Abstract
Early and reliable prediction of shunt-dependent hydrocephalus (SDHC) after aneurysmal subarachnoid hemorrhage (aSAH) may decrease the duration of in-hospital stay and reduce the risk of catheter-associated meningitis. Machine learning (ML) may improve predictions of SDHC in comparison to traditional non-ML methods. ML models were trained for CHESS and SDASH and two combined individual feature sets with clinical, radiographic, and laboratory variables. Seven different algorithms were used including three types of generalized linear models (GLM) as well as a tree boosting (CatBoost) algorithm, a Naive Bayes (NB) classifier, and a multilayer perceptron (MLP) artificial neural net. The discrimination of the area under the curve (AUC) was classified (0.7 ≤ AUC < 0.8, acceptable; 0.8 ≤ AUC < 0.9, excellent; AUC ≥ 0.9, outstanding). Of the 292 patients included with aSAH, 28.8% (n = 84) developed SDHC. Non-ML-based prediction of SDHC produced an acceptable performance with AUC values of 0.77 (CHESS) and 0.78 (SDASH). Using combined feature sets with more complex variables included than those incorporated in the scores, the ML models NB and MLP reached excellent performances, with an AUC of 0.80, respectively. After adding the amount of CSF drained within the first 14 days as a late feature to ML-based prediction, excellent performances were reached in the MLP (AUC 0.81), NB (AUC 0.80), and tree boosting model (AUC 0.81). ML models may enable clinicians to reliably predict the risk of SDHC after aSAH based exclusively on admission data. Future ML models may help optimize the management of SDHC in aSAH by avoiding delays in clinical decision-making.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Shunt-dependent hydrocephalus (SDHC) is common after aneurysmal subarachnoid hemorrhage (aSAH) with rates between 7 and 67% [1,2,3,4,5]. Based on different clinical and radiological factors, various scoring systems have been developed to predict the risk of SDHC after aSAH [6, 7]. The two validated scores with the best performance in SDHC prediction are the “Chronic Hydrocephalus Ensuing from SAH Score” (CHESS) and the “Shunt Dependency in aSAH Score” (SDASH) [2, 8, 9].
A recent trend in clinical prediction modelling is the introduction of machine learning (ML) algorithms allowing for the inclusion of a variety of additional complex variables [10, 11]. For example, in aSAH and stroke, such ML algorithms were shown to improve outcome prediction [12,13,14,15]. Even though ML-based prediction models have entered nearly all fields of medicine, there is limited evidence on direct comparisons between non-ML and ML-based prediction [13]. ML methods have been criticized for their supposed lack of transparency and confirmability of the impact of variables used, especially in the case of more modern ML techniques, such as deep neuronal networks [25]. Also, there is some debate on which data may be best suited to further enhance the predictive capabilities of ML models. For example, given that any prediction of an event should be done as early as possible in order to adjust care decisions and potentially influence outcomes, there is an inherent need to feed ML models with early variables, such as admission data, rather than variables from later stages of clinical management, such as, in the case of SDHC, the amount of cerebrospinal fluid (CSF) drained during the first 14 days after aSAH [16, 17].
We therefore performed a study to compare the performance of the non-ML-based scores CHESS and SDASH to different ML models in predicting SDHC after aSAH. We also compared various ML models among each other, some relying exclusively on variables available on admission, others adding the amount of 14-day CSF volumes as a late variable.
Methods and materials
Patient management and data collection
We retrospectively evaluated prospectively collected clinical, radiographic, and laboratory data of 408 consecutive patients hospitalized with aSAH at our department between January 1, 2009, and December 31, 2015. Local ethics committee approval was obtained (EA1/291/14). The only inclusion criterion was aSAH confirmed by CT or xanthochromic cerebrospinal fluid on admission. If the hemorrhage was due to trauma or we were not able to identify an aneurysmal source of the bleeding, patients were excluded. The clinical condition was assessed based on the Hunt & Hess grading system [18]. Radiographic parameters were assessed based on the admission CT. Semi-quantitative radiographic grading of the thickness of the subarachnoidal blood clot was performed according to the BNI grading system [19]. Other radiographic parameters on CT were as follows: the presence of intraventricular hemorrhage (IVH), intracerebral hemorrhage (ICH), early infarction (EI), posterior location of the aneurysm (post. loc. AY), and acute hydrocephalus (aHP). aHP was defined according to Bae and colleagues based on third ventricle enlargement and periventricular low density on CT within 72 h of the aSAH, combined with mental deterioration or impaired consciousness or memory, gait disturbance, and urinary incontinence [20]. The following laboratory serum parameters were assessed on admission: creatinine in mg/dl, glucose in mg/dl, and C-reactive protein (CRP) in mg/l.
For aSAH management, early aneurysm occlusion was attempted within 48 h after aSAH, according to previously published guidelines [21, 22]. For aHP management, the standard protocol established by Jabbarli and colleagues was followed [9]. In the acute phase, an external ventricular drain (EVD) or lumbar drain (LD) was placed. At a later stage, a ventriculoperitoneal shunt was placed if patients could not be weaned off the external ventricular drain (EVD) or lumbar drain (LD) within 14 days.
Outcome assessment
Shunt dependency was examined in patients that survived the index hospital stay. It was assessed based on patient files from routine control visits 6–12 months after aSAH. If no routine follow-up data was available, data were obtained by telephone interview.
Scores
For the assessment of the CHESS score to predict SDHC, the following variables were included: Hunt & Hess grade ≥ 4 (1 point), aneurysm location in the posterior circulation (1 point), aHP (4 points), IVH (1 point), and early cerebral infarction on CT (1 point) [9]. The BNI score was established according to the thickness of the subarachnoid blood clot perpendicular to a cistern or fissure (1: no visible SAH; 2: ≤ 5 mm; 3: 6–10 mm; 4: 11–15 mm; 5: 16–20 mm) [19]. The SDASH ranges from 1 to 4 points and was calculated as recently described [2]. It includes the following variables: presence of aHP (2 points), BNI score ≥ 3 (1 point), and Hunt & Hess grade ≥ 4 (1 point). SDASH and CHESS scoring systems were compared using a conventional area under the curve (AUC) calculation as previously described [2].
Feature selection
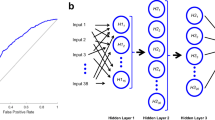
Among 408 patients with aSAH in the data base, 116 patients had to be excluded as they did not survive the initial phase of the disease or based on missing information on SDHC. We therefore included 292 patients in this study. Among those, only a few variables were missing (age: 0.7%, ICH: 0.3%, aneurysm location: 0.8%). Mean/mode imputation was used in each fold to impute missing values (see section “Model training and validation”). Input features were included if a ratio of at least 1 to 4 for binary variables (absence/presence) was reached. Features that were shown to be associated with SDHC were included (Table 1). This resulted in the following variables being used in the analyses: age, Hunt & Hess grade, BNI grade, presence of aHP, presence of ICH, presence of IVH, serum levels of CRP, and glucose on admission. As a further feature, we used the amount of cerebrospinal fluid (CSF) drainage (in ml) over the first 14 days after aSAH, as it is an established risk factor of SDHC after aSAH and as it represents a “late feature” in a separate run (Fig. 1) [8, 23]. Categorical features with at least three categories were transformed into binary features if they had too few instances per category. The following dichotomizations were used: radiologically defined ICH “yes”/ “no,” and aneurysm location in the anterior circulation “yes”/ “no.” Thus, all variables were either binary or continuous.
Prediction paradigms for shunt dependency after subarachnoid hemorrhage. Abbreviations: aHP, presence of acute hydrocephalus; AY, aneurysm; BNI, Barrow Neurological Institute scale for the thickness of subarachnoid hemorrhage; CC, conventional calculation; CRP, C-reactive protein; csf, cerebrospinal fluid; H&H, Hunt & Hess scale; ICH, intracerebral hemorrhage; IVH, intraventricular hemorrhage; tML, traditional machine learning with generalized linear models; Least absolute shrinkage and selection operator regression (LASSO) and ElasticNET; mML, modern machine learning with tree boosting (CatBoost), Naive Bayes (NV), and multilayer perceptron (MLP) neuronal network models
Model selection
Additional to the conventional area under the receiver operating characteristic curve calculation, ML models were trained for each score (run 1, CHESS; run 2, SDASH). A combined model with a set (run 3) of individual features available on admission was designed to include all clinically relevant (age, GCS) and laboratory parameters (glucose, CRP) as well as radiographically important parameters (aHP, IVH, ICH) on admission independent from SDASH and CHESS model calculation was trained. The additional run 4 included the features of run 3 and the amount of CSF drained during the first 14 days after aSAH (Fig. 1, Table 1).
Machine learning framework
To train ML models, a publicly available ML framework for predictive modelling was used utilizing standard ML libraries in Python. Its code can be accessed on GitHub (https://github.com/prediction2020/explainable-predictive-models), and details on the technical implementation can be found in previous open-access publications [14, 24]. A supervised ML approach was trained on all clinical parameters and scores listed in Table 1 to predict SDHC. Our dataset contained 84 positive (SDHC present) and 208 negative (no SDHC present) cases. This provided a reasonably balanced dataset and thus refrained from a sub-sampling approach that would have limited the amount of data for model training.
Applied algorithms
We used six different algorithms for all RUN selections. Three types of generalized linear models (GLM) represented traditional ML models: a plain GLM, an L1 regularized GLM (equivalent to LASSO logistic regression—LASSO), and a GLM elastic net with an additional L2 regularization (ElasticNET). We also included more modern ML models like a tree boosting algorithm (CatBoost), a Naive Bayes (NB) classifier, and a type of artificial neural network, the multilayer perceptron (MLP). For run 3 (“feature combination”) and run 4 (“feature combination with 14-day CSF drainage”), feature importance ratings were calculated for all algorithms using SHapley Additive exPlanations (SHAP) values (Table 2, Figs. 2 and 3). To minimize potential confounding effects on predictive performance, the variance inflation factor (VIF) was applied to assess multicollinearity for all features [25].
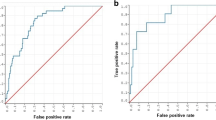
Performance and feature rating for the combined feature set for features available early in the clinical course of aneurysmal subarachnoid hemorrhage (run 3). A GLM, LASSO, and tree boosting (CatBoost) models reached the highest AUC in training (0.84/0.88) and MLP and NB in the test (0.80, respectively). A large difference between training and test was seen in tree boosting which may be an indication of overfitting. B GCS, Hunt & Hess, and the presence of early hydrocephalus were defined as the most important factors in the majority of models. Abbreviations: aHP, presence of acute hydrocephalus; BNI, Barrow Neurological Institute scale for thickness of subarachnoid hemorrhage; CRP, C-reactive protein early; GLM, generalized linear model; HCP, presence of early hydrocephalus; H&H, Hunt & Hess scale; ICH, intracerebral hemorrhage; IVH, intraventricular hemorrhage; MLP, multilayer perceptron; NB, Naive Bayes
Performance and feature rating for the combined feature set including 14-day cerebrospinal fluid drainage (run 4). A The tree boosting (CatBoost) model reached the highest AUC in training (0.89) and so did MLP and tree boosting in the test (0.81, respectively). Also, a larger difference between the training and test set in the tree boosting model was noted which may be indicative of overfitting. B The presence of early hydrocephalus, GCS, Hunt & Hess, and the amount of cerebrospinal fluid drainage within the first 14 days after the hemorrhage were defined as the most important factors in the majority of models. IVH, BNI, creatinine, and glucose were rated next important with varying extent throughout the models. Abbreviations: aHP, presence of acute hydrocephalus; BNI, Barrow Neurological Institute scale for thickness of subarachnoid hemorrhage; CRP, C-reactive protein; 14d CSF, the volume of drained cerebrospinal fluid within the first 14 days after the hemorrhage; GLM, generalized linear model; HCP, presence of early hydrocephalus; H&H, Hunt & Hess scale; ICH, intracerebral hemorrhage; IVH, intraventricular hemorrhage; MLP, multilayer perceptron; NB, Naive Bayes
Model training and validation
All data were split randomly into training and test sets at a 4:1 ratio. On both sets, we performed mean/mode imputation and feature scaling using zero-mean unit variance normalization based on the training set. A tenfold cross-validation was used for hyperparameter tuning. This process was repeated in 50 shuffles.
Performance assessment
Receiver operating characteristic (ROC) analysis was used to test the model performance on the test set by measuring the AUC. Performance was also assessed based on accuracy, average class accuracy, precision, recall, F1 score, negative predictive value, and specificity. The Brier score was used to quantify model calibration. According to Hosmer and Lemeshow, we used the following classification system for AUC: 0.7 ≤ AUC < 0.8, “acceptable”; 0.8 ≤ AUC < 0.9, “excellent”; AUC ≥ 0.9, “outstanding” [26, 27].
Interpretability assessment
To facilitate the comparability of feature ratings across models, we scaled the absolute values of the SHAP feature importance scores to the unit norm and, for each of the 50 shuffles, rescaled them to a range from 0 to 1 with their sum equal to 1. Each features’ means and standard deviations, calculated on the test sets over all shuffles, were reported as the final rating measures.
Results
Patient characteristics
Two hundred ninety-two patients with a median age of 53 years [IQR 45; 61] and a female to male ratio of 2:1 were included in the analysis. Of these, 28.8% (n = 84) developed SDHC. Clinical, radiographic, and laboratory data in the entire patient cohort are depicted in Table 1.
SDASH and CHESS score validity in conventional and ML-based outcome prediction
A conventional AUC calculation without ML algorithms revealed an AUC of 0.77 for CHESS and 0.78 for SDASH. To maintain comparability of scores in the ML-based models, CHESS and SDASH score values were introduced to the ML framework. Here, both scores reached acceptable prediction in training (AUC range: 0.77–0.78) and test (AUC range: 0.74–0.77) without remarkable differences between classic ML models (GLM, LASSO, ElasticNET) and more modern ML models (MLP, NB, CatBoost) (Table 2).
ML-based prediction with early parameters
The feature combination set with variables available on admission (run 3) revealed excellent predictive performance in training (AUC range: 0.82–0.88 [tree boost]) and reached acceptable performances in the GLM models and tree boosting model in the test set (AUC range: 0.78–0.79). In the test set, excellent performance was reached for the MLP and NB model (AUC 0.80, respectively (Table 2, Fig. 2A). The most important features were aHP, GCS, or Hunt & Hess H, and to varying extents in different models, BNI, IVH, CRP, and glucose (Fig. 2B).
ML-based prediction with early parameters and CSF volume drained within 14 days
The addition of the volume of CSF drained within the first 14 days as a late parameter after aSAH revealed further increases of the AUC to excellent performances in the test for the GLM (0.81), MLP (0.81), NB (0.80), and tree boosting (0.81) model (Table 2, Fig. 3A). When CSF was added to run 4, this variable gained importance being rated second or third highest after GCS or Hunt & Hess and HCP in most models (Fig. 3B).
Discussion
We present the first study on the prediction of SDHC after aSAH comparing traditional scoring systems to ML-based models and different ML-based models among each other. The main result of this analysis is that predictive performances of conventional scores SDASH and CHESS were reproduced with an ML-based analysis of these scores. A combination of features that were available already at admission and that were shown to be relevant for SDHC was introduced to an ML model approach, which resulted in an increase of predictive performance to an AUC of 0.80, which represents excellent prediction performance. Adding the late variable “14-day CSF volume” further improved the prediction of SDHC.
The fact that, in our study, the examined ML-based calculation of SDASH and CHESS scores reached predictive performances comparable to traditional and validated SDASH and CHESS scores suggests that ML approaches are valid and reproducible in predicting SDHC after aSAH [2, 8, 9]. This is of particular importance, as ML methods have previously been labeled “black-box-models” [28]. Especially more modern ML models, e.g., deep neural networks, have been criticized for being designed to identify and make use of associations between features rather than describe those associations in detail, which may limit the interpretability of the results [29, 30]. ML methods are therefore still predominantly used in the domain of predicting outcomes and complications, where their application is considered safe enough to allow them to learn continuously, as standard care is not affected while model updates can be carried out [31]. As ML models learn, it is important to identify the point at which their predictive performance surpasses validated non-ML prediction models. To this end, in our view, a continuous direct comparison between ML output and corresponding non-ML results is needed to prevent ML models from being perceived as black boxes simply producing different levels of AUC values. Critics of ML models in outcome prediction stress the lack of transparency as shown in a recent study [32]. Currently unanswered questions such as “What level of model transparency is required?” and “Do we understand the model outputs and whether they are unreliable and therefore not to be trusted?” may influence the future clinical utility of ML-based applications [32, 33]. This prompted us to compare ML outputs to well-established standards that are currently used in clinical practice. Moreover, our study specifically aims to identify and document factors contributing to potentially superior outputs of ML models and may therefore serve as a basis for future research.
It is somewhat surprising that this step of ML model validation is rarely done. A strength of the presented analysis is that it allows for a direct comparison of established, validated score calculations with traditional and more modern ML methods, which, in the case of predicting SDHC after aSAH, has never been done before. Only one other study exists examining ML methods in predicting SDHC after aSAH, albeit without comparison to non-ML methods [29]. In that study, on the basis of 368 patients and 32 variables, various ML algorithms were used, and the highest performance was reached using a distributed random forest model based on 21 variables, leading to a predictive performance with an AUC of 0.88 in validation and 0.85 in test with sixfold cross-validation [34]. That model included clinical and radiographic variables available on admission but also late variables, such as type of aneurysm treatment, ICU stay, time from symptom onset to treatment initiation, presence of fever, meningitis, treatment complications, and other infections. In our study, the ML-based feature combination (run 3) was solely based on features available at admission but also reached excellent prediction with the MLP and NB model.
Given that the prediction of any event becomes more valuable the sooner it is made, our finding that an ML algorithm based exclusively on admission data can predict SDHC with excellent performance is encouraging [17]. Of note, we did in fact observe a mild increase in predictive performance once the amount of 14-day CSF drainage was added to ML algorithms. In discussing the merits of late versus early variables, it is important to note that it hardly surprises that, in the case of our study, the late variable “14-day CSF volumes” led to improved prediction of SDHC, since this feature merely quantifies the failed attempt to wean patients off of EVD or LD, which is basically a symptom of SDHC rather than a risk factor. It is important to stress that any delay, in the case of our analysis a delay of 14 days in order to assess total CSF drainage, may outweigh the benefits of improved predictive performance, since it can increase the risk of complications. For example, delayed prediction of SDHC after aSAH may mean extended duration of external ventricular catheterization, which has been shown to be associated with increased rates of catheter-associated infection [35]. The finding that later variables may, to a certain degree, improve ML performance is in line with evidence from a recent study on ML-based prediction of discharge outcomes after aSAH, which describes the improvement of prediction once features from later phases of in-hospital stay are added to ML algorithms [16]. However, in that study, as well, the predictive performance of ML models including later features was not substantially better than ML algorithms based exclusively on admission data. In the case of SDHC prediction in aSAH, later variables that were shown to be associated with SDHC in non-ML prediction models were rebleeding and in-hospital complications such as meningitis, pneumonia, vasospasm, and ischemic stroke [3,4,5,6,7]. Whether the inclusion of these additional late factors may have improved our model remains elusive.
Another finding in our study was that ML models using only CHESS or SDASH data in the prediction of SDHC showed inferior performance when compared with ML models using variables more complex and comprehensive than CHESS or SDASH, such as the semi-quantitative measure BNI for the thickness of subarachnoidal blood, the presence of intracerebral hemorrhage, early infarction, and the age and laboratory serum parameters that were included. These additional factors were chosen, because their predictive value for SDHC was established in previous studies [2, 6, 8]. Our more multilayered ML models resulted in prediction improvements by 0.01 to 0.04 AUC points compared to CHESS and SDASH. It is reasonable to assume that future ML models with more multi-layered sets of admission features may further enhance predictive performances on admission. This could significantly influence treatment strategies, such as the timing of implantation of a ventriculoperitoneal shunt, or transfer to intensive care. Whether the addition of radiographic source data or other parameters previously shown to be associated with SDHC, e.g., cerebrospinal fluid markers such as total protein, red blood cell count, interleukin-6, or glucose, would have improved the predictive performance of the models used in our study remains to be tested [36]. The same goes for later variables previously shown to be associated with SDHC in non-ML prediction models, such as rebleeding and in-hospital complications such as meningitis, pneumonia, vasospasm, and ischemic stroke [37,38,39,40,41]. Whether the inclusion of such later factors would have impacted our models’ predictions is uncertain. Our data suggest that ML methods are suited for testing this, with more modern ML models like tree boosting, NB, and the MLP model generating better predictions, especially for the mixed parameter sets in run 3 and run 4.
The main strength of our study is the maintenance of transparency and comparability to existing models as well as between the different models, as our current approach examined established scoring systems with established statistical models in comparison to ML techniques. Nevertheless, the following limitations deserve to be mentioned. Our study was conducted at a single institution, which may limit the generalizability of our findings. Also, the retrospective and non-randomized nature of the analysis may introduce some selection bias and does not allow for causational interpretation of our results. Given the exclusion of non-survivors from our analysis, a bias towards patients with comparably good Hunt & Hess grades cannot be ruled out. However, this method is in line with previous studies on predictive factors for SDHC in aSAH and allows for comparability of our results to those reports [1,2,3,4,5]. A factor that may decrease comparability to other studies are variations in local treatment strategies concerning the timing or even the necessity to place a shunt after surgery. One may argue that true objective cut-offs for whether or when best to implant a shunt system do not exist. In our study, we adhered to an established protocol, as mentioned previously [2, 9]. However, the retrospective nature of our study has limits in terms of adherence to this protocol which we did not examine within this analysis. Given that shunt rates described in the literature range widely between 7 and 67%, our rate of 28.8% reflects a reasonable value, which is comparable to shunt rates published in studies using the same protocol [1,2,3,4,5, 9].
The fact that, in our study, the predictive capabilities of ML methods were comparable or better to the standard scores SDASH and CHESS suggests that future continual learning could one day enable the ML models to outmatch SDASH and CHESS. Once the superiority of ML prediction in SDHC after aSAH is reached, a randomized controlled trial will be necessary to validate these findings, before ML methods can become standard tools optimizing SDHC prediction in real time.
Conclusions
Our study is the first to present comparative data for SDHC prediction after aSAH for validated scores and state-of-the-art ML techniques. It suggests that ML models may enable clinicians to reliably predict the risk of SDHC based exclusively on admission data. Since early prediction of SDHC is key in aSAH management, future ML models could help optimize care for SDHC in aSAH by avoiding delays in clinical decision-making.
Data availability
Data is available upon reasonable request.
References
Chan M, Alaraj A, Calderon M et al (2009) Prediction of ventriculoperitoneal shunt dependency in patients with aneurysmal subarachnoid hemorrhage. J Neurosurg 110(1):44–49. https://doi.org/10.3171/2008.5.17560
Diesing D, Wolf S, Sommerfeld J, Sarrafzadeh A, Vajkoczy P, Dengler NF (2018) A novel score to predict shunt dependency after aneurysmal subarachnoid hemorrhage. J Neurosurg 128(5):1273–1279. https://doi.org/10.3171/2016.12.JNS162400
Erixon HO, Sorteberg A, Sorteberg W, Eide PK (2014) Predictors of shunt dependency after aneurysmal subarachnoid hemorrhage: results of a single-center clinical trial. Acta Neurochir (Wien) 156(11):2059–2069. https://doi.org/10.1007/s00701-014-2200-z
Lin CL, Kwan AL, Howng SL (1999) Acute hydrocephalus and chronic hydrocephalus with the need of postoperative shunting after aneurysmal subarachnoid hemorrhage. Kaohsiung J Med Sci 15(3):137–145
Kang P, Raya A, Zipfel GJ, Dhar R (2016) Factors associated with acute and chronic hydrocephalus in nonaneurysmal subarachnoid hemorrhage. Neurocrit Care 24(1):104–109. https://doi.org/10.1007/s12028-015-0152-7
Wilson CD, Safavi-Abbasi S, Sun H et al (2017) Meta-analysis and systematic review of risk factors for shunt dependency after aneurysmal subarachnoid hemorrhage. J Neurosurg 126(2):586–595. https://doi.org/10.3171/2015.11.JNS152094
Xie Z, Hu X, Zan X, Lin S, Li H, You C (2017) Predictors of shunt-dependent hydrocephalus after aneurysmal subarachnoid hemorrhage? A systematic review and meta-analysis. World Neurosurg 106:844–860 e6. https://doi.org/10.1016/j.wneu.2017.06.119
Garcia-Armengol R, Puyalto de Pablo P, Misis M et al (2021) Validation of shunt dependency prediction scores after aneurysmal spontaneous subarachnoid hemorrhage. Acta Neurochir (Wien). https://doi.org/10.1007/s00701-020-04688-w
Jabbarli R, Bohrer AM, Pierscianek D et al (2016) The CHESS score: a simple tool for early prediction of shunt dependency after aneurysmal subarachnoid hemorrhage. Eur J Neurol 23(5):912–918. https://doi.org/10.1111/ene.12962
Esteva A, Robicquet A, Ramsundar B et al (2019) A guide to deep learning in healthcare. Nat Med 25(1):24–29. https://doi.org/10.1038/s41591-018-0316-z
Beam AL, Kohane IS (2018) Big data and machine learning in health care. JAMA 319(13):1317–1318. https://doi.org/10.1001/jama.2017.18391
Dengler NF, Madai VI, Unteroberdorster M et al (2021) Outcome prediction in aneurysmal subarachnoid hemorrhage: a comparison of machine learning methods and established clinico-radiological scores. Neurosurg Rev 44(5):2837–2846. https://doi.org/10.1007/s10143-020-01453-6
Heo J, Yoon JG, Park H, Kim YD, Nam HS, Heo JH (2019) Machine learning-based model for prediction of outcomes in acute stroke. Stroke 50(5):1263–1265. https://doi.org/10.1161/STROKEAHA.118.024293
Mutke MA, Madai VI, Hilbert A et al (2022) Comparing poor and favorable outcome prediction with machine learning after mechanical thrombectomy in acute ischemic stroke. Front Neurol 13:737667. https://doi.org/10.3389/fneur.2022.737667
Martini ML, Neifert SN, Shuman WH et al (2022) Rescue therapy for vasospasm following aneurysmal subarachnoid hemorrhage: a propensity score-matched analysis with machine learning. J Neurosurg 136(1):134–147. https://doi.org/10.3171/2020.12.JNS203778
Maldaner N, Zeitlberger AM, Sosnova M et al (2021) Development of a complication- and treatment-aware prediction model for favorable functional outcome in aneurysmal subarachnoid hemorrhage based on machine learning. Neurosurgery 88(2):E150–E157. https://doi.org/10.1093/neuros/nyaa401
Escobar GJ, Turk BJ, Ragins A et al (2016) Piloting electronic medical record-based early detection of inpatient deterioration in community hospitals. J Hosp Med 11(Suppl 1):S18–S24. https://doi.org/10.1002/jhm.2652
Hunt WE, Hess RM (1968) Surgical risk as related to time of intervention in the repair of intracranial aneurysms. J Neurosurg 28(1):14–20. https://doi.org/10.3171/jns.1968.28.1.0014
Wilson DA, Nakaji P, Abla AA et al (2012) A simple and quantitative method to predict symptomatic vasospasm after subarachnoid hemorrhage based on computed tomography: beyond the Fisher scale. Neurosurgery 71(4):869–875. https://doi.org/10.1227/NEU.0b013e318267360f
Bae IS, Yi HJ, Choi KS, Chun HJ (2014) Comparison of incidence and risk factors for shunt-dependent hydrocephalus in aneurysmal subarachnoid hemorrhage patients. J Cerebrovasc Endovasc Neurosurg 16(2):78–84. https://doi.org/10.7461/jcen.2014.16.2.78
Steiner T, Juvela S, Unterberg A et al (2013) European Stroke Organization guidelines for the management of intracranial aneurysms and subarachnoid haemorrhage. Cerebrovasc Dis 35(2):93–112. https://doi.org/10.1159/000346087
Diringer MN, Bleck TP, Claude Hemphill J 3rd et al (2011) Critical care management of patients following aneurysmal subarachnoid hemorrhage: recommendations from the Neurocritical Care Society’s Multidisciplinary Consensus Conference. Neurocrit Care 15(2):211–240. https://doi.org/10.1007/s12028-011-9605-9
Tso MK, Ibrahim GM, Macdonald RL (2016) Predictors of shunt-dependent hydrocephalus following aneurysmal subarachnoid hemorrhage. World Neurosurg 86:226–232. https://doi.org/10.1016/j.wneu.2015.09.056
Zihni E, Madai VI, Livne M et al (2020) Opening the black box of artificial intelligence for clinical decision support: a study predicting stroke outcome. PLoS One 15(4):e0231166. https://doi.org/10.1371/journal.pone.0231166
Miles J (2014) Tolerance and variance inflation factor. Wiley Stats: Ref: Statistics Reference online. (American Cancer Society)
Hosmer DW, Lemeshow S (2000) Applied logistic regression. Wiley Online Books. 2nd edition, pp 156–164. https://doi.org/10.1002/0471722146
Mandrekar JN, Mandrekar SJ (2009) Biostatistics: a toolkit for exploration, validation, and interpretation of clinical data. J Thorac Oncol 4(12):1447–1449. https://doi.org/10.1097/JTO.0b013e3181c0a329
Rudin C (2019) Stop explaining black box machine learning models for high stakes decisions and use interpretable models instead. Nat Mach Intell 1(5):206–215. https://doi.org/10.1038/s42256-019-0048-x
Lakkaraju H, Bach SH, Jure L (2016) Interpretable decision sets: a joint framework for description and prediction. KDD 2016:1675–1684. https://doi.org/10.1145/2939672.2939874
Lipton ZC (2016) The mythos of model interpretability. arXiv https://doi.org/10.48550/arXiv.1606.03490
Lee CS, Lee AY (2020) Clinical applications of continual learning machine learning. Lancet Digit Health 2(6):e279–e281. https://doi.org/10.1016/S2589-7500(20)30102-3
Antes AL, Burrous S, Sisk BA et al (2021) Exploring perceptions of healthcare technologies enabled by artificial intelligence: an online, scenario-based survey. BMC Med Inform Decis Mak 21(1):221
Wiens J, Saria S, Sendak M, Ghassemi M et al (2019) Do no harm: a roadmap for responsible machine learning for health care. Nat Med 25(9):1337–1340
Muscas G, Matteuzzi T, Becattini E et al (2020) Development of machine learning models to prognosticate chronic shunt-dependent hydrocephalus after aneurysmal subarachnoid hemorrhage. Acta Neurochir (Wien) 162(12):3093–3105. https://doi.org/10.1007/s00701-020-04484-6
Sorinola A, Buki A, Sandor J, Czeiter E (2019) Risk factors of external ventricular drain infection: proposing a model for future studies. Front Neurol 10:226. https://doi.org/10.3389/fneur.2019.00226
Lenski M, Biczok A, Huge V et al (2019) Role of cerebrospinal fluid markers for predicting shunt-dependent hydrocephalus in patients with subarachnoid hemorrhage and external ventricular drain placement. World Neurosurg 121:e535–e542
de Oliveira JG, Beck J, Setzer M et al (2007) Risk of shunt-dependent hydrocephalus after occlusion of ruptured intracranial aneurysms by surgical clipping or endovascular coiling: a single-institution series and meta-analysis. Neurosurgery 61(5):924–33 (discussion 33-4)
Dorai Z, Hynan LS, Kopitnik TA et al (2003) Factors related to hydrocephalus after aneurysmal subarachnoid hemorrhage. Neurosurgery 52(4):763–9 (discussion 9-71)
Graff-Radford NR, Torner J, Adams HP et al (1989) Factors associated with hydrocephalus after subarachnoid hemorrhage. A report of the Cooperative Aneurysm Study. Arch Neurol 46(7):744–52
Kwon JH, Sung SK, Song YJ et al (2008) Predisposing factors related to shunt-dependent chronic hydrocephalus after aneurysmal subarachnoid hemorrhage. J Korean Neurosurg Soc 43(4):177–181
Lai L, Morgan MK (2013) Predictors of in-hospital shunt-dependent hydrocephalus following rupture of cerebral aneurysms. J Clin Neurosci 20(8):1134–1138
Funding
Open Access funding enabled and organized by Projekt DEAL. ND was funded by the institutional Rahel Hirsch and Lydia Rabinowitsch scholarships, received public body funding for the project GoSafe (Horizon 2020), accepted speaker honoraria from Integra LifeSciences, and serves as an advisor for Alexion Pharmaceuticals.
DF reported receiving grants from the European Commission Horizon2020 PRECISE4Q No. 777107. No funding bodies had any role in study design, data collection and analysis, decision to publish, or preparation of the manuscript.
Author information
Authors and Affiliations
Contributions
All authors contributed to the study conception and design. Material preparation, data collection, and analysis were performed by Dietmar Frey, Nora F. Dengler, Adam Hilbert, Anton Früh, Jenny Sommerfeld, Meike Unteroberdoerster, and Sophie Charlotte Brune. The first draft of the manuscript was written by Dietmar Frey and Nora Dengler, and all authors commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Ethical approval
This study was performed in line with the principles of the Declaration of Helsinki. Approval was granted by the Ethics Committee of Charité – Universitätsmedizin Berlin (EA1/291/14). As this is a retrospective collection of data, no consent to participate was necessary in accordance with our local ethical standards. No personal information in terms of images of any of the patients included in this study was published in this article.
Competing interests
DF reported receiving personal fees from and holding an equity interest in ai4medicine outside the submitted work. AH and TK reported receiving personal fees from ai4medicine outside the submitted work.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Previous presentations
An abstract of this paper was presented at the 73rd annual meeting of the German Neurosurgical Society (DGNC) in Cologne, Germany, in 2022.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Frey, D., Hilbert, A., Früh, A. et al. Enhancing the prediction for shunt-dependent hydrocephalus after aneurysmal subarachnoid hemorrhage using a machine learning approach. Neurosurg Rev 46, 206 (2023). https://doi.org/10.1007/s10143-023-02114-0
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s10143-023-02114-0