Abstract
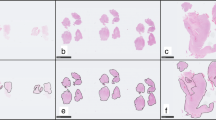
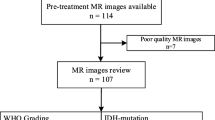
High-grade gliomas (HGG) have high malignancy, high heterogeneity, and a poor prognosis. Tumor purity is an intrinsic feature of the HGG microenvironment and an independent prognostic factor. The purpose of this study was to analyze the correlation of tumor purity with clinicopathological, molecular, and imaging features. We performed a retrospective analysis of 112 patients diagnosed with HGG (grades III and IV) in our center. Eleven regions of interest (ROI) were randomly selected on whole-slide images (WSI, 40 × magnification) based on HGG tissue paraffin sections and hematoxylin–eosin (H&E) staining. Of these 11 ROIs, five ROIs were visually estimated by pathologists and six ROIs were automatically analyzed using ImageJ software. Last, the average tumor purity (%) of the 11 ROIs was calculated. Correlation analysis of tumor purity with clinicopathological, molecular, and imaging features was conducted. Of the 112 patients included in the study, the mean tumor purity of HGG was 70.96%. There were differences in tumor purity between WHO grades III and IV; the tumor purity of grade IV patients (67.59%) was lower than that of grade III patients (76.00%) (p < 0.001). There were also differences in tumor purity between IDH1 mutant and wild type, and the tumor purity of IDH1 mutant patients was higher than that of IDH1 wild-type patients (p = 0.006). The average range of peritumoral edema was about 19.18 mm, and the diameter of edema, ADCmean, and ADCmin were negatively correlated with tumor purity(r = − 0.236, r = − 0.306, and r = − 0.242; p < 0.05). The grade of HGG, IDH1 mutant/wild type, peritumoral edema, and ADC value were correlated with tumor purity. HGG grade, IDH1 mutant/wild type, peritumoral edema, and ADC value can predict tumor purity and indirectly reflect patient prognosis.




Similar content being viewed by others
Data availability
Not applicable.
References
Aran D, Sirota M, Butte AJ (2015) Systematic pan-cancer analysis of tumour purity. Nat Commun 6:8971
Azimi V, Chang YH, Thibault G, Smith J, Tsujikawa T, Kukull B, Jensen B, Corless C, Margolin A, Gray JW (2017) Breast cancer histopathology image analysis pipeline for tumor purity estimation. Proc IEEE Int Symp Biomed Imaging 2017:1137–1140
Biswas NK, Chandra V, Sarkar-Roy N, Das T, Bhattacharya RN, Tripathy LN, Basu SK, Kumar S, Das S, Chatterjee A, Mukherjee A, Basu P, Maitra A, Chattopadhyay A, Basu A, Dhara S (2015) Variant allele frequency enrichment analysis in vitro reveals sonic hedgehog pathway to impede sustained temozolomide response in GBM. Sci Rep 5:7915
Chang PD, Malone HR, Bowden SG, Chow DS, Gill BJA, Ung TH, Samanamud J, Englander ZK, Sonabend AM, Sheth SA, McKhann GM 2nd, Sisti MB, Schwartz LH, Lignelli A, Grinband J, Bruce JN, Canoll P (2017) A multiparametric model for mapping cellularity in glioblastoma using radiographically localized biopsies. AJNR Am J Neuroradiol 38(5):890–898
Hu X, Martinez-Ledesma E, Zheng S, Kim H, Barthel F, Jiang T, Hess KR, Verhaak RGW (2017) Multigene signature for predicting prognosis of patients with 1p19q co-deletion diffuse glioma. Neuro Oncol 19(6):786–795
Li F, Sun H, Li Y, Bai X, Dong X, Zhao N, Meng J, Sun B, Zhang D (2021) High expression of eIF4E is associated with tumor macrophage infiltration and leads to poor prognosis in breast cancer. BMC Cancer 21(1):1305
Li H, Li J, Zhang C, Zhang C, Wang H (2020) TERT mutations correlate with higher TMB value and unique tumor microenvironment and may be a potential biomarker for anti-CTLA4 treatment. Cancer Med 9(19):7151–7160
Lou S, Zhang J, Yin X, Zhang Y, Fang T, Wang Y, Xue Y (2021) Comprehensive characterization of tumor purity and its clinical implications in gastric cancer. Front Cell Dev Biol 9:782529
Lu X, Li C, Xu W, Wu Y, Wang J, Chen S, Zhang H, Huang H, Huang H, Liu W (2021) Malignant tumor purity reveals the driven and prognostic role of CD3E in low-grade glioma Microenvironment. Front Oncol 11:676124
Patel NM, Jo H, Eberhard DA, Yin X, Hayward MC, Stein MK, Hayes DN, Grilley-Olson JE (2019) Improved tumor purity metrics in next-generation sequencing for clinical practice: the integrated interpretation of neoplastic cellularity and sequencing results (IINCaSe) approach. Appl Immunohistochem Mol Morphol 27(10):764–772
Pei L, Jones KA, Shboul ZA, Chen JY, Iftekharuddin KM (2021) Deep neural network analysis of pathology images with integrated molecular data for enhanced glioma classification and grading. Front Oncol 11:668694
Qi T, Meng X, Wang Z, Wang X, Sun N, Ming J, Ren L, Jiang C, Cai J (2021) A voxel-based radiographic analysis reveals the biological character of proneural-mesenchymal transition in glioblastoma. Front Oncol 11:595259
Qin Y, Feng H, Chen M, Wu H, Zheng X (2018) InfiniumPurify: an R package for estimating and accounting for tumor purity in cancer methylation research. Genes Dis 5(1):43–45
Rajan R, Poniecka A, Smith TL, Yang Y, Frye D, Pusztai L, Fiterman DJ, Gal-Gombos E, Whitman G, Rouzier R, Green M, Kuerer H, Buzdar AU, Hortobagyi GN, Symmans WF (2004) Change in tumor cellularity of breast carcinoma after neoadjuvant chemotherapy as a variable in the pathologic assessment of response. Cancer-Am Cancer Soc 100(7):1365–1373
Sallman DA, Komrokji R, Vaupel C, Cluzeau T, Geyer SM, McGraw KL, Al Ali NH, Lancet J, McGinniss MJ, Nahas S, Smith AE, Kulasekararaj A, Mufti G, List A, Hall J, Padron E (2016) Impact of TP53 mutation variant allele frequency on phenotype and outcomes in myelodysplastic syndromes. Leukemia 30(3):666–673
Sallman DA, Padron E (2016) Integrating mutation variant allele frequency into clinical practice in myeloid malignancies. Hematol Oncol Stem Cell Ther 9(3):89–95
Schulze Heuling E, Knab F, Radke J, Eskilsson E, Martinez-Ledesma E, Koch A, Czabanka M, Dieterich C, Verhaak RG, Harms C, Euskirchen P (2017) Prognostic relevance of tumor purity and Interaction with MGMT methylation in glioblastoma. Mol Cancer Res 15(5):532–540
Xiong Y, Xiong Z, Cao H, Li C, Wanggou S, Li X (2020) Multi-dimensional omics characterization in glioblastoma identifies the purity-associated pattern and prognostic gene signatures. Cancer Cell Int 20:37
Yang J, Wang J, Tian S, Wang Q, Zhao Y, Wang B, Cao L, Liang Z, Zhao H, Lian H, Ma J (2021) An integrated analysis of tumor purity of common central nervous system tumors in children based on machine learning methods. Front Genet 12:707802
Zhang C, Cheng W, Ren X, Wang Z, Liu X, Li G, Han S, Jiang T, Wu A (2017) Tumor purity as an underlying key factor in glioma. Clin Cancer Res 23(20):6279–6291
Zhao B, Xia Y, Yang F, Wang Y, Wang Y, Wang Y, Dai C, Wang Y, Ma W (2022) Molecular landscape of IDH-mutant astrocytoma and oligodendroglioma grade 2 indicate tumor purity as an underlying genomic factor. Mol Med 28(1):34
Acknowledgements
We thank all the staff and participants for their contribution to this study. The authors would like to thank Editage (www.editage.cn) for English language editing.
Funding
This work was supported by the National Natural Science Foundation of China (grant number 82071872), the Science and Technology Program Funding Project of Gansu Province (grant number 21YF5FA123), and the Science and Technology Program of Gansu Province (grant number 21JR7RA427).
Author information
Authors and Affiliations
Contributions
Conception and design: Qing Zhou, Peng Zhang, Jiangwei Man, and Junlin Zhou. Acquisition of data: Qing Zhou, Peng Zhang, Jiangwei Man, Bin Zhang, Cai-qiang Xue, and Xiaoai Ke. Analysis and interpretation of data: All authors. Drafting the article: Qing Zhou. Reviewed submitted version of manuscript: Junlin Zhou. Statistical analysis: Qing Zhou, Cai-qiang Xue, and Xiaoai Ke. Administrative/technical/material support: Qing Zhou, Peng Zhang, Jiangwei Man, and Junlin Zhou. Study supervision: Junlin Zhou.
Corresponding author
Ethics declarations
Ethics approval
This study was approved by the Medical Ethics Committee of the Second Hospital of Lanzhou University, and informed consent was waived.
Human and animal ethics
This retrospective study was approved by our hospital’s human research ethics committee and followed the tenets of the Declaration of Helsinki (Ethics Number: 2020A-070).
Consent to participate
All authors have read and approved submission of the revised manuscript. The material in the abstract has not been published and is not being considered for publication elsewhere in whole or in part in any language.
Consent for publication
All of the authors gave consent for the publication of this manuscript.
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Qing Zhou, Peng Zhang, and Jiangwei Man contributed equally to this work as co-first authors.
Rights and permissions
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Zhou, Q., Zhang, P., Man, J. et al. Correlation analysis of tumor purity with clinicopathological, molecular, and imaging features in high-grade gliomas. Neurosurg Rev 45, 3699–3708 (2022). https://doi.org/10.1007/s10143-022-01871-8
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10143-022-01871-8