Abstract
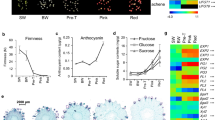
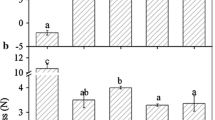
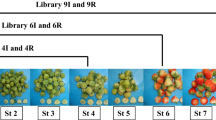
Strawberry is an ideal model for studying the molecular biology of the development and ripening of non-climacteric fruits. Hormonal regulation of gene expression along all these processes in strawberries is still to be fully elucidated. Although auxins and ABA have been pointed out as the major regulatory hormones, few high-throughput analyses have been carried out to date. The role for ethylene and gibberellins as regulatory hormones during the development and ripening of the strawberry fruit remain still elusive. By using a custom-made and high-quality oligo microarray platform done with over 32,000 probes including all of the genes actually described in the strawberry genome, we have analysed the expression of genes during the development and ripening in the receptacles of these fruits. We classify these genes into two major groups depending upon their temporal and developmental expression. First group are genes induced during the initial development stages. The second group encompasses genes induced during the final maturation and ripening processes. Each of these two groups has been also divided into four sub-groups according their pattern of hormonal regulation. By analyzing gene expression, we clearly show that auxins and ABA are the main and key hormones that combined or independently are responsible of the development and ripening process. Auxins are responsible for the receptacle fruit development and, at the same time¸ prevent ripening by repressing crucial genes. ABA regulates the expression of the vast majority of genes involved in the ripening. The main genes expressed under the control of these hormones are presented and their physiological rule discussed. We also conclude that ethylene and gibberellins do not seem to play a prominent role during these processes.



Similar content being viewed by others
Abbreviations
- ABA:
-
Abscisic acid
- Ct :
-
Threshold cycle
- EST:
-
Expressed sequence tag
- G1:
-
Green1 development stage
- G3:
-
Green3 development stage
- GAs:
-
Gibberellins
- GO:
-
Gene ontology
- IAA:
-
Indole-3-acetic acid
- MEP:
-
2-C-methyl-d-erithritol 4-phosphate pathway
- MVA:
-
Mevalonate pathway
- NCED:
-
9-cis-Epoxycarotenoid dioxygenase
- NDGA:
-
Nordihydroguaiaretic acid
- qRT-PCR:
-
Quantitative real-time-polymerase chain reaction
- R:
-
Red ripe ripening stage
- TFs:
-
Transcription factors
- Tm:
-
Melting temperature
- u.a.i:
-
Arbitrary units of intensity
References
Aharoni A, Keizer LC, Bouwmeester HJ, Sun Z, Alvarez-Huerta M, Verhoeven HA, Blaas J, van Houwelingen AM, De Vos RC, van der Voet H, Jansen RC, Guis M, Mol J, Davis RW, Schena M, van Tunen AJ, O’Connell AP (2000) Identification of the SAAT gene involved in strawberry flavour biogenesis by use of DNA microarrays. Plant Cell 12:647–662
Aharoni A, Keizer LCP, Van Den Broeck HC, Blanco-Portales R, Muñoz-Blanco J, Bois G, Smit P, De Vos RCH, O’Connell AP (2002) Novel insight into vascular, stress, and auxin-dependent and -independent gene expression programs in strawberry, a non-climacteric fruit. Plant Physiol 129:1019–1031
Asif M, Dhawan P, Nath P (2000) A simple procedure for the isolation of high quality RNA from ripening banana fruit. Plant Mol Biol Rep 18:109–115
Benítez-Burraco A, Blanco-Portales R, Redondo-Nevado J, Bellido ML, Moyano E, Caballero J-L, Muñoz-Blanco J (2003) Cloning and characterization of two ripening-related strawberry (Fragaria x ananassa cv. Chandler) pectate lyase genes. J Exp Bot 54:633–645
Benjamini Y, Hochberg H (1995) Controlling the false discovery rate: a practical and powerful approach to multiple testing. J R Statist Soc B 1:289–300
Bitto E, Bingman CA, Bittova L, Houston NL, Boston RS, Fox BG, Phillips GN (2009) X-ray structure of ILL2, an auxin-conjugate amidohydrolase from Arabidopsis thaliana. Proteins 74:61–71
Blanco-Portales R, Medina-Escobar N, López-Ráez JA, González-Reyes JA, Villalba JM, Moyano E, Caballero JL, Muñoz-Blanco J (2002) Cloning, expression and immunolocalization pattern of a cinnamyl alcohol dehydrogenase gene from strawberry (Fragaria x ananassa cv. Chandler). J Exp Bot 53:1723–1734
Bombarely A, Merchante C, Csukasi F, Cruz-Rus E, Caballero JL, Medina-Escobar N, Blanco-Portales R, Botella MA, Muñoz-Blanco J, Sánchez-Sevilla JF, Valpuesta V (2010) Generation and analysis of ESTs from strawberry (Fragaria xananassa) fruits and evaluation of their utility in genetic and molecular studies. BMC Genomics 11:503
Böttcher C, Burbidge CA, Boss PK, Davies C (2013) Interactions between ethylene and auxin are crucial to the control of grape (Vitis vinifera L.) berry ripening. BMC Plant Biol 13:222
Bustamante CA, Civello PM, Martínez GA (2009) Cloning of the promoter region of β-xylosidase (FaXyl1) gene and effect of plant growth regulators on the expression of FaXyl1 in strawberry fruit. Plant Sci 177:49–56
Castillejo C, de la Fuente JI, Iannetta P, Botella MA, Valpuesta V (2004) Pectin esterase gene family in strawberry fruit: study of FaPE1, a ripening-specific isoform. J Exp Bot 55:909–918
Cazzonelli CI, Pogson BJ (2010) Source to sink: regulation of carotenoid biosynthesis in plants. Trends Plant Sci 15:266–274
Chai Y-M, Jia H-F, Li C-L, Dong Q-H, Shen Y-Y (2011) FaPYR1 is involved in strawberry fruit ripening. J Exp Bot 62:5079–5089
Chen J-Y, Liu D-J, Jiang Y-M, Zhao M-L, Shan W, Kuang J-F, Ahn J-H, (2011) Molecular Characterization of a Strawberry FaASR Gene in Relation to Fruit Ripening. PLoS ONE 6(9):e24649
Chen J, Linchun M, Wenjing L, Tiejin Y, Zisheng L (2015) Transcriptome profiling of postharvest strawberry fruit in response to exogenous auxin and abscisic acid. Planta. doi:10.1007/s00425-015-2402-5
Cherian S, Figueroa CR, Nair H (2014) ‘Movers and shakers’ in the regulation of fruit ripening: a cross-dissection of climacteric versus non-climacteric fruit. J Exp Bot 65:4705–4722
Chervin C, El-Kereamy A, Roustan J-P, Latché A, Lamon J, Bouzayen M (2004) Ethylene seems required for the berry development and ripening in grape, a non-climacteric fruit. Plant Sci 167:1301–1305
Conesa A, Götz S, García-Gómez JM, Terol J, Talón M, Robles M (2005) Blast2GO: a universal tool for annotation, visualization and analysis in functional genomics research. Bioinformatics 21:3674–3676
Conn S, Curtin C, Bézier A, Franco C, Zhang W (2008) Purification, molecular cloning, and characterization of glutathione S-transferases (GSTs) from pigmented Vitis vinifera L. cell suspension cultures as putative anthocyanin transport proteins. J Exp Bot 59:3621–3634
Creelman RA, Bell E, Mullet JE (1992) Involvement of a lipoxygenase-like enzyme in abscisic acid biosynthesis. Plant Physiol 99:1258–1260
Csukasi F, Osorio S, Gutierrez JR, Kitamura J, Giavalisco P, Nakajima M, Fernie AR, Rathjen JP, Botella MA, Valpuesta V, Medina-Escobar N (2011) Gibberellin biosynthesis and signalling during development of the strawberry receptacle. New Phytol 191:376–390
Cumplido-Laso G, Medina-Puche L, Moyano E, Hoffmann T, Sinz Q, Ring L, Studart-Wittkowski C, Luis Caballero J, Schwab W, Munoz-Blanco J, Blanco-Portales R (2012) The fruit ripening-related gene FaAAT2 encodes an acyl transferase involved in strawberry aroma biogenesis. J Exp Bot 63:4275–4290
Cutler SR, Rodríguez PL, Finkelstein RR, Abrams SR (2010) Abscisic acid: emergence of a core signaling network. Annu Rev Plant Biol 61:651–679
Daminato M, Guzzo F, Casadoro G (2013) A shatterproof-like gene controls ripening in non-climacteric strawberries, and auxin and abscisic acid antagonistically affect its expression. J Exp Bot 64:3775–3786
Dreher T, Poovaiah B (1982) Changes in auxin content during development in strawberry fruits. In: Hortscience, vol 17. Amer Soc horticultural science 701 North Saint Asaph street, Alexandria, VA 22314-1998, pp 475-475
Encinas-Villarejo S, Maldonado AM, Amil-Ruiz F, de los Santos B, Romero F, Pliego-Alfaro F, Muñoz-Blanco J, Caballero JL (2009) Evidence for a positive regulatory role of strawberry (Fragaria x ananassa) Fa WRKY1 and arabidopsis at WRKY75 proteins in resistance. J Exp Bot 60:3043–3065
Fait A, Hanhineva K, Beleggia R, Dai N, Rogachev I, Nikiforova VJ, Fernie AR, Aharoni A (2008) Reconfiguration of the achene and receptacle metabolic networks during strawberry fruit development. Plant Physiol 148:730–750
Fortes A, Teixeira R, Agudelo-Romero P (2015) Complex interplay of hormonal signals during grape berry ripening. Molecules 20:9326
Giovannoni J (2001) Molecular biology of fruit maturation and ripening. Annu Rev Plant Phys 52:725–749
Giovannoni JJ (2004) Genetic regulation of fruit development and ripening. Plant Cell Online 16:S170–S180
Giovannoni JJ (2007) Fruit ripening mutants yield insights into ripening control. Curr Opin Plant Biol 10:283–289
Gomez C, Terrier N, Torregrosa L, Vialet S, Fournier-Level A, Verriès C, Souquet J-M, Mazauric J-P, Klein M, Cheynier V, Ageorges A (2009) Grapevine MATE-type proteins act as vacuolar H + -dependent acylated anthocyanin transporters. Plant Physiol 150:402–415
Gomez C, Conejero G, Torregrosa L, Cheynier V, Terrier N, Ageorges A (2011) In vivo grapevine anthocyanin transport involves vesicle-mediated trafficking and the contribution of anthoMATE transporters and GST. Plant J 67:960–970
Guzmán P (2012) The prolific ATL family of RING-H2 ubiquitin ligases. Plant Signal Behav 7:1014–1021
Hua Z, Vierstra RD (2011) The cullin-ring ubiquitin-protein ligases. Annu Rev Plant Biol 62:299–334
Hua C, Linling L, Feng X, Yan W, Honghui Y, Conghua W, Shaobing W, Zhiqin L, Juan H, Yuping W, Shuiyuan C, Fuliang C (2013) Expression patterns of an isoflavone reductase-like gene and its possible roles in secondary metabolism in Ginkgo biloba. Plant Cell Rep 32:637–650
Hugueney P, Provenzano S, Verriès C, Ferrandino A, Meudec E, Batelli G, Merdinoglu D, Cheynier V, Schubert A, Ageorges A (2009) A novel cation-dependent O-methyltransferase involved in anthocyanin methylation in grapevine. Plant Physiol 150:2057–2070
Iannetta PPM, Laarhoven L-J, Medina-Escobar N, James EK, McManus MT, Davies HV, Harren FJM (2006) Ethylene and carbon dioxide production by developing strawberries show a correlative pattern that is indicative of ripening climacteric fruit. Physiol Plantarum 127:247–259
Ibdah M, Pichersky E (2009) Arabidopsis Chy1 null mutants are deficient in benzoic acid-containing glucosinolates in the seeds. Plant Biol (Stuttg) 11:574–581
Jia H-F, Chai Y-M, Li C-L, Lu D, Luo J-J, Qin L, Shen Y-Y (2011) Abscisic acid plays an important role in the regulation of strawberry fruit ripening. Plant Physiol 157:188–199
Jia H, Wang Y, Sun M, Li B, Han Y, Zhao Y, Li X, Ding N, Li C, Ji W, Jia W (2013) Sucrose functions as a signal involved in the regulation of strawberry fruit development and ripening. New Phytol 198:453–465
Khan M, Xu H, Hepworth SR (2014) Blade-on-petiole genes: setting boundaries in development and defense. Plant Sci 215–216:157–171
Klee HJ, Giovannoni JJ (2011) Genetics and control of tomato fruit ripening and quality attributes. Annu Rev Genet 45:41–59
Kong Y, Peña MJ, Renna L, Avci U, Pattathil S, Tuomivaara ST, Li X, Reiter W-D, Brandizzi F, Hahn MG, Darvill AG, York WS, O’Neill MA (2015) Galactose-depleted xyloglucan is dysfunctional and leads to dwarfism in arabidopsis. Plant Physiol 167:1296–1306
Koyama K, Sadamatsu K, Goto-Yamamoto N (2010) Abscisic acid stimulated ripening and gene expression in berry skins of the cabernet sauvignon grape. Funct Integr Genomics 10:367–381
Kuhn N, Guan L, Dai ZW, Wu B-H, Lauvergeat V, Gomès E, Li S-H, Godoy F, Arce-Johnson P, Delrot S (2014) Berry ripening: recently heard through the grapevine. J Exp Bot 65:4543–4559
Kumar R, Khurana A, Sharma AK (2014) Role of plant hormones and their interplay in development and ripening of fleshy fruits. J Exp Bot 65:4561–4575
Lange PR, Eastmond PJ, Madagan K, Graham IA (2004) An arabidopsis mutant disrupted in valine catabolism is also compromised in peroxisomal fatty acid beta-oxidation. FEBS Lett 571:147–153
Lawlor DW, Paul MJ (2014) Source/sink interactions underpin crop yield: the case for trehalose 6-phosphate/SnRK1 in improvement of wheat. Front Plant Sci 5:418
Lee S, Chung EJ, Joung YH, Choi D (2010) Non-climacteric fruit ripening in pepper: increased transcription of EIL-like genes normally regulated by ethylene. Funct Integr Genomics 10:135–146
Leshem YY, Pinchasov Y (2000) Non-invasive photoacoustic spectroscopic determination of relative endogenous nitric oxide and ethylene content stoichiometry during the ripening of strawberries Fragaria anannasa (Duch.) and avocados Persea americana (Mill.). J Exp Bot 51:1471–1473
Li C, Liu Z, Zhang Q, Wang R, Xiao L, Ma H, Chong K, Xu Y (2012) SKP1 is involved in abscisic acid signalling to regulate seed germination, stomatal opening and root growth in Arabidopsis thaliana. Plant Cell Environ 35:952–965
Manríquez D, El-Sharkawy I, Flores FB, El-Yahyaoui F, Regad F, Bouzayen M, Latché A, Pech J-C (2006) Two highly divergent alcohol dehydrogenases of melon exhibit fruit ripening-specific expression and distinct biochemical characteristics. Plant Mol Biol 61:675–685
Martínez GA, Chaves AR, Añón MC (1996) Effect of exogenous application of gibberellic acid on colour change and phenylalanine ammonia-lyase, chlorophyllase, and peroxidase activities during ripening of strawberry fruit (Fragaria x ananassa Duch.). J Plant Growth Regul 15:139–146
Martinoia E, Maeshima M, Neuhaus HE (2007) Vacuolar transporters and their essential role in plant metabolism. J Exp Bot 58:83–102
Mata R (2007) Flavonoids, Chemistry, Biochemistry and Applications By Ø. M. Andersen (University of Bergen) and K. R. Markham (Industrial Research Ltd.). CRC Press/Taylor & Francis, Boca Raton. 2006. xiv + 1237 pp. 7 × 101/4 in. $249.95. ISBN 0-8493-2021-6. J Nat Prod 70: 140-140
McAtee P, Karim S, Schaffer R, David K (2013) A dynamic interplay between phytohormones is required for fruit development, maturation, and ripening. Front Plant Sci 4:79
Medina-Escobar N, Cárdenas J, Moyano E, Caballero JL, Muñoz-Blanco J (1997) Cloning, molecular characterization and expression pattern of a strawberry ripening-specific cDNA with sequence homology to pectate lyase from higher plants. Plant Mol Biol 34:867–877
Medina-Puche L, Cumplido-Laso G, Amil-Ruiz F, Hoffmann T, Ring L, Rodríguez-Franco A, Caballero JL, Schwab W, Muñoz-Blanco J, Blanco-Portales R (2014) MYB10 plays a major role in the regulation of flavonoid/phenylpropanoid metabolism during ripening of Fragaria × ananassa fruits. J Exp Bot 65:401–417
Medina-Puche L, Molina-Hidalgo FJ, Boersma M, Schuurink RC, López-Vidriero I, Solano R, Franco-Zorrilla J-M, Caballero JL, Blanco-Portales R, Muñoz-Blanco J (2015) An R2R3-MYB transcription factor regulates eugenol production in ripe strawberry fruit receptacles. Plant Physiol 168:598–614
Merchante C, Vallarino JG, Osorio S, Aragüez I, Villarreal N, Ariza MT, Martínez GA, Medina-Escobar N, Civello MP, Fernie AR, Botella MA, Valpuesta V (2013) Ethylene is involved in strawberry fruit ripening in an organ-specific manner. J Exp Bot 64:4421–4439
Molina-Hidalgo FJ, Franco AR, Villatoro C, Medina-Puche L, Mercado JA, Hidalgo MA, Monfort A, Luis Caballero J, Munoz-Blanco J, Blanco-Portales R (2013) The strawberry (Fragariaananassa) fruit-specific rhamnogalacturonate lyase 1 (FaRGLyase1) gene encodes an enzyme involved in the degradation of cell-wall middle lamellae. J Exp Bot 64:1471–1483
Molina-Hidalgo FJ, Medina-Puche L, Gelis S, Ramos J, Sabir F, Soveral G, Prista C, Iglesias-Fernández R, Caballero JL, Muñoz-Blanco J, Blanco-Portales R (2015) Functional characterization of FaNIP1;1 gene, a ripening-related and receptacle-specific aquaporin in strawberry fruit. Plant Sci 238:198–211
Mukhopadhyay D, Riezman H (2007) Proteasome-independent functions of ubiquitin in endocytosis and signaling. Science 315:201–205
Opazo MC, Lizana R, Pimentel P, Herrera R, Moya-León MA (2013) Changes in the mRNA abundance of FcXTH1 and FcXTH2 promoted by hormonal treatments of Fragaria chiloensis fruit. Postharvest Biol Tec 77:28–34
Pedersen S, Amtssygehus A (2001) Multiplex relative gene expression analysis by real-time RT-PCR using the iCycler iQ™ detection system. BioRadiations (BioRad) 107:10–11
Perkins-Veazie P (1995) Growth and ripening of strawberry fruit. Hortic Rev 17:267–297
Perkins-Veazie PM, Huber DJ, Brecht JK (1996) In vitro growth and ripening of strawberry fruit in the presence of ACC, STS or propylene. Ann Appl Biol 128:105–116
Pillet J, Yu H-W, Chambers AH, Whitaker VM, Folta KM (2015) Identification of candidate flavonoid pathway genes using transcriptome correlation network analysis in ripe strawberry (Fragaria × ananassa) fruits. J Exp Bot 66:4455–4467
Raab T, López-Ráez JA, Klein D, Caballero JL, Moyano E, Schwab W, Muñoz-Blanco J (2006) FaQR, required for the biosynthesis of the strawberry flavour compound 4-hydroxy-2,5-dimethyl-3(2H)-furanone, encodes an enone oxidoreductase. Plant Cell 18:1023–1037
Redondo-Nevado J, Moyano E, Medina-Escobar N, Caballero JL, Muñoz-Blanco J (2001) A fruit-specific and developmentally regulated endopolygalacturonase gene from strawberry (Fragaria x ananassa cv. Chandler). J Exp Bot 52:1941–1945
Rodríguez L, Gonzalez-Guzman M, Diaz M, Rodrígues A, Izquierdo-Garcia AC, Peirats-Llobet M, Fernandez MA, Antoni R, Fernandez D, Marquez JA, Mulet JM, Albert A, Rodríguez PL (2014) C2-domain abscisic acid-related proteins mediate the interaction of PYR/PYL/RCAR abscisic acid receptors with the plasma membrane and regulate abscisic acid sensitivity in arabidopsis. Plant Cell 26:4802–4820
Salvatierra A, Pimentel P, Moya-León MA, Herrera R (2013) Increased accumulation of anthocyanins in Fragaria chiloensis fruits by transient suppression of FcMYB1 gene. Phytochemistry 90:25–36
Schwab W, Davidovich-Rikanati R, Lewinsohn E (2008) Biosynthesis of plant-derived flavour compounds. Plant J 54:712–732
Seki M, Ishida J, Narusaka M, Fujita M, Nanjo T, Umezawa T, Kamiya A, Nakajima M, Enju A, Sakurai T, Satou M, Akiyama K, Yamaguchi-Shinozaki K, Carninci P, Kawai J, Hayashizaki Y, Shinozaki K (2002) Funct Integr Genomics 2:282–291
Showalter AM, Keppler B, Lichtenberg J, Gu D, Welch LR (2010) A bioinformatics approach to the identification, classification, and analysis of hydroxyproline-rich glycoproteins. Plant Physiol 153:485–513
Symons GM, Davies C, Shavrukov Y, Dry IB, Reid JB, Thomas MR (2006) Grapes on steroids. Brassinosteroids are involved in grape berry ripening. Plant Physiol 140:150–158
Symons GM, Chua Y-J, Ross JJ, Quittenden LJ, Davies NW, Reid JB (2012) Hormonal changes during non-climacteric ripening in strawberry. J Exp Bot 63:4741–4750
Trainotti L, Spolaore S, Pavanello A, Baldan B, Casadoro G (1999) A novel E-type endo-β-1,4-glucanase with a putative cellulose-binding domain is highly expressed in ripening strawberry fruits. Plant Mol Biol 40:323–332
Trainotti L, Spinello R, Piovan A, Spolaore S, Casadoro G (2001) beta-Galactosidases with a lectin-like domain are expressed in strawberry. J Exp Bot 52:1635–1645
Verweij W, Spelt C, Di Sansebastiano G-P, Vermeer J, Reale L, Ferranti F, Koes R, Quattrocchio F (2008) An H+ P-ATPase on the tonoplast determines vacuolar pH and flower colour. Nat Cell Biol 10:1456–1462
Villarreal NM, Bustamante CA, Civello PM, Martínez GA (2010) Effect of ethylene and 1-MCP treatments on strawberry fruit ripening. J Sci Food Agr 90:683–689
Waters DLE, Holton TA, Ablett EM, Lee LS, Henry RJ (2005) cDNA microarray analysis of developing grape (Vitis vinifera cv. Shiraz) berry skin. Funct Integr Genomics 5:40–58
Wechter WP, Levi A, Harris KR, Davis AR, Fei Z, Katzir N, Giovannoni JJ, Salman-Minkov A, Hernandez A, Thimmapuram J, Tadmor Y, Portnoy V, Trebitsh T (2008) Gene expression in developing watermelon fruit. BMC Genomics 9:275
Wurmbach E (2009) Real-time PCR: current technology and applications, Chapter 10 validation of array data. Caister Academic Press, London
Xu P, Cai W (2014) RAN1 is involved in plant cold resistance and development in rice (Oryza sativa). J Exp Bot 65:3277–3287
Zang A, Xu X, Neill S, Cai W (2010) Overexpression of OsRAN2 in rice and Arabidopsis renders transgenic plants hypersensitive to salinity and osmotic stress. J Exp Bot 61:777–789
Zhang D, Aravind L (2010) Identification of novel families and classification of the C2 domain superfamily elucidate the origin and evolution of membrane targeting activities in eukaryotes. Gene 469:18–30
Zhang Y, Li B, Huai D, Zhou Y, Kliebenstein DJ (2015) The conserved transcription factors, MYB115 and MYB118, control expression of the newly evolved benzoyloxy glucosinolate pathway in arabidopsis thaliana. Front Plant Sci 6:343
Acknowledgments
This work was supported by a grant from the Ministerio Español de Ciencia e Innovación (MICINN) (BIO2010-19322). LMP and RBP are grateful for the award of a Ph.D. fellowship within the framework of Formación del Personal Universitario (FPU) program implemented by the Ministerio de Educación y Ciencia (MEC) (Spain) and a post-doctoral contract from Campus de Excelencia Internacional Agroalimentario (CEIA3) from University of Córdoba, respectively.
Author information
Authors and Affiliations
Corresponding author
Additional information
Laura Medina-Puche and Rosario Blanco-Portales contributed equally to this work.
Additional files
Below is the link to the electronic supplementary material.
ESM 1
Table S1. List of primer sequences used for validation through qRT-PCR. Fw: forward; Rv: reverse. (DOCX 26 kb)
ESM 2
Table S2. Gene expression values of different housekeeping genes obtained by qRT-PCR analysis. Experiment A: throughout the development and ripening of strawberry fruit. Experiment B: in vegetative tissues against strawberry red fruit. Experiment C: in de-achened G2 fruits against control G2 fruits. Reactions normalized using the Ct value corresponding to a strawberry interspacer 26S-18S gene for quantification as described in Materials and Methods. Ct values used as reference were assigned an arbitrary value equal to unity. The increase in mRNA value was relative to the receptacle G1-Ct value for experiment A, to the receptacle R-Ct value for experiment B and to the control G2-fruits-Ct for experiment C. G1: small-sized green fruits; G2: middle-sized green fruits; G3: full-sized green fruits; W: white fruits; R: full-ripe red fruits. (XLSX 11 kb)
ESM 3
Table S3. Complete list of genes expressed during fruit ripening. Data provided correspond to strawberry genes whose expression change from green inmature to red ripe fruit receptacles. Gene ID as reported in Fragaria vesca Genome Database (http://www.strawberrygenome.org/). (XLSX 1808 kb)
ESM 4
Table S4. List of genes deregulated after ABA depletion by using NDGA. List of strawberry genes whose expression is modified after NDGA treatment respect to untreated control fruits. Gene ID reported in Fragaria vesca Genome Database (http://www.strawberrygenome.org/). (XLSX 1157 kb)
ESM 5
Table S5. Complete list of differential gene expression in red ripe strawberry fruit receptacles compared to vegetative tissues. Gene ID reported in Fragaria vesca Genome Database (http://www.strawberrygenome.org/). (XLSX 1218 kb)
ESM 6
Table S6. Correlation coefficients (r-value) and coefficients of determination (r-squared) calculated with data obtained from each of the three biological replicates under the experimental conditions summarized in Fig. 1. This was calculated by fitting the normalized data to a linear mode using the lm() R function. (DOCX 13 kb)
ESM 7
Table S7. Validation of microarray data by qRT-PCR. (XLSX 15 kb)
ESM 8
Tables S8-S15. GO annotation of differentially regulated genes in Clusters UP and DOWN regulated. (XLSX 341 kb)
ESM 9
Tables S16-S25. Annotation of differentially regulated genes in each functional group sorted into the four ripening Up-regulated clusters. Transcriptomic analyses of genes whose expression changes during fruit ripening, in fruits treated with NDGA, in de-achened fruits and specificity in receptacle. NDGA and minus achenes indicate the expression obtained for the genes studied in green–white fruits injected with NDGA 100 μm and green2 fruits whose achenes were removed, respectively. Intensity (a.u.i.) indicates the total signal intensity for each feature on the microarray platform. Gene ID as reported in Fragaria vesca Genome Database (http://www.strawberrygenome.org/). (XLSX 1135 kb)
ESM 10
Tables S26-S29. Annotation of differentially down regulated genes sorted into functional groups. Transcriptomic analyses of genes whose expression changes during fruit ripening, in fruits treated with NDGA, in de-achened fruits and specificity in receptacle. NDGA and minus achenes indicate the expression obtained for the genes studied in green–white fruits injected with NDGA 100 μm and green2 fruits whose achenes were removed, respectively. Intensity (a.u.i.) indicates the total signal intensity for each feature on the microarray platform. Gene ID as reported in Fragaria vesca Genome Database (http://www.strawberrygenome.org/) (XLSX 123 kb)
ESM 11
Fig. S1. Distribution of the differentially expressed genes according their gene onthologies functional terms (GO terms) for categories Biological Process, Molecular Function and Cellular Component. (PDF 796 kb)
Rights and permissions
About this article
Cite this article
Medina-Puche, L., Blanco-Portales, R., Molina-Hidalgo, F.J. et al. Extensive transcriptomic studies on the roles played by abscisic acid and auxins in the development and ripening of strawberry fruits. Funct Integr Genomics 16, 671–692 (2016). https://doi.org/10.1007/s10142-016-0510-3
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10142-016-0510-3