Abstract
Neurosurgery is a therapeutic option for patients with refractory obsessive–compulsive disorder who do not respond to previous treatments. Although its efficacy in reducing clinical symptomatology has been proven, few studies have analyzed its effects at the cognitive level. The aim of this systematic review was to describe the cognitive outcomes of functional neurosurgery in patients that went through capsulotomies or cingulotomies. PubMed, Medline, Scopus, PsycInfo, PsyArticles, and Web of Knowledge were searched for studies reporting cognitive outcomes in refractory obsessive–compulsive patients after capsulotomies and cingulotomies. The risk of bias was assessed with the Assessment Tool for Before-After (Pre-Post) Studies With No Control Group tool; 13 studies met inclusion criteria, including 205 refractory obsessive–compulsive disorder patients for both surgical procedures. Results showed a substantial number of studies that did report significant cognitive improvement after surgery, being this improvement specially related to memory and executive functions. The second-most frequent finding is the maintenance of cognitive performance (nor improvement or worsening). From a neuropsychological point of view, this outcome might be considered a success, given that it is accompanied by amelioration of obsessive–compulsive symptoms. Subtle cognitive adverse effects have also been reported. Neurosurgery procedures appear to be safe from a cognitive point of view. Methodological issues must be improved to draw clearer conclusions, but capsulotomies and cingulotomies constitute an effective alternative treatment for refractory obsessive–compulsive disorder patients.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Obsessive–compulsive disorder (OCD) is a severe and disabling psychiatric disorder characterized by the presence of obsessions and/or compulsions [1, 2]. The international prevalence of this disorder is 1,1–1,8%, and of course, if not treated, it is chronic with oscillating symptoms [3]. In addition to clinical symptoms, cognitive impairments had been observed in this population in different domains, such as memory, executive functions, processing speed, and working memory [4,5,6]. First-line psychological and pharmacological treatments for patients with OCD are Cognitive-Behavioral Therapy (CBT) using Exposure with Response Prevention and pharmacotherapy with Selective Serotonin Reuptake Inhibitors (SSRI) [7,8,9,10,11]. If patients do not fully respond to one of these treatments or to the conjunction of both, augmentation strategies (e.g., adding antipsychotics) or switching to other drugs (e.g., clomipramine or other SSRIs) are recommended. However, even though many patients respond to these treatments, it is estimated that 40–60% of the patients remain refractory to pharmacological treatment [12] and that drop-out rates of CBT are between 9 and 16% [13]. For these cases with severe and highly refractory OCD, neurosurgery appears as a feasible therapeutic option [14, 15].
Neurosurgical procedures are used to improve specific symptoms caused by mental illnesses [16]. These are usually carried out by radiofrequency and radiosurgery but also by magnetic resonance-guided focused ultrasound (MRGFU) [17,18,19]. Specifically, capsulotomies and cingulotomies are considered the principal ablative procedures [19]. Capsulotomies refer to a technique in which a lesion is made in the anterior limb of the internal capsule (ALIC) from the frontal horn of the lateral ventricle along the head of the caudate nucleus [20]. By this lesion, frontothalamic connections are disrupted [21] and, specifically, the fibers of passage between the prefrontal cortex and subcortical nuclei, also comprising the dorsomedial thalamus [17]. On the other hand, cingulotomies refer to the procedures in which a lesion is made within the anterior cingulate cortex (ACC) [17], involving the cingulate gyrus and the white matter fibers of the cingulum bundle [21]. Even though the development of ablative surgery had been slowed in the past decades because of intense and public criticism [21], many studies have reported the efficacy of these procedures in reducing obsessive–compulsive symptomatology [e.g., 22, 23]. Moreover, considering the results of 278 patients who underwent anterior capsulotomies, Pepper, Zrinzo, and Hariz [24] reported clinical response in 73% of the patients and remission in 24% of patients, with a low rate of complications. In addition to capsulotomies, Brown et al. [25] also described improvement in OCD symptomatology after cingulotomy. Furthermore, neuroablation appears to have a lower complication rate than other procedures, such as deep brain stimulation (DBS) [26].
As observed, the assessment of the effectiveness of these procedures has been analyzed in terms of obsessive–compulsive symptom reduction, but results in terms of safety regarding the comparison of cognitive performance before and after surgery are scarce. Considering that the presence of different behavioral or cognitive impairments is frequent after brain damage [27], it is essential to describe whether the ablation of gray and white matter structures and whether the modification or interruption of brain tracts in controlled injuries does have a deleterious effect on a patient’s cognitive outcomes. In contrast, a recent systematic review and meta-analysis (i.e., Lai et al. [28]) about the clinical advantages and disadvantages of different neurosurgical interventions did not include or have omitted a detailed analysis of cognitive outcomes after surgery. This is an important issue, given that the analysis of the safety and the effectiveness of surgical procedures could be limited to the study of impairments at psychopathological and physiological levels (e.g., hemorrhages, infections) while ignoring the possibility of adverse cognitive effects. Derived conclusions could help to determine whether carrying out these interventions is convenient. In that case, empirical support would validate this technique, which in the most severe cases could help to improve the patient’s symptoms, and also their cognitive performance.
For this reason, the aim of the present study was to describe the cognitive outcomes of neurosurgery in patients that went through ablative functional neurosurgeries. To do so, a systematic review was conducted reporting the changes in neuropsychological performance found in studies of patients with refractory OCD who had undergone capsulotomies and cingulotomies. In this way, it is defined which cognitive functions are improved or impaired by neurosurgeries and which procedure seems to be safer when considering cognitive performance after functional neurosurgery in the treatment of refractory OCD.
Materials and methods
Literature search
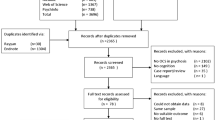
The systematic review has been conducted following the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) [29,30,31]. The PRISMA-based flow diagram (Fig. 1) illustrates the selection procedure. Specific search procedures and inclusion and exclusion criteria were established in a protocol and registered in PROSPERO (International Prospective Register of Systematic Reviews; registration number: CRD42023400038). This study was made in compliance with the Declaration of Helsinki.
Search strategy
Publications about cognitive outcomes following neurosurgery (capsulotomies and cingulotomies) in patients with OCD were obtained from PubMed, Medline, Scopus, PsycInfo, PsyArticles, and Web of Knowledge databases and from backward searching citation chaining. In these six bibliographic databases, relevant published literature until January 2023 was searched using a combination of optimal search strategies including MESH terms “Obsessive–compulsive disorder,” “Cognitive Dysfunction,” “Neurosurgery,” and “Neurosurgical Procedures” if applied, or the terms “OCD,” “obsessive–compulsive disorder,” “obsessive–compulsive disorder,” “obsessive–compulsive,” “obsessive compulsive,” “cogn*,” “neuropsychol*,” “neurocogni*,” “neurosurgery,” “capsulotomy,” “cingulotomy,” “radiosurgery,” “radiofrequency,” “capsuloto*,” “cinguloto*,” and “psychosurgery” (Syntax, see Online resource 1). As mentioned, reference lists of included studies were consulted as an additional source for literature. At this point, no restrictions were included.
Study selection and inclusion and exclusion criteria
Titles and abstracts of all identified were screened independently by two reviewers (G.L. and M.R). Duplicates of the reports were removed. In case of doubt, discrepancies in eligibility were resolved through discussion and consensus. After the selection of literature based on titles and abstracts, the full texts of the retained papers were assessed by reviewers to apply inclusion and exclusion criteria.
For the study selection, clear inclusion and exclusion criteria were followed and applied. Regarding study design, clinical trials (including randomized controlled trials, RCT) and observational studies (including cohort studies and case studies) with OCD patients that underwent neurosurgery (i.e., capsulotomies and cingulotomies) were included. Not only published articles but also data included in book chapters and conference abstracts were included. The included publications were all studies (a) which were published in English, (b) included patients with diagnosed OCD, and (c) 18–65 years aged. Finally, studies must report cognitive outcomes from patients with OCD who underwent capsulotomies and/or cingulotomies.
In addition, exclusion criteria included (systematic) reviews, letters, and animal studies, studies not reporting cognitive outcomes after neurosurgery or assessing those outcomes without standardized instruments (test or neuropsychological batteries), those including interventions other than neurosurgery, articles with designs different from pre-post design, those with samples which had a primary diagnosis other than OCD, and those with samples including patients with different diagnosis (without the possibility of extracting independently OCD data). Also, articles published in languages other than English, articles including patients aged < 18 or > 65 years were excluded, or those including patients with OCD symptoms attributable to medical cause (e.g., patients with the presence of OCD symptoms after streptococcal infection) [6].
Qualitative assessment
The methodological quality and risk of bias assessment was performed using the National Heart, Lung, and Blood Institute Assessment Tool for Before-After (Pre-Post) Studies With No Control Group tool [32], according to Ma et al. [33] recommendations. Based on this resource, 12 questions about methodological issues were answered and studies were classified by the study overall quality (“poor,” “fair,” or “good”). No minimum quality score for inclusion was stated.
Data collection
The data was extracted by one author (G.L.) using a standardized form and revised by two authors (J.A.P. and G.Lu). Patient data was compared among the studies to avoid the inclusion of the same sample in more than one report. In overlapping cohorts, the study with the longest follow-up was included. Where possible, details of the study design (sample), employed surgical technique, assessment (used cognitive tests or batteries), and other outcomes (subjective cognitive complaints or observations by relatives or physicians) were reported. Instruments and their scores were described and classified according to relevant OCD cognitive meta-analysis classification [6], reference guidelines in the neuropsychological field [34, 35], and validity studies [e.g., 36, 37]. In cases where the cognitive scores used were not explicitly available in the revised articles, the interpretation made by the authors was preserved, or no interpretation was included (reflected in Tables 2 and 3).
Results
Search results
In the initial search, 702 articles were identified, of which only 689 could be retrieved. After excluding 294 duplicates, 395 articles remained, of which titles and abstracts were screened. Then, 357 were excluded because the searched terms were not included or explored. Afterward, 38 publications remained, of which the full text was examined, which results in the exclusion of 25 other articles due to different reasons (see Fig. 1). The final sample comprised 13 articles. This final sample included 205 OCD refractory patients, of which 189 belong to studies that assessed cognitive outcomes after capsulotomies and only 19 to studies analyzing cognitive outcomes after cingulotomies. An overview of sample size, surgical procedures, and clinical features is included in Table 1. Cognitive measures, assessment times, and cognitive outcomes are reported in Table 2 and Table 3.
The set of 13 revised manuscripts showed that up to 40 neuropsychological tests and batteries have been used to assess cognitive outcomes in patients with refractory OCD who had undergone capsulotomies and cingulotomies. In order to analyze the reported information, the following cognitive domains were distinguished (see Tables 2 and 3): attention, visuospatial abilities, memory (including verbal and visual memory), language, working memory, executive functions, verbal fluency, information processing speed, intelligence, motor functioning, and overall cognitive functioning. This was made according to the reference guidelines reported in the “Materials and method” section. Overall, 7 (54%) studies reported significant changes in neuropsychological scores after surgery (all of them reporting cognitive amelioration, and only one reporting cognitive worsening for one score analyzed), and 6 (46%) found no significant changes. The presented results are differentiated according to the target of the lesion performed (capsulotomies or cingulotomies). Finally, the results of the quality assessment are reported.
Cognitive outcomes depending on the anatomical target
Capsulotomies
As observed in Table 2, capsulotomies are procedures in which the cognitive outcomes of OCD patients after surgery have been examined, showing significant changes in several domains.
First, there have been improvements consistently reported in the domains of memory and executive functions. Regarding memory, improvement has been found globally (memory quotient of the Rey-Kim memory test) [38] and in both verbal [39] and visual [39,40,41] modalities, the latter being more prominent. It should be noted that improvements occurred in tests that included both immediate and delayed recall scores [39,40,41], being the improvement registered by the Rey-Osterrieth complex figure test (ROCFT) the most frequent one (immediate recall [41], delayed Recall [40, 41]). Additionally, it should be noted that improvements in verbal memory were reported (scores of the immediate and delayed recall of the logical memory subtest of the Weschler memory scale-revised (WMS-R) [39]). Regarding executive functions, improvements are found specifically in California sorting test-A (right concept) [42], Wisconsin card sorting test (WCST) (hits, errors, perseverative errors, and categories scores) [39, 40], TMT effect-time score [40], and stroop (interference time score) [42]. However, the interpretation of this last score is not straightforward (since it differs from the standardized form and the authors did not provide additional information).
Secondly, scores in motor functioning, visuospatial abilities, language, and verbal fluency domains have shown significant changes. These scores show improvements in the grooved pegboard test (dominant hand-time), hand dynamometer (non-dominant hand), trail making test A (TMT-A), vocabulary subtest from Weschler abbreviated scale of intelligence (WASI) [40], and verbal fluency test (number of words) [42]. In addition, there have also been reported improvements in general intelligence [total intelligence quotient (IQ) of WASI, IQ of Hungarian version of the Weschler intelligence test (MAWI) [40, 42] and specific components of intelligence (performance IQ, block design subtest of WASI) [39, 40].
Thirdly, the only worsening recorded to date after capsulotomies has been found in verbal fluency tasks (intrusion errors in the category fluency test) [42].
Fourthly, no significant changes were found in neuropsychological performance in attention, working memory, information processing speed, and overall cognitive functioning (see Table 2).
Fifthly, when studying subjective complaints, or behavioral observations, 7 studies reported no cognitive adverse effects [38, 41,42,43,44,45,46], while other 4 reported the presence of executive dysfunction, apathy, disinhibition symptoms, disorientation (temporal and spatial), childish behavior, lack of interest, decreased motivation, inappropriate laughter and impaired memory, and confusion [39, 40, 47, 48] after surgery. Only one study used a scale to register subjective complaints (EAD scale [47]), while the others used clinical observation or spontaneous reports of the patients or caregivers [38, 45], but in most cases, the method used to register the presence of subjective effects was not stated [39,40,41, 43, 44, 46].
Finally, regarding assessment times, of the 12 studies that examined changes in neuropsychological performance, most conducted the post-surgical assessments within the first year after surgery [39, 41,42,43,44,45,46, 4948], extending the post-assessments in some cases to two years [38, 40, 42] or more [47,48,49]. Of those that performed post-evaluation during the first year after surgery, the majority were radiofrequency studies [39, 41, 43, 46], followed by radiosurgeries [47] and thermal lesioning [44]. The remaining studies performed evaluations in a period equal to or longer than two years and included radiofrequency [42, 47, 48], radiosurgery [40, 47, 49], and thermal lesioning [38] procedures. Considering the assessment times of those 11 studies analyzing subjective complaints, two reported subjective cognitive adverse effects immediately after surgery [47, 48], another one at 8 months [39], and two at 12 months after surgery [39, 47]. Three studies reported the assessment times of subjective complaints: Jung et al. [44] reported that this assessment was made at 6 months, Kim et al. [38] in every follow-up visit (i.e., 1 week, 1 month, 3 months, 6 months, 12 months, and 24 months after surgery) and Peker et al. [45] also in every appointment (i.e., 6 months and annually until fifth year after surgery). However, the eight remaining articles did not indicate an exact indication of which time they carried out the assessment of subjective complaints [39,40,41,42,43, 46,47,48].
Cingulotomies
As seen in Table 3, to date, only one study reported significant improvements, found in the domain of executive functions, as reflected in the scores of WCST (total number of errors, perseverative responses, perseverative errors, and non-perseverative errors) [50]. In addition, it should be noted that no pre-post cognitive neuropsychological changes had been studied in attention, language, information processing speed, and overall cognitive functioning domains. Regarding subjective complaints, one study reported no worsening [43], and one study described the presence of subjective immediate memory dysfunction complaints that were not found by means of standardized neuropsychological assessment [50].
Considering the cognitive assessment times, the two studies included for cingulotomies performed neuropsychological assessments within the first year after surgery, and both followed radiofrequency procedures [43, 50]. One of them reported subjective complaints that were registered 2–3 months after surgery [50].
Quality assessment
In brief, qualitative assessment rated five studies as “poor” (38%), four as “fair” (31%), and four as “good” (31%) (see Online resource 2) [32].
Discussion
In the present review, studies analyzing cognitive outcomes in patients with OCD after ablative neurosurgery (capsulotomies and cingulotomies) have been revised. The main result was that a substantial number of studies report cognitive amelioration in neuropsychological performance after surgery. This improvement appears to be multi-domain, although it is more frequently reported for memory and executive functions for both types of surgeries. In addition, many studies have shown no cognitive changes (nor improvement nor worsening), what can be considered a successful outcome, suggesting that surgery is a safe and potentially curative procedure for many patients without showing adverse effects. It appears that increases in neuropsychological performance may be quite extensive in capsulotomies, but subtle deleterious effects have also been reported. No worsening in neuropsychological functioning has been reported for cingulotomies, although some patients exposed subjective complaints that should be studied in more detail. Overall, neurosurgery has shown the presence of improvements that are clearly superior to the possible worsening detected.
In capsulotomies, a heterogeneous pattern of cognitive improvements has been reported, although the body of the results is consistently related to memory and executive functions. Improvements have also been found in domains such as motor functioning, visuospatial abilities, language, and verbal fluency. In this regard, the pathological activity of the hyperactive orbito-fronto-striato-thalamo-cortical circuitry that is associated with OCD [51,52,53] appears to be modified by the intervention in fiber tracts between orbitofrontal cortex (OFC) and thalamus by targeting the ventral fibers of ALIC [54]. An interruption of this circuit, and the amelioration of obsessive–compulsive symptoms, may lead to secondary restoration of the activity of the networks that relate to frontal executive abilities [55]. Indeed, executive impairments appear to promote the exacerbation of OCD symptoms [56]. So, an improvement in neuropsychological performance might relate to a redistribution of cognitive resources that previously could be involved in clinical symptoms. This last idea is also similar to the capacity-reduction hypothesis in disorders such as depression. People with depression present a cognitive capacity reduction that is exhibited in effortful tasks (including cognitive tasks) because available cognitive resources are focused on negative self-content and fewer attentional resources are displayed. Certainly, interference in effortful processing has been associated with psychopathology in general [57, 58]. Therefore, effective treatment of clinical symptoms (also in OCD) may allow for optimum distribution of the available attentional capacity and, in consequence, better neuropsychological performance. Along with the mentioned improvement, worsening has also been found for one executive score in only one study (i.e., intrusion errors of category fluency test) [42], as well as executive and memory subjective cognitive complaints in 4 studies [39, 40, 47, 48]. According to the literature, if worsening occurs, it might be related to the localization of the target area within the ALIC, where more dorsally located lesions might be related to impairments in different cognitive domains (i.e., executive functions and working memory) [59]. However, ventral lesions of this structure seem to involve the longitudinal fiber tracts that connect anterior cingulate and orbitofrontal projections to the striatum, thalamus, and brainstem, reducing the adverse effects without compromising the therapeutic effects [28]. In this way, results with an almost complete absence of cognitive worsening could denote preservation of the ALIC fibers that could cause cognitive impairments when lesioned [59].
In cingulotomies, the explanation of cognitive changes associated with neurosurgery is challenging given that the available literature is modest and the cognitive domains studied had been a few. On the one hand, the main finding is a general cognitive improvement, especially in executive functions, while no significant changes have been found in other cognitive functions. Kim et al. [60] stated that improvements in WCST scores (similar to those of Jung et al. [50]) reflect improvements in the capacity to flexibly change behavior as the environment changes (cognitive flexibility) and also to monitor and adapt behavior (cognitive control) [50, 60,61,62]. This last domain is specially related to the main target of cingulotomies, the dorsal anterior cingulate cortex (dACC), which contributes to response monitoring, response inhibition, and goal selection [59]. According to the expected value of control theory, the dACC would exhibit an abnormal function in OCD (i.e., would be pathologically active) and would lead to behaviors related to OCD symptoms. Particularly, the dACC would present problems in the specification of the identity and/or intensity of signals (called cognitive control signals) produced to alert of possible dangers. Even if the threat is not real or does not longer exist, the control signals persist, leading to the emergence of repetitive behaviors that are not adaptative to respond to stimuli interpreted as highly threating [63, 64]. In this regard, it is possible that cingulotomies would reduce the activity of the dACC and normalize its function. This would lead to the allocation of cognitive resources that were previously involved in generating behaviors in response to perceived threats [59] to the tasks of interest. On the other hand, and considering subjective cognitive complaints, only one of the two studies on cingulotomies stated cognitive adverse effects related to memory [50]. In this view, the cingulate gyrus has been associated with memory function by means of its connections with the retrosplenial cortex, hippocampal, and parahippocampal regions [65, 66], so the extension of the lesion in cingulotomies to proximal regions could, in turn, have an impact on memory mechanisms. However, in the absence of a more detailed study of cognitive control and the role and connections of the cingulate cortex, determining how cingulotomies impact executive functions and memory in OCD is still a complex purpose.
From a methodological point of view, the revised studies show a heterogeneity that makes it difficult to implement a direct comparison among them. Firstly, the differences in study design have possibly conditioned the reporting of results (i.e., case studies [43]). Secondly, the inclusion and exclusion criteria may have influenced the possibility of detecting cognitive changes due to a ceiling effect. Thus, Jung et al. [44] and Kim et al. [38] excluded all patients that had an MMSE score lower or equal to 24, Zhan et al. [48] did not include patients that had cognitive deficits, and Peker et al. [45] excluded those patients with cognition in the “low range.” Given this, the exclusion of participants with the worst cognitive performance might cause, in an artifactual way, difficulties in detecting cognitive improvement that could occur since patients whose performance would likely improve are eliminated. In those studies in which only patients with normal cognitive performance are evaluated, the possibilities of detecting changes are clearly limited by the small margin for improvement that exists. In fact, in three of these four studies, no significant changes were detected in neuropsychological pre-post comparisons. Thirdly, three different neurosurgery techniques have been detected in the revised literature (radiosurgery, radiofrequency, and thermal lesioning by focused ultrasound) (e.g., [44, 46, 49]), which could influence the results according to their mechanisms of action. Indeed, the time course of clinical response to radiosurgery (onset of symptom improvement has been reported from 3 months to 3 years; [14]) is different from other procedures such as radiofrequency, where the gradual nature of the radiation effects is characteristic [19]. This raises the idea that the timing of neuropsychological assessments may be conditioned by the time course of the effects of such techniques. In the case of Peker’s et al. [45] radiosurgery study, this assumption is relevant given that the follow-up was made 6 months after surgery which could be a premature time for the detection of the changes. However, for all the other studies, the times selected for the cognitive assessment were adequate according to the type of technique, so the cognitive changes that might have taken place have probably been captured. By extension, this issue ties in with the possibility of detecting cognitive changes (improvement or worsening) within the first year after surgery. Along with this idea, improvement of executive functions and memory may evolve in the second year after surgery [38, 42]. Therefore, for studies that did not show changes in cognitive performance, and with the neuropsychological follow-up taking place in the first six or twelve months after surgery (e.g., [44,45,46], it cannot be ruled out that a longer period of time was necessary for the improvement in cognitive performance to take place and, consequently, be detected. Fourthly, some studies included tests that were difficult to access (e.g., [43]), or have used versions adapted to demographic characteristics of the sample (but with the not available explanation of these adaptations), so direct interpretation and comparison of the results was not possible (e.g., K-WAIS [44]; Korean colour word stroop test [38]; category fluency test [42]). In other cases, no scores were indicated or relevant data were missing (e.g., numerical data or p-value), making it difficult to interpret the results [38, 43, 44, 47,48,49,50]. Moreover, the realization of an adequate neuropsychological assessment along with standardized instruments for the detection of cognitive impairments is a fundamental issue in the neurosurgical context since patients with different cognitive profiles are linked to distinct clinical profiles [67] that appear to benefit differently from capsulotomies [68]. In this line, the differences between the worsening detected by neuropsychological performance and by subjective cognitive adverse reporting for both surgeries must be considered, and it may be appropriate to suggest that some impairments may not have been recorded in neuropsychological assessment. This fact is relevant since it was not possible to assess how cognitive impairments have impacted the patient’s daily functioning through the tests and batteries employed in the revised studies. It can also be added that those patients referring to subjective complaints could be more aware of executive and memory impairments, considering their notable interference with daily life [e.g., 69]. Thus, it would be necessary to build a protocol in which subjective complaints are registered in a systematic way. Lastly, the quality of the studies that make up this review should be afforded. The number of studies that have been assessed to be “poor” or “fair” quality is noteworthy (69%), exceeding the number of those considered to have “good” quality (31%). In this vein, there is a clear need to improve the methodological quality of studies with standardized procedures according to the established consensus of the neurosurgery community [e.g., 70]. However, when the studies rated as “good” are examined, it is possible to observe a very similar pattern of results showing consistent improvements in executive functions and memory [39,40,41]. Indeed, no worsening scores are found in this group of studies. Therefore, although methodological difficulties are reported in most of the revised manuscripts, and results should be addressed with caution, derived conclusions converge among studies.
Conclusions
Among the revised literature, ablative functional neurosurgery appears to be a safe procedure from the cognitive or neuropsychological point of view. Memory and executive functions appear to be the domains in which the most marked improvement has been found. In addition, a great proportion of studies did not show significant cognitive changes after neurosurgery which might also be considered a successful outcome after surgery. In conclusion, neurosurgery procedures appear to be an alternative treatment option for refractory OCD patients, but methodological issues related to neuropsychological or cognitive assessment should be improved to further detail the changes and cognitive outcomes and to draw clearer conclusions about the efficacy of the procedures.
Data availability
Not applicable.
References
Robbins TW, Vaghi MM, Banca P (2019) Obsessive-compulsive disorder: puzzles and prospects. Neuron 102:27–47. https://doi.org/10.1016/j.neuron.2019.01.046
Stein DJ, Costa DLC, Lochner C, Miguel EC, Reddy YCJ, Shavitt RG, van den Heuvel OA, Simpson HB (2019) Obsessive-compulsive disorder Nat Rev Dis Primers 5:52. https://doi.org/10.1038/s41572-019-0102-3
American Psychiatric Association (2013) Diagnostic and statistical manual of mental disorders, 5th edn. American Psychiatric Publishing, Washington, DC
Abramovitch A, Abramowitz JS, Mittelman A (2013) The neuropsychology of adult obsessive-compulsive disorder: a meta-analysis. Clin Psychol Rev 33:1163–1171. https://doi.org/10.1016/j.cpr.2013.09.004
Shin NY, Lee TY, Kim E, Kwon JS (2014) Cognitive functioning in obsessive-compulsive disorder: a meta-analysis. Psychol Med 44:1121–1130. https://doi.org/10.1017/S0033291713001803
Snyder HR, Kaiser RH, Warren SL, Heller W (2014) Obsessive-compulsive disorder is associated with broad impairments in executive function: a meta-analysis. Clin Psychol Sci 3:301–330. https://doi.org/10.1177/2167702614534210
Koran LM (2007) Obsessive-compulsive disorder: an update for the clinician. Focus 5:299–313. https://doi.org/10.1176/foc.5.3.foc299
Baldwin DS, Anderson IM, Nutt DJ, Allgulander C, Bandelow B, den Boer JA, Christmas DM, Davies S, Fineberg N, Lidbetter N, Malizia A, McCrone P, Nabarro D, O’Neill C, Scott J, van der Wee N, Wittchen H (2014) Evidence-based pharmacological treatment of anxiety disorders, post-traumatic stress disorder and obsessive-compulsive disorder: a revision of the 2005 guidelines from the British Association for Psychopharmacology. J Psychopharmacol 28:403–439. https://doi.org/10.1177/0269881114525674
Katzman MA, Bleau P, Chokka P, Kjernisted K, Van Ameringen M, The Canadian Anxiety Guidelines Initiative Group on behalf of the Anxiety Disorders Association of Canada/Association Canadienne des troubles anxieux and McGill University (2014) The Canadian clinical practice guidelines for the management of anxiety, posttraumatic stress and obsessive-compulsive disorders. BMC Psychiatry 14:S1. https://doi.org/10.1186/1471-244X-14-S1-S1
National Institute for Health and Care Excellence (NICE) (2005) Obsessive-compulsive disorder and body dysmorphic disorder: treatment. https://www.nice.org.uk/guidance/cg31
National Institute for Health and Care Excellence (NICE) (2013) Obsessive-compulsive disorder. Evidence Update September 2013. https://www.nice.org.uk/guidance/cg31/evidence/evidence-update-194847085
Drubach DA (2015) Obsessive-compulsive disorder. Continuum (Minneap Minn) 21:783–788. https://doi.org/10.1212/01.CON.0000466666.12779.07
Pozza A, Dèttore D (2017) Drop-out and efficacy of group versus individual cognitive behavioral therapy: what works best for obsessive-compulsive disorder? A systematic review and meta-analysis of direct comparisons. Psychiatry Res 258:24–36. https://doi.org/10.1016/j.psychres.2017.09.056
Miguel EC, Lopes AC, McLaughlin NCR, Norén G, Gentil AF, Hamani C, Shavitt RG, Matustuzzo MC, Vattimo EFQ, Canteras M, De Salles A, Gorgulho A, Salvajoli JV, Fonoff ET, Paddich I, Hoexter MQ, Lingquist C, Haber SN, Greenberg BD, Sheth SA (2018) Evolution of gamma knife capsulotomy for intractable obsessive-compulsive disorder. Mol Psychiatry 24:218–240. https://doi.org/10.1038/s41380-018-0054-0
Sinha S, McGovern RA, Mikell CB, Banks GP, Sheth SA (2015) Ablative limbic system surgery: review and future directions. Curr Behav Neurosci Rep 2:49–59. https://doi.org/10.1007/s40473-015-0038-1
Martínez-Álvarez R (2019) Radiosurgery for behavioral disorders. Prog Neurol Surg 34:289–297. https://doi.org/10.1159/000493076
Greenberg BD, Rauch SL, Haber SN (2010) Invasive circuitry-based neurotherapeutics: stereotactic ablation and deep brain stimulation for OCD. Neuropsychopharmacology 35:317–336. https://doi.org/10.1038/npp.2009.128
Jung HH, Chang WS, Kim SJ, Kim C, Chang JW (2018) The potential usefulness of magnetic resonance guided focused ultrasound for obsessive compulsive disorders. J Korean Neurosurg Soc 61:427–433. https://doi.org/10.3340/jkns.2017.0505.004
Levequé M (2014) Psychosurgery. Springer International Publishing, Switzerland, New techniques for brain disorders
Martínez-Álvarez R (2014) Ablative surgery for obsessive-compulsive disorders. In: Sun B, De Salles A (eds) Neurosurgical Treatments for Psychiatric Disorders. Springer, Dordrecht, Shanghai, China, pp 105–112
Staudt MD, Herring EZ, Gao K, Miller JP, Sweet JA (2019) Evolution in the treatment of psychiatric disorders: from psychosurgery to psychopharmacology to neuromodulation. Front Neurosci 13:108. https://doi.org/10.3389/fnins.2019.00108
Dougherty DD, Baer L, Cosgrove GR, Cassem EH, Price BH, Nierenberg AA, Jenike MA, Rauch SL (2002) Prospective long-term follow-up of 44 patients who received cingulotomy for treatment-refractory obsessive-compulsive disorder. Am J Psychiatry 159:269–275. https://doi.org/10.1176/appi.ajp.159.2.269
Spatolla G, Martinez-Alvarez R, Martinez-Moreno N, Rey G, Linera J, Rios-Lago M, Sanz M, Gutiérrez J, Vidal P, Richieri R, Régis J (2018) Results of Gamma Knife anterior capsulotomy for refractory obsessive-compulsive disorder: results in a series of 10 consecutive patients. J Neurosurg 131:376–383. https://doi.org/10.3171/2018.4.JNS171525
Pepper J, Zrinzo L, Hariz M (2019) Anterior capsulotomy for obsessive-compulsive disorder: a review of old and new literature. J Neurosurg 11:1–10. https://doi.org/10.3171/2019.4.JNS19275
Brown LT, Mikell CB, Youngerman BE, Zhang Y, McKhann GM, Sheth SA (2016) Dorsal anterior cingulotomy and anterior capsulotomy for severe, refractory obsessive-compulsive disorder: a systematic review of observational studies. J Neurosurg 124:77–89. https://doi.org/10.3171/2015.1.JNS14681
Kumar KK, Bhati MT, Ravikumar VK, Ghanouni P, Stein SC, Halpern CH (2019) MR-guided focused ultrasound versus radiofrequency capsulotomy for treatment-refractory obsessive-compulsive disorder: a cost-effectiveness threshold analysis. Front Neurosci 13:66. https://doi.org/10.3389/fnins.2019.00066
Lezak M, Howieson DB, Bigler ED, Tranel D (2012) Neuropsychological assesment. Oxford University Press, New York
Lai Y, Wang T, Zhang C, Lin G, Voon V, Chang J, Sun B (2020) Effectiveness and safety of neuroablation for severe and treatment-resistant obsessive-compulsive disorder: a systematic review and meta-analysis. J Psychiatry Neurosci 45:356–369. https://doi.org/10.1503/jpn.190079
Liberati A, Altman DG, Tetzlaff J, Mulrow C, Getzsche PC, Ionnidis JPA, Clarke M, Devereaux PJ, Kleijnen J, Moher D (2009) The PRISMA statement for reporting systematic reviews and meta-analyses of studies that evaluate health care interventions: explanation and elaboration. Plos Med 6:e1000100. https://doi.org/10.1371/journal.pmed.1000100
Moher D, Liberati A, Tetzlaff J, Altman DG, the PRISMA group (2009) Prefered reporting items for systematic reviews and meta-analyses: the PRISMA statement. PLoS Med 21:e1000097. https://doi.org/10.1371/journal.pmed.1000097
Page MJ, McKenzie JE, Bossuyt PM, Boutron I, Hoffmann TC, Mulrow CD, Shamseer L, Tetzlaff JM, Akl EA, Brennan SE, Chou R, Glanville J, Grimshaw JM, Hróbjartsson A, Lalu MM, Li T, Loder EW, Mayo-Wilson E, McDonald S, McGuinness LA, Stewart LA, Thomas J, Tricco AC, Welch VA, Whiting P, Moher D (2021) The PRISMA 2020 statement: an updated guideline for reporting systematic reviews. BMJ 372:n71. https://doi.org/10.1136/bmj.n71
National Heart, Lung and Blood Institute (NIH) (2021) Study quality assessment tools. https://www.nhlbi.nih.gov/health-topics/study-quality-assessment-tools
Ma L, Wang Y, Yang Z, Huang D, Weng H, Zeng X (2020) Methodological quality (risk of bias) assessment tools for primary and secondary medical studies: what are they and which is better? Mil Med Res 7:7. https://doi.org/10.1186/s40779-020-00238-8
Kreutzer JS, DeLuca J, Caplan B (2018) Encyclopedia of clinical neuropsychology. Springer Intenational Publishing, Switzerland
Strauss E, Sherman E, Spreen O (2006) A compendium of neuropsychological tests. Oxford University Press, New York
Sánchez-Cubillo I, Periañez JA, Adrover-Roig D, Rodríguez-Sánchez JM, Ríos-Lago M, Tirapu J, Barceló F (2009) Construct validity of the trail making test: role of task-switching, working memory, inhibition/interference control, and visuomotor abilities. J Int Neuropsychol Soc 15:438–450. https://doi.org/10.1017/S1355617709090626
Periañez JA, Lubrini G, García-Gutiérrez A, Ríos-Lago M (2020) Construct validity of the stroop color-word test: influence of speed of visual search, verbal fluency, working memory, cognitive flexibility, and conflict monitoring. Arch Clin Neuropsychol 36:99–111. https://doi.org/10.1093/arclin/acaa034
Sj K, Roh D, Jung HH, Chang WS, Kim C, Chang JW (2018) A study of novel bilateral thermal capsulotomy with focused ultrasound for treatment-refractory obsessive-compulsive disorder: 2-year follow-up. J Psychiatry Neurosci 43:327–337. https://doi.org/10.1503/jpn.170188
Gong F, Li P, Li B, Zhang S, Zhang X, Yang S, Liu H, Wang W (2018) A study of cognitive function in treatment-refractory obsessive-compulsive disorder treated with capsulotomy. J Neurosurg 128:583–595. https://doi.org/10.3171/2016.9.JNS152494
Batistuzzo MC, Hoexter MQ, Taub A, Gentil AF, Cesar RCC, Joaquim MA, D’Alcante MA, McLaughlin NC, Canteras MM, Shavitt RG, Savage CR, Greenberg BD, Norén G, Miguel EC, Lopes AC (2015) Visuospatial memory improvement after gamma ventral capsulotomy in treatment refractory obsessive-compulsive disorder patients. NPP 40:1837–1845. https://doi.org/10.1038/npp.2015.33
Krámska L, Urgošik D, Liščák R, Hrešková L, Skopová J (2021) Neuropsychological outcome in refractory obsessive-compulsive disorder treated with anterior capsulotomy including repeated surgery. Psychiatry Clin Neurosci 75:101–197. https://doi.org/10.1111/pcn.13190
Csigó K, Harsányi A, Demeter G, Cs R, Németh A, Racsmány M (2010) Long-term follow-up of patients with obsessive-compulsive disorder treated by anterior capsulotomy: a neuropsychological study. J Affect Disord 126:198–205. https://doi.org/10.1016/j.jad.2010.02.127
Fodstad H, Strandman E, Karlsson B, West KA (1982) Treatment of chronic obsessive compulsive states with stereotactic anterior capsulotomy or cingulotomy. Acta Neurochir Suppl 62:1–23. https://doi.org/10.1007/BF01402207
Jung HH, Kim SJ, Roh D, Chang JG, Chang WS, Kweon EJ, Kim C, Chang JW (2015) Bilateral thermal capsulotomy with MR-guided focused ultrasound for patients with treatment-refractory obsessive-compulsive disorder: a proof-of-concept study. Mol Psychiatry 20:1205–1211. https://doi.org/10.1038/mp.2014.154
Peker S, Yavuz M, Yilmaz M, Sengoz M, Ulku N, Ogel K (2020) Efficacy and safety of gamma ventral capsulotomy for treatment-resistant obsessive-compulsive disorder: a single-center experience. World Neurosurg 141:e941–e952. https://doi.org/10.1016/j.wneu.2020.06.098
Oliver B, Gascón J, Aparicio A, Ayats E, Rodríguez R, Maestro de León JL, García-Bach M, Anton Soler P (2003) Bilateral anterior capsulotomy for refractory obsessive-compulsive disorders. Stereotact Funct Neurosurg 81:90–95. https://doi.org/10.1159/000075110
Rück C, Karlsson A, Steele D, Edman G, Meyerson BA, Ericson K, Nyman H, Åsberg M, Svanborg P (2008) Capsulotomy for obsessive-compulsive disorder. Long-term follow-up of 25 patients. Arch Gen Psychiatry 65:914–922. https://doi.org/10.1001/archpsyc.65.8.914
Zhan S, Liu W, Li D, Pan S, Pan Y, Li Y, Sun B (2014) Long-term follow-up of bilateral anterior capsulotomy in patients with refractory obsessive-compulsive disorder. Clin Neurol Neurosurg 119:91–95. https://doi.org/10.1016/j.clineuro.2014.01.009
Kassel MT, Lositsky O, Vaidya AR, Badre D, Malloy PF, Greenberg BD, Marsland R, Noren G, Sherman A, Rasmussen SA, McLaughlin NCR (2022) Differential assessment of frontally-mediated behaviors between self- and informant-report in patients with obsessive-compulsive disorder following gamma ventral capsulotomy. Neuropsychologia 170:108211. https://doi.org/10.1016/j.neuropsychologia.2022.108211
Jung HH, Kim C, Chang JH, Park YG, Chung SS, Chang JW (2006) Bilateral anterior cingulotomy for refractory obsessive-compulsive disorder: long-term follow-up results. Stereotact Funct Neurosurg 84:184–189. https://doi.org/10.1159/000095031
Saxena S, Brody AL, Schwartz JM, Baxter LR (1998) Neuroimaging and frontal-subcortical circuitry in obsessive-compulsive disorder. Br J Psychiatry Suppl 35:26–37. https://doi.org/10.1192/S0007125000297870
Chamberlain SR, Blackwell AD, Fineberg NA, Robbins TW, Sahakian BJ (2005) The neuropsychology of obsessive compulsive disorder: the importance of failures in cognitive and behavioral inhibition as candidate endophenotypic markers. Neurosci Biobehav Rev 29:399–419. https://doi.org/10.1016/j.neubiorev.2004.11.006
Menzies L, Chamberlain SR, Laird AR, Thelen SM, Sahakian BJ, Bullmore ET (2008) Integrating evidence from neuroimaging and neuropsychological studies of obsessive-compulsive disorder: the orbitofronto-striatal model revisited. Neurosci Biobehav Rev 32:525–549. https://doi.org/10.1016/j.neubiorev.2007.09.005
Oliveira BF, Gorgulho A, Saraiva CWC, Lopes AC, Ribeiro JG, Pássaro AM, Hoexter MQ, Miguel EC, De Salles AEF (2019) Understanding gamma ventral capsulotomy: potential implications of difusión tensor image tractography on target selectivity. Surg Neurol Int 10:136. https://doi.org/10.25259/SNI-65-2019
Savage CR (1998) Neuropsychology of obsessive-compulsive disorder: research findings and treatment implications. In: Jenike M, Baer L, Minichiello W (eds) Obsessive-compulsive disorders: practical management, 3rd edn. Mosby, St.Louis, MO, pp 254–275
Shepard E, Stern ER, van den Heuvel OA, Costa DLC, Batistuzzo MC, Godoy PBG, Lopes AC, Brunoni AR, Hoexter MQ, Shavitt RG, Reddy YCJ, Lochner C, Stein DJ, Simpson HB, Miguel EC (2021) Toward a neurocircuit-based taxonomy to guide treatment of obsessive-compulsive disorder. Mol Psychiatry 26:4583–4604. https://doi.org/10.1038/s41380-020-01007-8
Hartlage S, Alloy LB, Vázquez C, Dykman B (1993) Automatic and effortful processing in depression. Psychol Bull 113:247–278. https://doi.org/10.1037/0033-2909.113.2.247
Nuño L, Gómez-Benito J, Carmona VR, Pino O (2021) A systematic review of executive function and information processing speed in major depression disorder. Brain Sci 11:147. https://doi.org/10.3390/brainsci11020147
Avecillas JM, Hurwitz TA, Bogod NM, Honey CR (2021) Tractography-guided anterior capsulotomy for major depression and obsessive-compulsive disorder: targeting the emotion network. Oper Neurosurg (Hagerstown) 20:406–412. https://doi.org/10.1093/ons/opaa420
Kim CH, Chang JW, Koo M, Kim JW, Suh HS, Park IH, Hs L (2003) Anterior cingulotomy for refractory obsessive-compulsive disorder. Acta Psychiatr Scand 107:283–290. https://doi.org/10.1034/j.1600-0447.2003.00087.x
Heaton, RK (1981) Wisconsin card sorting test manual. Psychological Association Resources, Odessa
Diamond A (2013) Executive functions. Annu Rev Psychol 64:135–168. https://doi.org/10.1146/annurev-psych-113011-143750
McGovern RA, Sameer S (2017) Role of the dorsal anterior cingulate in obsessive-compulsive disorder: converging evidence from cognitive neuroscience and psychiatric neurosurgery. J Neurosurg 126(1):132–147. https://doi.org/10.3171/2016.1.JNS15601
Gehring WJ, Himle J, Nisenson LG (2000) Action-monitoring dysfunction in obsessive-compulsive disorder. Psychol Sci 11(1):1–6. https://doi.org/10.1111/1467-9280.00206
Beckman M, Johansen Berg H, Rushworth MF (2009) Connectivity-based parcellation of human cingulate cortex and its relation to functional specialization. J Neurosci 29:1175–1190. https://doi.org/10.1523/JNEUROSCI.3328-08.2009
Bubb EJ, Metzler-Baddeley C, Aggleton JP (2018) The cingulum bundle: anatomy, function, and dysfunction. Neurosci Biobehav Rev 92:104–127. https://doi.org/10.1016/j.neubiorev.2018.05.008
Bragdon LB, Gibb BE, Coles ME (2017) Does neuropsychological performance in OCD relate to different symptoms? A meta-analysis comparing the symmetry and obsessing dimensions. Depress Anxiety 35:761–774. https://doi.org/10.1002/da.22785
Gong F, Li B, Zhang S, Wang Y, Gao Y, Xu Y, Wang X, Xiong B, Li D, Wen R, Qin Z, Wang W (2019) The suitability of different subtypes and dimensions of obsessive-compulsive disorder for treatment with anterior capsulotomy: a long-term follow-up study. Stereotact Funct Neurosurg 97:319–336. https://doi.org/10.1159/000500137
Overdorp EJ, Kessels RPC, Claassen JA, Oosterman JM (2016) The combined effect of neuropsychological and neuropathological deficits on instrumental activities of daily living in older adults: a systematic review. Neuropsychol Rev 26:92–106. https://doi.org/10.1007/s11065-015-9312-y
Nuttin B, Wu G, Mayberg H, Hariz M, Gabriëls L, Galert T, Merkel R, Kubu C, Vilela-Filho O, Matthews K, Taira T, Lozano AM, Schechtmann G, Doshi P, Broggi G, Régis J, Alkhani A, Sin B, Elijamel S, Schulder M, Kaplitt M, Eskandar E, Rezai A, Krauss JK, Hilven P, Schuurman R, Ruiz P, Chang JW, Cosyns P, Lipsman N, Voges J, Cosgrove R, Li Y, Schlaepfer T (2014) Consensus on guidelines for stereotactic neurosurgery for psychiatric disorders. J Neurol Neurosurg Psychiatry 85:1003–1008. https://doi.org/10.1136/jnnp-2013-306580
Acknowledgements
G. L-Z. was supported by a fellowship from the Fundación Tatiana Pérez de Guzman el Bueno. We would like to thank the helpful comments and suggestions of Arantza Zaballa Ayestaran for the writing style of the manuscript.
Funding
Open Access funding provided thanks to the CRUE-CSIC / UNED agreement with Springer Nature. G. L-Z. was supported by a fellowship from the Fundación Tatiana Pérez de Guzman el Bueno during the preparation of this manuscript.
Author information
Authors and Affiliations
Contributions
All authors contributed to the study conception and design. Literature search, data collection, and analysis were performed by Garazi Laseca-Zaballa and Marcos Ríos-Lago. Review of the literature search and qualitative assessment was made by Jose Antonio Periañez Morales and Genny Lubrini. The first draft of the manuscript was written by Garazi Laseca-Zaballa and Vanesa Simon, and Manuela Martín revised it for intellectual content. Final draft was critically revised and edited by Marcos Ríos-Lago, Genny Lubrini, Cristina Torres, Nuria Martínez, Juan Álvarez-Linera, and Roberto Martinez. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Ethical approval
Not applicable.
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Laseca-Zaballa, G., Lubrini, G., Periañez, J.A. et al. Cognitive outcomes following functional neurosurgery in refractory OCD patients: a systematic review. Neurosurg Rev 46, 145 (2023). https://doi.org/10.1007/s10143-023-02037-w
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s10143-023-02037-w