renal function in sirolimus-treated renal transplant patients after early cyclosporine elimination. Transplantation. 2002;74(11):1560–7.
Stallone G, Di Paolo S, Schena A, et al. Early withdrawal of cyclosporine A improves 1-year kidney graft structure and function in sirolimus-treated patients. Transplantation. 2003;75(7):998–1003.
Grinyo JM, Campistol JM, Paul J, et al. Pilot randomized study of early tacrolimus withdrawal from a regimen with sirolimus plus tacrolimus in kidney transplantation. Am J Transplant. 2004;4(8):1308–14.
Lebranchu Y, Thierry A, Toupance O, et al. Efficacy on renal function of early conversion from cyclosporine to sirolimus 3 months after renal transplantation: concept study. Am J Transplant. 2009;9(5):1115–23.
Schena FP, Pascoe MD, Alberu J, et al. Conversion from calcineurin inhibitors to sirolimus maintenance therapy in renal allograft recipients: 24-month efficacy and safety results from the CONVERT trial. Transplantation. 2009;87(2):233–42.
Hausmann H. [Ernst Unger (1875–1938)—a pioneer in kidney transplantation. On the 50th anniversary of the death of the Berlin surgeon]. Z Arztl Fortbild (Jena). 1988;82(17):843–5.
Mantel N, Haenszel W. Statistical aspects of the analysis of data from retrospective studies of disease. J Natl Cancer Inst. 1959;22(4):719–48.
DerSimonian R, Laird N. Meta-analysis in clinical trials. Control Clin Trials. 1986;7(3):177–88.
Lau J, Ioannidis JP, Schmid CH. Quantitative synthesis in systematic reviews. Ann Intern Med. 1997;127(9):820–6.
Higgins JP, Thompson SG, Deeks JJ, Altman DG. Measuring inconsistency in meta-analyses. BMJ. 2003;327(7414):557–60.
Egger M, Davey Smith G, Schneider M, Minder C. Bias in meta-analysis detected by a simple, graphical test. BMJ. 1997;315(7109):629–34.
Podder H, Podbielski J, Hussein I, Katz S, Van Buren C, Kahan BD. Sirolimus improves the two-year outcome of renal allografts in African-American patients. Transpl Int. 2001;14(3):135–42.
Johnson RW, Kreis H, Oberbauer R, Brattstrom C, Claesson K, Eris J. Sirolimus allows early cyclosporine withdrawal in renal transplantation resulting in improved renal function and lower blood pressure. Transplantation. 2001;72(5):777–86.
Baboolal K. A phase III prospective, randomized study to evaluate concentration-controlled sirolimus (rapamune) with cyclosporine dose minimization or elimination at six months in de novo renal allograft recipients. Transplantation. 2003;75(8):1404–8.
Redmond A, Barnes S. Acute renal failure: recognition and treatment in ward patients. Nurs Stand. 2004;18(22):46–53.
Sousa M, Linhares M, Salzedas-Netto A, et al. Risk factors of pancreatic graft loss and death of receptor after simultaneous pancreas/kidney transplantation. Paper presented at: Transplantation proceedings, 2014.
de Mattos AM, Olyaei AJ, Bennett WM. Nephrotoxicity of immunosuppressive drugs: long-term consequences and challenges for the future. Am J Kidney Dis. 2000;35(2):333–46.
Mulay AV, Hussain N, Fergusson D, Knoll GA. Calcineurin inhibitor withdrawal from sirolimus-based therapy in kidney transplantation: a systematic review of randomized trials. Am J Transplant. 2005;5(7):1748–56.
Frohn C, Fricke L, Puchta JC, Kirchner H. The effect of HLA-C matching on acute renal transplant rejection. Nephrol Dial Transplant. 2001;16(2):355–60.
Halloran PF. Immunosuppressive drugs for kidney transplantation. N Engl J Med. 2004;351(26):2715–29.
Andreoni KA, Brayman KL, Guidinger MK, Sommers CM, Sung RS. Kidney and pancreas transplantation in the United States, 1996–2005. Am J Transplant. 2007;7(5 Pt 2):1359–75.
Wiederrecht G, Lam E, Hung S, Martin M, Sigal N. The mechanism of action of FK-506 and cyclosporin A. Ann N Y Acad Sci. 1993;696:9–19.
Kasiske BL, Chakkera HA, Louis TA, Ma JZ. A meta-analysis of immunosuppression withdrawal trials in renal transplantation. J Am Soc Nephrol. 2000;11(10):1910–7.
Basadonna GP, Matas AJ, Gillingham KJ, et al. Early versus late acute renal allograft rejection: impact on chronic rejection. Transplantation. 1993;55(5):993–5.
Massy ZA, Guijarro C, Wiederkehr MR, Ma JZ, Kasiske BL. Chronic renal allograft rejection: immunologic and nonimmunologic risk factors. Kidney Int. 1996;49(2):518–24.
Acknowledgments
We would like to thank all respondents of the study and all the people who give the help for this study.
Conflict of interest
The authors have declared that no competing interests exist.
Author information
Authors and Affiliations
Corresponding author
Additional information
H. Bai and Y. Qian are co-first authors.
About this article
Cite this article
Bai, H., Qian, Y., Shi, B. et al. Effectiveness and safety of calcineurin inhibitor withdrawal in kidney transplantation: a meta-analysis of randomized controlled trials. Clin Exp Nephrol 19, 1189–1198 (2015). https://doi.org/10.1007/s10157-015-1109-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10157-015-1109-z