Abstract
Background
Little information has been published on the use of tyrosine kinase inhibitors for treatment of patients undergoing hemodialysis (HD). We investigated the efficacy and safety of sorafenib for metastatic renal cell carcinoma (mRCC) patients undergoing HD.
Methods
Twenty patients undergoing HD were treated with sorafenib as first-line therapy for mRCC at our hospital between April 2008 and August 2014. Patient medical records were retrospectively reviewed to evaluate the response to sorafenib and treatment-related toxicity.
Results
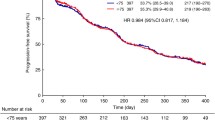
Fifteen and 5 patients were classified in the intermediate and poor risk groups, respectively, of the Memorial Sloan–Kettering Cancer Center risk model. Eighteen patients had 3 or more metastatic lesions, and 7 patients had metastases in 2 or more organs. Of 16 patients who had previously undergone nephrectomy, 8 were pathologically diagnosed with non-clear-cell carcinoma. The median duration of sorafenib therapy was 4.7 months. Sorafenib was discontinued owing to progressing disease for 15 patients and because of serious adverse events (AE) (≥grade 3) for 4 patients, i.e. subarachnoid hemorrhage, cerebral hemorrhage, sepsis, and syncope for 1 patient each. Median time to progression was 6.3 months, and median overall survival was 14.2 months.
Conclusions
In this study, many patients had unfavorable clinical features, for example poor risk classification and metastases in multiple organs. Although sorafenib treatment of HD patients seems feasible, careful monitoring is needed because of the tendency for a high incidence of serious AE, even when a reduced dose is administered.



Similar content being viewed by others
References
Ishikawa I, Saito Y, Shikura N et al (1990) Ten-year prospective study on the development of renal cell carcinoma in dialysis patients. Am J Kidney Dis 16:452–458
Terasawa Y, Suzuki Y, Morita M et al (1994) Ultrasonic diagnosis of renal cell carcinoma in hemodialysis patients. J Urol 152:846–851
Denton MD, Magee CC, Ovuworie C et al (2002) Prevalence of renal cell carcinoma in patients with ESRD pre-transplantation: a pathologic analysis. Kidney Int 61:2201–2209
Schwarz A, Vatandaslar S, Merkel S et al (2007) Renal cell carcinoma in transplant recipients with acquired cystic kidney disease. Clin J Am Soc Nephrol 2:750–756
Neuzillet Y, Tillou X, Mathieu R et al (2011) Renal cell carcinoma (RCC) in patients with end-stage renal disease exhibits many favourable clinical, pathologic, and outcome features compared with RCC in the general population. Eur Urol 60:366–373
Nouh MA, Kuroda N, Yamashita M et al (2010) Renal cell carcinoma in patients with end-stage renal disease: relationship between histological type and duration of dialysis. BJU Int 105:620–627
Syrios J, Kechagias G, Tsavaris N (2013) Treatment of patients with metastatic renal cell carcinoma undergoing hemodialysis: case report of two patients and short literature review. BMC Nephrol 14:84
Escudier B, Eisen T, Stadler WM et al (2007) Sorafenib in advanced clear-cell renal-cell carcinoma. N Engl J Med 356:125–134
Motzer RJ, Bacik J, Murphy BA et al (2002) Interferon-alfa as a comparative treatment for clinical trials of new therapies against advanced renal cell carcinoma. J Clin Oncol 20:289–296
Strumberg D, Richly H, Hilger RA et al (2005) Phase I clinical and pharmacokinetic study of the Novel Raf kinase and vascular endothelial growth factor receptor inhibitor BAY 43-9006 in patients with advanced refractory solid tumors. J Clin Oncol 23:965–972
Clark JW, Eder JP, Ryan D et al (2005) Safety and pharmacokinetics of the dual action Raf kinase and vascular endothelial growth factor receptor inhibitor, BAY 43-9006, in patients with advanced, refractory solid tumors. Clin Cancer Res 11:5472–5480
Moore M, Hirte HW, Siu L et al (2005) Phase I study to determine the safety and pharmacokinetics of the novel Raf kinase and VEGFR inhibitor BAY 43-9006, administered for 28 days on/7 days off in patients with advanced, refractory solid tumors. Ann Oncol 16:1688–1694
Ruppin S, Protzel C, Klebingat KJ et al (2009) Successful sorafenib treatment for metastatic renal cell carcinoma in a case with chronic renal failure. Eur Urol 55:986–988 (quiz 988)
Shinsako K, Mizuno T, Terada T et al (2010) Tolerable sorafenib therapy for a renal cell carcinoma patient with hemodialysis: a case study. Int J Clin Oncol 15:512–514
Rey PM, Villavicencio H (2008) Sorafenib: tolerance in patients on chronic hemodialysis: a single-center experience. Oncology 74:245–246
Masini C, Sabbatini R, Porta C et al (2012) Use of tyrosine kinase inhibitors in patients with metastatic kidney cancer receiving haemodialysis: a retrospective Italian survey. BJU Int 110:692–698
Kennoki T, Kondo T, Kimata N et al (2011) Clinical results and pharmacokinetics of sorafenib in chronic hemodialysis patients with metastatic renal cell carcinoma in a single center. Jpn J Clin Oncol 41:647–655
Saito K, Tatokoro M, Fujii Y et al (2009) Impact of C-reactive protein kinetics on survival of patients with metastatic renal cell carcinoma. Eur Urol 55:1145–1153
Heng DY, Choueiri TK, Rini BI et al (2014) Outcomes of patients with metastatic renal cell carcinoma that do not meet eligibility criteria for clinical trials. Ann Oncol 25:149–154
Motzer RJ, Hutson TE, Tomczak P et al (2009) Overall survival and updated results for sunitinib compared with interferon alfa in patients with metastatic renal cell carcinoma. J Clin Oncol 27:3584–3590
Escudier B, Szczylik C, Hutson TE et al (2009) Randomized phase II trial of first-line treatment with sorafenib versus interferon Alfa-2a in patients with metastatic renal cell carcinoma. J Clin Oncol 27:1280–1289
Akaza H, Tsukamoto T, Murai M et al (2007) Phase II study to investigate the efficacy, safety, and pharmacokinetics of sorafenib in Japanese patients with advanced renal cell carcinoma. Jpn J Clin Oncol 37:755–762
Uemura H, Shinohara N, Yuasa T et al (2010) A phase II study of sunitinib in Japanese patients with metastatic renal cell carcinoma: insights into the treatment, efficacy and safety. Jpn J Clin Oncol 40:194–202
Procopio G, Bellmunt J, Dutcher J et al (2013) Sorafenib tolerability in elderly patients with advanced renal cell carcinoma: results from a large pooled analysis. Br J Cancer 108:311–318
Hutson TE, Bellmunt J, Porta C et al (2010) Long-term safety of sorafenib in advanced renal cell carcinoma: follow-up of patients from phase III TARGET. Eur J Cancer 46:2432–2440
Stadler WM, Figlin RA, McDermott DF et al (2010) Safety and efficacy results of the advanced renal cell carcinoma sorafenib expanded access program in North America. Cancer 116:1272–1280
Hutson TE, Lesovoy V, Al-Shukri S et al (2013) Axitinib vs. sorafenib as first-line therapy in patients with metastatic renal-cell carcinoma: a randomised open-label phase 3 trial. Lancet Oncol 14:1287–1294
Motzer RJ, Nosov D, Eisen T et al (2013) Tivozanib versus sorafenib as initial targeted therapy for patients with metastatic renal cell carcinoma: results from a phase III trial. J Clin Oncol 31:3791–3799
Harnett JD, Foley RN, Kent GM et al (1995) Congestive heart failure in dialysis patients: prevalence, incidence, prognosis and risk factors. Kidney Int 47:884–890
Agarwal R (2005) Hypertension and survival in chronic hemodialysis patients–past lessons and future opportunities. Kidney Int 67:1–13
Longenecker JC, Coresh J, Powe NR et al (2002) Traditional cardiovascular disease risk factors in dialysis patients compared with the general population: the CHOICE Study. J Am Soc Nephrol 13:1918–1927
Yasuda Y, Saito K, Yuasa T et al (2013) Prognostic impact of pretreatment C-reactive protein for patients with metastatic renal cell carcinoma treated with tyrosine kinase inhibitors. Int J Clin Oncol 18(5):884–889
Omae K, Kondo T, Tanabe K (2014) High preoperative C-reactive protein values predict poor survival in patients on chronic hemodialysis undergoing nephrectomy for renal cancer. Urol Oncol 33(2):67.e9–67.e13
Miki S, Iwano M, Miki Y et al (1989) Interleukin-6 (IL-6) functions as an in vitro autocrine growth factor in renal cell carcinomas. FEBS Lett 250:607–610
Koo AS, Armstrong C, Bochner B et al (1992) Interleukin-6 and renal cell cancer: production, regulation, and growth effects. Cancer Immunol Immunother 35:97–105
Trédan O, Galmarini CM, Patel K et al (2007) Drug resistance and the solid tumor microenvironment. J Natl Cancer Inst 99:1441–1454
Mancino A, Lawrence T (2010) Nuclear factor-kappaB and tumor-associated macrophages. Clin Cancer Res 16:784–789
Hanahan D, Coussens LM (2012) Accessories to the crime: functions of cells recruited to the tumor microenvironment. Cancer Cell 21:309–322
Filiopoulos V, Vlassopoulos D (2009) Inflammatory syndrome in chronic kidney disease: pathogenesis and influence on outcomes. Inflamm Allergy Drug Targets 8:369–382
Acknowledgments
The authors thank Noriko Hata for secretarial assistance.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
The authors declare that they have no conflict of interest.
About this article
Cite this article
Omae, K., Kondo, T., Kennoki, T. et al. Efficacy and safety of sorafenib for treatment of Japanese metastatic renal cell carcinoma patients undergoing hemodialysis. Int J Clin Oncol 21, 126–132 (2016). https://doi.org/10.1007/s10147-015-0871-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10147-015-0871-y