Abstract
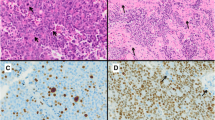
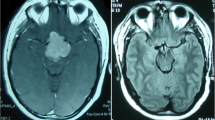
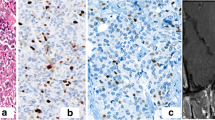
The aim of the study was to assess incidence rate, hormonal activity, and local invasiveness and evaluate outcomes of so-diagnosed atypical pituitary adenomas that underwent endoscopic endonasal surgery at the Division of Neurosurgery of Università degli Studi di Napoli Federico II. According to the 2004 WHO classification, atypical pituitary adenomas are defined by an invasive growth, Ki-67/MIB-1 proliferative index greater than 3 %, high p53 immunoreactivity, and increased mitotic activity. A retrospective analysis of a series of 434 pituitary adenomas that underwent endoscopic endonasal surgery at our department between March 2007 and February 2013 was performed. Fifty adenomas (11.5 %) met the criteria of diagnosis of atypical lesions; 10 (21.6 %) of the 50 patients were recurrent tumors with a previous transsphenoidal surgery. Forty-one (82 %) were macroadenomas, and 21/50 (42 %) showed a clear invasion of the cavernous sinus. Histotype of atypical adenomas figured out to be nonfunctioning in 23 cases (46 %), PRL secreting in 10 cases (20 %), ACTH secreting and GH secreting each apart in 8 patients (16 %), and in a single case a GH/PRL secreting adenoma (2 %). The Ki-67 labeling index ranged from 3.5 to 22.5 % (mean 5.6 %). Tumor recurrence was observed in six cases (12 %) after a mean time of 18 months (range 9–24 months). Mean follow-up was 36.5 months (range 2–80 months). Atypical pituitary adenomas account for ca. 10 % of all pituitary adenomas; these lesions have peculiar features. It should be considered that a strong immunopositivity of p53 and higher Ki-67 LI could predict an increased risk of tumor recurrence, but more studies and larger series are expected to confirm and enlarge the diagnostic and therapeutic management process of these lesions.



Similar content being viewed by others
References
Scheithauer BW, Gaffey TA, Lloyd RV, Sebo TJ, Kovacs KT, Horvath E, Yapicier O, Young WF Jr, Meyer FB, Kuroki T, Riehle DL, Laws ER Jr (2006) Pathobiology of pituitary adenomas and carcinomas. Neurosurgery 59:341–353. doi:10.1227/01.NEU.0000223437.51435.6E, discussion 341–353
Mamelak AN, Carmichael JD, Park P, Bannykh S, Fan X, Bonert HV (2011) Atypical pituitary adenoma with malignant features. Pituitary 14:92–97. doi:10.1007/s11102-008-0151-1
Kovacs K, Scheithauer BW, Horvath E, Lloyd RV (1996) The World Health Organization classification of adenohypophysial neoplasms A proposed five-tier scheme. Cancer 78:502–510. doi:10.1002/(SICI)1097-0142(19960801)78:3<502::AID-CNCR18>3.0.CO;2-2
Chiloiro S, Bianchi A, Doglietto F, de Waure C, Giampietro A, Fusco A, Iacovazzo D, Tartaglione L, Di Nardo F, Signorelli F, Lauriola L, Anile C, Rindi G, Maira G, Pontecorvi A, De Marinis L (2014) Radically resected pituitary adenomas: prognostic role of Ki 67 labeling index in a monocentric retrospective series and literature review. Pituitary 17:267–276. doi:10.1007/s11102-013-0500-6
de Aguiar PH, Aires R, Laws ER, Isolan GR, Logullo A, Patil C, Katznelson L (2010) Labeling index in pituitary adenomas evaluated by means of MIB-1: is there a prognostic role? A critical review. Neurol Res 32:1060–1071. doi:10.1179/016164110X12670144737855
Filippella M, Galland F, Kujas M, Young J, Faggiano A, Lombardi G, Colao A, Meduri G, Chanson P (2006) Pituitary tumour transforming gene (PTTG) expression correlates with the proliferative activity and recurrence status of pituitary adenomas: a clinical and immunohistochemical study. Clin Endocrinol 65:536–543. doi:10.1111/j.1365-2265.2006.02630.x
Kontogeorgos G (2006) Predictive markers of pituitary adenoma behavior. Neuroendocrinology 83:179–188. doi:10.1159/000095526
Steno A, Bocko J, Rychly B, Chorvath M, Celec P, Fabian M, Belan V, Steno J (2014) Nonfunctioning pituitary adenomas: association of Ki-67 and HMGA-1 labeling indices with residual tumor growth. Acta Neurochir 156:451–461. doi:10.1007/s00701-014-1993-0, Discussion 461
Zada G, Lin N, Laws ER Jr (2010) Patterns of extrasellar extension in growth hormone-secreting and nonfunctional pituitary macroadenomas. Neurosurg Focus 29, E4. doi:10.3171/2010.7.FOCUS10155
Zhang X, Horwitz GA, Heaney AP, Nakashima M, Prezant TR, Bronstein MD, Melmed S (1999) Pituitary tumor transforming gene (PTTG) expression in pituitary adenomas. J Clin Endocrinol Metab 84:761–767
Ezzat S, Zheng L, Asa SL (2004) Pituitary tumor-derived fibroblast growth factor receptor 4 isoform disrupts neural cell-adhesion molecule/N-cadherin signaling to diminish cell adhesiveness: a mechanism underlying pituitary neoplasia. Mol Endocrinol 18:2543–2552. doi:10.1210/me.2004-0182
Kawamoto H, Kawamoto K, Mizoue T, Uozumi T, Arita K, Kurisu K (1996) Matrix metalloproteinase-9 secretion by human pituitary adenomas detected by cell immunoblot analysis. Acta Neurochir 138:1442–1448
Wierinckx A, Auger C, Devauchelle P, Reynaud A, Chevallier P, Jan M, Perrin G, Fevre-Montange M, Rey C, Figarella-Branger D, Raverot G, Belin MF, Lachuer J, Trouillas J (2007) A diagnostic marker set for invasion, proliferation, and aggressiveness of prolactin pituitary tumors. Endocr Relat Cancer 14:887–900. doi:10.1677/ERC-07-0062
DeLellis RA (2004) Pathology and genetics of tumours of endocrine organs. World Health Organization classification of tumours, vol 8. IARC Press, Lyon
Figarella-Branger D, Trouillas J (2006) The new WHO classification of human pituitary tumors: comments. Acta Neuropathol 111:71–72. doi:10.1007/s00401-005-1099-0
Grossman AB (2006) The 2004 World Health Organization classification of pituitary tumors: is it clinically helpful? Acta Neuropathol 111:76–77. doi:10.1007/s00401-005-1101-x
Kleinschmidt-DeMasters BK (2006) Subtyping does matter in pituitary adenomas. Acta Neuropathol 111:84–85. doi:10.1007/s00401-005-1105-6
Kontogeorgos G (2006) Innovations and controversies in the WHO classification of pituitary adenomas. Acta Neuropathol 111:73–75. doi:10.1007/s00401-005-1100-y
Kovacs K (2006) The 2004 WHO classification of pituitary tumors: comments. Acta Neuropathol 111:62–63. doi:10.1007/s00401-005-1095-4
Laws ER Jr, Lopes MB (2006) The new WHO classification of pituitary tumors: highlights and areas of controversy. Acta Neuropathol 111:80–81. doi:10.1007/s00401-005-1103-8
Marucci G, Faustini-Fustini M, Righi A, Pasquini E, Frank G, Agati R, Foschini MP (2009) Thyrotropin-secreting pituitary tumours: significance of “atypical adenomas” in a series of 10 patients and association with Hashimoto thyroiditis as a cause of delay in diagnosis. J Clin Pathol 62:455–459. doi:10.1136/jcp.2008.061523
Ogawa Y, Ikeda H, Tominaga T (2009) Clinicopathological study of prognostic factors in patients with pituitary adenomas and Ki-67 labeling index of more than 3%. J Endocrinol Invest 32:581–584. doi:10.3275/6300
Perry A, Scheithauer BW (2006) Commentary: classification and grading of pituitary tumors. Observations of two working neuropathologists. Acta Neuropathol 111:68–70. doi:10.1007/s00401-005-1098-1
Saeger W, Ludecke DK, Buchfelder M, Fahlbusch R, Quabbe HJ, Petersenn S (2007) Pathohistological classification of pituitary tumors: 10 years of experience with the German Pituitary Tumor Registry. Eur J Endocrinol Eur Fed Endocrine Societies 156:203–216. doi:10.1530/eje.1.02326
Wolfsberger S, Knosp E (2006) Comments on the WHO 2004 classification of pituitary tumors. Acta Neuropathol 111:66–67. doi:10.1007/s00401-005-1097-2
Yildirim AE, Divanlioglu D, Nacar OA, Dursun E, Sahinoglu M, Unal T, Belen AD (2013) Incidence, hormonal distribution and postoperative follow up of atypical pituitary adenomas. Turkish Neurosurg 23:226–231. doi:10.5137/1019-5149.JTN.6828-12.1
Zada G, Woodmansee WW, Ramkissoon S, Amadio J, Nose V, Laws ER Jr (2011) Atypical pituitary adenomas: incidence, clinical characteristics, and implications. J Neurosurg 114:336–344. doi:10.3171/2010.8.JNS10290
Ogawa Y, Tominaga T (2008) A case of atypical thyrotroph cell adenoma, which re-grew within 3 months after surgery and required multimodal treatment. J Neuro-Oncol 87:91–95. doi:10.1007/s11060-007-9492-2
Losa M, Mazza E, Terreni MR, McCormack A, Gill AJ, Motta M, Cangi MG, Talarico A, Mortini P, Reni M (2010) Salvage therapy with temozolomide in patients with aggressive or metastatic pituitary adenomas: experience in six cases. Eur J Endocrinol Eur Fed Endocrine Soc 163:843–851. doi:10.1530/EJE-10-0629
Knosp E, Steiner E, Kitz K, Matula C (1993) Pituitary adenomas with invasion of the cavernous sinus space: a magnetic resonance imaging classification compared with surgical findings. Neurosurgery 33:610–617, discussion 617–618
Aflorei ED, Korbonits M (2014) Epidemiology and etiopathogenesis of pituitary adenomas. J Neuro-Oncol 117:379–394. doi:10.1007/s11060-013-1354-5
Jane JA, Jr., Thapar K, Laws ER, Jr. (2011) Pituitary tumors: functioning and nonfunctioning. In: Winn RH (ed) Youmans Neurological Surgery. 6th edn. Elsevier Saunders, pp 1476–1510
Cappabianca P, Esposito F, Cavallo LM, Colao A (2012) Endocrinologically silent pituitary tumors. In: Quiñones-Hinojosa A (ed) Schmidek and Sweet operative neurosurgical techniques indications, methods and results. vol 21, 6th edn. Elsevier Saunders, pp 246–256
Chen L, White WL, Spetzler RF, Xu B (2011) A prospective study of nonfunctioning pituitary adenomas: presentation, management, and clinical outcome. J Neuro-Oncol 102:129–138. doi:10.1007/s11060-010-0302-x
Colao A, Di Somma C, Pivonello R, Faggiano A, Lombardi G, Savastano S (2008) Medical therapy for clinically non-functioning pituitary adenomas. Endocr Relat cancer 15:905–915
Ferrante E, Ferraroni M, Castrignano T, Menicatti L, Anagni M, Reimondo G, Del Monte P, Bernasconi D, Loli P, Faustini-Fustini M, Borretta G, Terzolo M, Losa M, Morabito A, Spada A, Beck-Peccoz P, Lania AG (2006) Non-functioning pituitary adenoma database: a useful resource to improve the clinical management of pituitary tumors. Eur J Endocrinol Eur Fed Endocr Soc 155:823–829
Matsuyama J (2012) Ki-67 expression for predicting progression of postoperative residual pituitary adenomas: correlations with clinical variables. Neurol Med Chir 52:563–569
Cavallo LM, Solari D, Tasiou A, Esposito F, de Angelis M, D'Enza AI, Cappabianca P (2012) Endoscopic endonasal transsphenoidal removal of recurrent and regrowing pituitary adenomas: experience on a 59-patient series. World Neurosurg. doi:10.1016/j.wneu.2012.10.008
Benveniste RJ, King WA, Walsh J, Lee JS, Delman BN, Post KD (2005) Repeated transsphenoidal surgery to treat recurrent or residual pituitary adenoma. J Neurosurg 102:1004–1012. doi:10.3171/jns.2005.102.6.1004
Chang EF, Sughrue ME, Zada G, Wilson CB, Blevins LS Jr, Kunwar S (2010) Long term outcome following repeat transsphenoidal surgery for recurrent endocrine-inactive pituitary adenomas. Pituitary 13:223–229. doi:10.1007/s11102-010-0221-z
Greenman Y, Ouaknine G, Veshchev I, Reider G II, Segev Y, Stern N (2003) Postoperative surveillance of clinically nonfunctioning pituitary macroadenomas: markers of tumour quiescence and regrowth. Clin Endocrinol 58:763–769
Laws ER Jr, Fode NC, Redmond MJ (1985) Transsphenoidal surgery following unsuccessful prior therapy. An assessment of benefits and risks in 158 patients. J Neurosurg 63:823–829. doi:10.3171/jns.1985.63.6.0823
Long H, Beauregard H, Somma M, Comtois R, Serri O, Hardy J (1996) Surgical outcome after repeated transsphenoidal surgery in acromegaly. J Neurosurg 85:239–247. doi:10.3171/jns.1996.85.2.0239
Noh TW, Jeong HJ, Lee MK, Kim TS, Kim SH, Lee EJ (2009) Predicting recurrence of nonfunctioning pituitary adenomas. J Clin Endocrinol Metab 94:4406–4413. doi:10.1210/jc.2009-0471
Wilson CB, Dempsey LC (1978) Transsphenoidal microsurgical removal of 250 pituitary adenomas. J Neurosurg 48:13–22
Gejman R, Swearingen B, Hedley-Whyte ET (2008) Role of Ki-67 proliferation index and p53 expression in predicting progression of pituitary adenomas. Hum Pathol 39:758–766. doi:10.1016/j.humpath.2007.10.004
Oliveira MC, Marroni CP, Pizarro CB, Pereira-Lima JF, Barbosa-Coutinho LM, Ferreira NP (2002) Expression of p53 protein in pituitary adenomas. Braz J Med Biol Res 35:561–565
Acknowledgments
No financial support was received. The manuscript or parts of it are not under consideration by another journal or electronic publication and have not been previously published or presented at a meeting. The authors have no proprietary or commercial interest in any materials or method discussed in this article.
Author contributions
Conception and design of the study: Luigi Maria Cavallo and Marialaura Del Basso De Caro; execution of the study, writing, drafting, and revising process: Domenico Solari, Alessandro Villa, and Francesca Pagliuca; data collection and literature research: Alessandro Villa and Francesca Pagliuca; analysis and interpretation of histopathological findings: Marialaura Del Basso De Caro and Elia Guadagno; and drafting and revising process as well as the final approval of the article: Guido Pettinato, Annamaria Colao, and Paolo Cappabianca.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
All authors certify that they have no affiliations with or involvement in any organization or entity with any financial interest (such as honoraria; educational grants; participation in speakers’ bureaus; membership, employment, consultancies, stock ownership, or other equity interest; and expert testimony or patent-licensing arrangements), or nonfinancial interest (such as personal or professional relationships, affiliations, knowledge, or beliefs) in the subject matter or materials discussed in this manuscript.
Rights and permissions
About this article
Cite this article
Del Basso De Caro, M., Solari, D., Pagliuca, F. et al. Atypical pituitary adenomas: clinical characteristics and role of ki-67 and p53 in prognostic and therapeutic evaluation. A series of 50 patients. Neurosurg Rev 40, 105–114 (2017). https://doi.org/10.1007/s10143-016-0740-9
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10143-016-0740-9