Abstract
Objectives
Only a few prospective studies have been conducted to examine the efficacy and safety of systemic chemotherapy for patients with pulmonary sarcomatoid carcinomas (PSCs). There is, thus, a crucial need to develop novel treatment strategies for this rare tumor.
Patients and methods
Chemotherapy-naïve patients with histologically confirmed PSCs were assigned to receive either carboplatin/paclitaxel alone (CP) or with bevacizumab (CPB) followed by bevacizumab maintenance. The primary endpoint was overall response rate (ORR). Secondary endpoints included overall survival (OS), progression-free survival (PFS), and safety.
Results
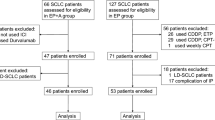
This study was closed before accumulating the expected number of cases due to slow patient accrual. Eventually, 16 patients were enrolled. The ORR was 25.0% and disease control rate was 56.3%. CPB was administered in all four patients with an objective response [partial response (PR)]; among the four PR cases, two patients had pleomorphic carcinoma, and two had carcinosarcoma. Median PFS and median survival time (MST) in all the enrolled patients were 2.6 months and 8.8 months, respectively. Median PFS was 1.2 months in the CP group and 4.2 months in the CPB group. In addition, MST was 7.9 months in the CP group and 11.2 months in the CPB group. Hematological and non-hematological adverse events were common and reversible, although ileus (grade 4) and nasal bleeding (grade 3) occurred in one case each in the CPB group.
Conclusions
CPB might be effective as first-line treatment for PSCs. Further study is warranted to clarify the role of cytotoxic chemotherapy for this rare and aggressive tumor.
Clinical trials registration
University Hospital Medical Information Network (UMIN) Clinical Trial Registry (UMIN000008707).



Similar content being viewed by others
References
Brambilla E, Travis WD, Colby TV et al (2001) The new World Health Organization classification of lung tumours. Eur Respir J 18(6):1059–1068. https://doi.org/10.1183/09031936.01.00275301
Travis WD, Brambilla E, Nicholson AG et al (2015) The 2015 World Health Organization Classification of Lung Tumors: impact of genetic, clinical and radiologic advances since the 2004 Classification. J Thorac Oncol 10(9):1243–1260. https://doi.org/10.1097/JTO.0000000000000630
Rossi G, Cavazza A, Sturm N et al (2003) Pulmonary carcinomas with pleomorphic, sarcomatoid, or sarcomatous elements: a clinicopathologic and immunohistochemical study of 75 cases. Am J Surg Pathol 27(3):311–324. https://doi.org/10.1097/00000478-200303000-00004
Martin LW, Correa AM, Ordonez NG et al (2007) Sarcomatoid carcinoma of the lung: a predictor of poor prognosis. Ann Thorac Surg 84(3):973–980. https://doi.org/10.1016/j.athoracsur.2007.03.099
Ito K, Oizumi S, Fukumoto S et al (2010) Clinical characteristics of pleomorphic carcinoma of the lung. Lung Cancer 68(2):204–210. https://doi.org/10.1016/j.lungcan.2009.06.002
Ung M, Rouquette I, Filleron T et al (2016) Characteristics and clinical outcomes of sarcomatoid carcinoma of the lung. Clin Lung Cancer 17(5):391–397. https://doi.org/10.1016/j.cllc.2016.03.001
Lococo F, Rapicetta C, Cardillo G et al (2017) Pathologic findings and long-term results after surgical treatment for pulmonary sarcomatoid tumors: a multicenter analysis. Ann Thorac Surg 103(4):1142–1150. https://doi.org/10.1016/j.athoracsur.2016.08.114
Bae HM, Min HS, Lee SH et al (2007) Palliative chemotherapy for pulmonary pleomorphic carcinoma. Lung Cancer 58(1):112–115. https://doi.org/10.1016/j.lungcan.2007.05.006
Hong JY, Choi MK, Uhm JE et al (2009) The role of palliative chemotherapy for advanced pulmonary pleomorphic carcinoma. Med Oncol 26(3):287–291. https://doi.org/10.1007/s12032-008-9117-4
Yendamuri S, Caty L, Pine M et al (2012) Outcomes of sarcomatoid carcinoma of the lung: a surveillance, epidemiology, and end results database analysis. Surgery 152(3):397–402. https://doi.org/10.1016/j.surg.2012.05.007
Tamura T, Ohara G, Kagohashi K et al (2016) Pemetrexed and bevacizumab-containing chemotherapy for pleomorphic carcinoma of the lung. Mol Clin Oncol 4(4):616–618. https://doi.org/10.3892/mco.2016.758
Sakata S, Saeki S, Sato R et al (2017) Long-term complete response to carboplatin plus paclitaxel combined with bevacizumab in a patient with metastatic spindle cell carcinoma. Respir Investig 55(6):372–375. https://doi.org/10.1016/j.resinv.2017.08.005
Vieira T, Girard N, Ung M et al (2013) Efficacy of first-line chemotherapy in patients with advanced lung sarcomatoid carcinoma. J Thorac Oncol 8(12):1574–1577. https://doi.org/10.1097/01.JTO.0000437008.00554.90
Huang SY, Shen SJ, Li XY (2013) Pulmonary sarcomatoid carcinoma: a clinicopathologic study and prognostic analysis of 51 cases. World J Surg Oncol 11:252. https://doi.org/10.1186/1477-7819-11-252
Chaft JE, Sima CS, Ginsberg MS et al (2012) Clinical outcomes with perioperative chemotherapy in sarcomatoid carcinomas of the lung. J Thorac Oncol 7(9):1400–1405. https://doi.org/10.1097/JTO.0b013e3182614856
Velcheti V, Rimm DL, Schalper KA (2013) Sarcomatoid lung carcinomas show high levels of programmed death ligand-1 (PD-L1). J Thorac Oncol 8(6):803–805. https://doi.org/10.1097/JTO.0b013e318292be18
Vieira T, Antoine M, Hamard C et al (2016) Sarcomatoid lung carcinomas show high levels of programmed death ligand-1 (PD-L1) and strong immune-cell infiltration by TCD3 cells and macrophages. Lung Cancer 98:51–58. https://doi.org/10.1016/j.lungcan.2016.05.013
Chen J, He Q, Liu J et al (2018) CD8+ tumor-infiltrating lymphocytes as a novel prognostic biomarker in lung sarcomatoid carcinoma, a rare subtype of lung cancer. Cancer Manag Res 10:3505–3511. https://doi.org/10.2147/CMAR.S169074
Domblides C, Leroy K, Monnet I et al (2020) Efficacy of immune checkpoint inhibitors in lung sarcomatoid carcinoma. J Thorac Oncol 15(5):860–866. https://doi.org/10.1016/j.jtho.2020.01.014
Mayenga M, Assie JB, Monnet I et al (2020) Durable responses to immunotherapy of non-small cell lung cancers harboring MET exon-14-skipping mutation: a series of 6 cases. Lung Cancer 150:21–25. https://doi.org/10.1016/j.lungcan.2020.09.008
Acknowledgements
We thank all participating patients, their families, and the site investigators. Authors also thank Dr. Satoshi Morita for his technical support for statistical considerations.
Funding
This study was supported by a Grant-in-Aid for Scientific Research (JSPS KAKENHI Grant Number 15K09164).
Author information
Authors and Affiliations
Consortia
Contributions
SO was the primary investigator and contributed to study design. KT, TH, MT, NM, RH, SW, TA, MK, TF, RN, EK, YT, and TT were involved in patient accrual and data collection. KK and HDA chaired the study group and contributed to the administrative management of the study. SO performed data analysis and interpretation. The first draft of the manuscript was written by SO. All authors contributed to subsequent editing, critical review, and final approval to submit the report for publication.
Corresponding author
Ethics declarations
Conflict of interest
SO received honoraria from AstraZeneca and Eli Lilly and research funding from Abbvie, Amgen, AstraZeneca, Bristol-Myers Squibb, Chugai Pharmaceutical, Kissei Pharmaceutical, Ono Pharmaceutical, Pfizer, Merck Biopharma, Sanofi, Taiho Pharmaceutical, and Takeda Pharmaceutical; MT received honoraria from AstraZeneca, Chugai Pharmaceutical, Eli Lilly, MSD, and Ono Pharmaceutical and research funding from AstraZeneca; SW received honoraria from AstraZeneca, Boehringer Ingelheim, Bristol-Myers Squibb, Chugai Pharmaceutical, Eli Lilly, MSD, Novartis, Ono Pharmaceutical, Pfizer, and Taiho Pharmaceutical and research funding from AstraZeneca and Boehringer Ingelheim; TA received honoraria from AstraZeneca, Boehringer Ingelheim, Chugai Pharmaceutical, Eli Lilly, MSD, Ono Pharmaceutical, Taiho Pharmaceutical, and Takeda Pharmaceutical; RN received honoraria from AstraZeneca and Novartis; YT received honoraria from AstraZeneca, Boehringer Ingelheim, Bristol-Myers Squibb, Chugai Pharmaceutical, Mochida Pharmaceutical, Nippon Kayaku, Ono Pharmaceutical, and Taiho Pharmaceutical; KK received honoraria from AstraZeneca; All other authors declare that they have no conflicts of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
About this article
Cite this article
Oizumi, S., Takamura, K., Harada, T. et al. Phase II study of carboplatin–paclitaxel alone or with bevacizumab in advanced sarcomatoid carcinoma of the lung: HOT1201/NEJ024. Int J Clin Oncol 27, 676–683 (2022). https://doi.org/10.1007/s10147-021-02113-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10147-021-02113-5