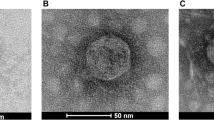
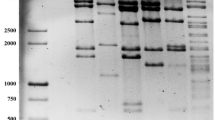
Abstract
Pseudomonas spp., such as P. fluorescens group, P. fragi, and P. putida, are the major psychrophilic spoilage bacteria in the food industry. Bacteriophages (phages) are a promising tool for controlling food-spoilage and food-poisoning bacteria; however, there are few reports on phages effective on food-spoilage bacteria such as Pseudomonas spp. In this study, 12 Pseudomonas phages were isolated from chicken and soil samples. Based on the host range and lytic activity at 30 °C and 4 °C and various combinations of phages, phages vB_PflP-PCS4 and vB_PflP-PCW2 were selected to prepare phage cocktails to control Pseudomonas spp. The phage cocktail consisting of vB_PflP-PCS4 and vB_PflP-PCW2 showed the strongest lytic activity and retarded regrowth of P. fluorescens and P. putida at 30 °C, 8 °C, and 4 °C at a multiplicity of infection of 100. Nucleotide sequence analysis of the genomic DNA indicated that vB_PflP-PCS4 and vB_PflP-PCW2 phages were lytic phages of the Podoviridae family and lacked tRNA, toxin, or virulence genes. A novel endolysin gene was found in the genomic DNA of phage vB_PflP-PCS4. The results of this study suggest that the phage cocktail consisting of vB_PflP-PCS4 and vB_PflP-PCW2 is a promising tool for the biocontrol of psychrophilic food-spoilage pseudomonads during cold storage and distribution.

Source of isolations was PCF1 from chicken fallopian tube, PCG1 from chicken gizzard; PCL1 from chicken liver; PCS1-7 from chicken skin, PCW2 from chicken wings; and PSP1 from soil. *Isolated from lettuce leaf






Similar content being viewed by others
Data availability
The authors confirm that the data supporting the findings of this study are available in the article and its supplementary information.
References
Bunker ST, Bates TC, Oliver JD (2004) Effects of temperature on detecting plasmid or chromosomally encoded gfp-and lux-labeled Pseudomonas fluorescens in soil. Environ Biosaf Res 3:83–90. https://doi.org/10.1051/ebr:2004008
Ceyssens PJ, Lavigne R (2010) Bacteriophages of Pseudomonas. Future Microbiol 5:1041–1055. https://doi.org/10.2217/fmb.10.66
Chan PP, Lin BY, Mak AJ, Lowe TM (2021) TRNAscan-SE 2.0: improved detection and functional classification of transfer RNA genes. Nucleic Acids Res 49:9077–9096. https://doi.org/10.1093/nar/gkab688
Clausen PTLC, Aarestrup FM, Lund O (2018) Rapid and precise alignment of raw reads against redundant databases with KMA. BMC Bioinforma 19:1–9. https://doi.org/10.1186/s12859-018-2336-6
Cornelissen A, Ceyssens PJ, Krylov VN, Noben JP, Volckaert G, Lavigne R (2012) Identification of EPS-degrading activity within the tail spikes of the novel Pseudomonas putida phage AF. Virology 434:251–256. https://doi.org/10.1016/j.virol.2012.09.030
Endersen L, Coffey A (2020) The use of bacteriophages for food safety. Curr Opin Food Sci 36:1–8. https://doi.org/10.1016/j.cofs.2020.10.006
Fanelli F, Caputo L, Quintieri L (2021) Phenotypic and genomic characterization of Pseudomonas putida ITEM 17297 spoiler of fresh vegetables: focus on biofilm and antibiotic resistance interaction. Curr Res Food Sci 4:74–82. https://doi.org/10.1016/j.crfs.2021.02.001
Flores O, Retamales J, Núñez M, León M, Salinas P, Besoain X, Yañez C, Bastías R (2020) Characterization of bacteriophages against Pseudomonas syringae pv. Actinidiae with potential use as natural antimicrobials in kiwifruit plants. Microorganisms 8(7):974. https://doi.org/10.3390/microorganisms8070974
Hirano SS, Upper CD (2000) Bacteria in the leaf ecosystem with emphasis on Pseudomonas syringae —a pathogen, ice nucleus, and epiphyte. Microbiol Mol Biol Rev 64:624–653. https://doi.org/10.1128/mmbr.64.3.624-653.2000
Holt A, Cahill J, Ramsey J, Martin C, O’Leary C, Moreland R, Maddox LT, Galbadage T, Sharan R, Sule P, Cirillo JD, Young R (2021) Phage-encoded cationic antimicrobial peptide required for lysis. J Bacteriol 204(1):e00214-221. https://doi.org/10.1128/jb.00214-21
Joensen KG, Scheutz F, Lund O, Hasman H, Kaas RS, Nielsen EM, Aarestrup FM (2014) Real-time whole-genome sequencing for routine typing, surveillance, and outbreak detection of verotoxigenic Escherichia coli. J Clin Microbiol 52:1501–1510. https://doi.org/10.1128/JCM.03617-13
Knecht LE, Veljkovic M, Fieseler L (2020) Diversity and function of phage encoded depolymerases. Front Microbiol 10:1–16. https://doi.org/10.3389/fmicb.2019.02949
Kosugi S, Hirakawa H, Tabata S (2015) GMcloser: closing gaps in assemblies accurately with a likelihood-based selection of contig or long-read alignments. Bioinformatics 31:3733–3741. https://doi.org/10.1093/bioinformatics/btv465
Kropinski AM, Prangishvili D, Lavigne R (2009) Position paper: the creation of a rational scheme for the nomenclature of viruses of Bacteria and Archaea. Environ Microbiol 11:2775–2777. https://doi.org/10.1111/j.1462-2920.2009.01970.x
Lammens EM, Nikel PI, Lavigne R (2020) Exploring the synthetic biology potential of bacteriophages for engineering nonmodel bacteria. Nat Commun 11:5294. https://doi.org/10.1038/s41467-020-19124-x
Lane DJ (1991) 16S/23S rRNA sequencing. In: Stackebrandt E, Goodfellow M (eds) Nucleic acid techniques in bacterial systematic. John Wiley and Sons, New York, pp 115–175
Lister PD, Wolter DJ, Hanson ND (2009) Antibacterial-resistant Pseudomonas aeruginosa: clinical impact and complex regulation of chromosomally encoded resistance mechanisms. Clin Microbiol Rev 22:582–610. https://doi.org/10.1128/CMR.00040-09
Lowe TM, Chan PP (2016) tRNAscan-SE On-line: integrating search and context for analysis of transfer RNA genes. Nucleic Acids Res 44:W54–W57. https://doi.org/10.1093/nar/gkw413
Lu G, Luhr J, Stoecklein A, Warner P, Tapprich W (2017) Complete genome sequences of Pseudomonas fluorescens bacteriophages isolated from freshwater samples in Omaha, Nebraska. Genome Announc 5(12):e01501-e1516. https://doi.org/10.1128/genomeA.01501-16
Malberg Tetzschner AM, Johnson JR, Johnston BD, Lund O, Scheutz F (2020) In Silico Genotyping ofE scherichia coli Isolates for Extraintestinal Virulence Genes by Use of Whole-Genome Sequencing Data. J Clin Microbiol 58(10):e01269-20. https://doi.org/10.1128/JCM.01269-20
Masuda Y, Kawabata S, Uedoi T, Honjoh KI, Miyamoto (2021) Construction of leaderless-bacteriocin-producing bacteriophage targeting E. coli and neighboring gram-positive pathogens. Microbiol Spectr 9(1):e0014121. https://doi.org/10.1111/1758-2229.12150
Naknaen A, Samernate T, Wannasrichan W (2023) Combination of genetically diverse Pseudomonas phages enhances the cocktail efficiency against bacteria. Sci Rep 13:8921. https://doi.org/10.1038/s41598-023-36034-2
Nishimori E, Kita-Tsukamoto K, Wakabayashi H (2000) Pseudomonas plecoglossicida sp. nov., the causative agent of bacterial hemorrhagic ascites of ayu, Plecoglossus altivelis. Int J Syst Evol Microbiol 50:83–89. https://doi.org/10.1099/00207713-50-1-83
Niu YD, Liu H, Du H, Meng R, Sayed Mahmoud E, Wang G, McAllister TA, Stanford K (2021) Efficacy of individual bacteriophages does not predict efficacy of bacteriophage cocktails for control of Escherichia coli O157. Front Microbiol 12:616712. https://doi.org/10.3389/fmicb.2021.616712
Papadopoulou OS, Iliopoulos V, Mallouchos A, Panagou EZ, Chorianopoulos N, Tassou CC, Nychas G-J (2020) Spoilage potential of Pseudomonas (P. fragi, P. putida) and LAB (Leuconostoc mesenteroides, Lactobacillus sakei) strains and their volatilome profile during storage of sterile pork meat using GC/MS and data analytics. Foods 9:633
Peng X, Nguyen A, Ghosh D (2018) Quantification of M13 and T7 bacteriophages by TaqMan and SYBR Green qPCR. J Virol Methods 252:100–107. https://doi.org/10.1016/j.jviromet.2017.11.012
Ramsay L, Macaulay M, Degli Ivanissevich S, MacLean K, Cardle L, Fuller J, Edwards KJ, Tuvesson S, Morgante M, Massari A, Maestri E, Marmiroli N, Sjakste T, Ganal M, Powell W, Waugh R (2000) Gapped BLAST and PSI-BLAST: a new generation of protein database search programs. Genetics 156:1997–2005. https://doi.org/10.1093/genetics/156.4.1997
Raposo A, Pérez E, dFaria CT, Ferrús MA, Carrascosa C (2017) Food Spoilage byP seudomonas spp.—An overview. In: Singh OV (ed) Foodborne Pathogens and Antibiotic Resistance. Wiley, Hoboken, pp 41–58. https://doi.org/10.1002/9781119139188.ch3
Sajben-Nagy E, Maróti G, Kredics L, Horváth B, Párducz Á, Vágvölgyi C, Manczinger L (2012) Isolation of new Pseudomonas tolaasii bacteriophages and genomic investigation of the lytic phage BF7. FEMS Microbiol Lett 332:162–169. https://doi.org/10.1111/j.1574-6968.2012.02592.x
Scales BS, Dickson RP, Lipuma JJ, Huffnagle GB (2014) Microbiology, genomics, and clinical significance of the Pseudomonas fluorescens species complex, an unappreciated colonizer of humans. Clin Microbiol Rev 27:927–948. https://doi.org/10.1128/CMR.00044-14
Schmelcher M, Donovan DM, Loessner MJ (2012) Bacteriophage endolysins as novel antimicrobials. Future Microbiol 7:1147–1171. https://doi.org/10.2217/fmb.12.97
Seemann T (2014) Prokka: rapid prokaryotic genome annotation. Bioinformatics 30:2068–2069. https://doi.org/10.1093/bioinformatics/btu153
Simões M, Simões LC, Vieira MJ (2010) A review of current and emergent biofilm control strategies. LWT - Food Sci Technol 43:573–583. https://doi.org/10.1016/J.LWT.2009.12.008
Son HM, Duc HM, Masuda Y, Honjoh K, Miyamoto T (2018) Application of bacteriophages in simultaneously controlling Escherichia coli O157:H7 and extended-spectrum beta-lactamase producing Escherichia coli. Appl Microbiol Biotechnol 102:10259–10271. https://doi.org/10.1007/s00253-018-9399-1
Sulakvelidze A, Alavidze, Morris J (2001) Bacteriophage therapy. Antimicrob Agents Chemother 45:649–659. https://doi.org/10.1128/AAC.45.3.649-659.2001
Summer EJ, Berry J, Tran TAT, Niu L, Struck DK, Young R (2007) Rz/Rz1 lysis gene equivalents in phages of gram-negative hosts. J Mol Biol 373:1098–1112. https://doi.org/10.1016/j.jmb.2007.08.045
Sutherland IW, Hughes KA, Skillman LC, Tait K (2004) The interaction of phage and biofilms. FEMS Microbiol Lett 232(1):1–6. https://doi.org/10.1016/S0378-1097(04)00041-2
Tanaka C, Yamada K, Takeuchi H, Inokuchi Y, Kashiwagi A, Toba T (2018) A lytic bacteriophage for controlling Pseudomonas lactis in raw cow’s milk. Appl Environ Microbiol 84(18):e00111-e118. https://doi.org/10.1128/AEM.00111-18
Thiel K (2004) Old dogma, new tricks - 21st century phage therapy. Nat Biotechnol 22:31–36. https://doi.org/10.1038/nbt0104-31
Xiang Y, Wang S, Li J, Wei Y, Zhang Q, Lin L, Ji X (2018) Isolation and characterization of two lytic cold-active bacteriophages infecting Pseudomonas fluorescens from the Napahai plateau wetland. Can J Microbiol 64:183–190. https://doi.org/10.1139/cjm-2017-0572
Zerbino DR, Birney E (2008) Velvet: algorithms for de novo short read assembly using de Bruijn graphs. Genome Res 18:821–829. https://doi.org/10.1101/gr.074492.107
Funding
This research was supported by JSPS KAKENHI (grant number JP23H02162).
Author information
Authors and Affiliations
Contributions
All authors contributed to the conception and design of this study. Takahisa Miyamoto contributed to the conceptualization, writing, review, editing, and supervision of the study. Material preparation, data collection, analysis, and the first draft of the manuscript were conducted by Daisuke Johno and Yu Zhang. Ken-ichi Honjoh and Yoshimitsu Masuda reviewed and edited the manuscript. Tahir Noor Mohammadi, Junxin Zhao, and Yunzhi Lin contributed to the methodology, investigation, and data curation. Aye Thida Maung, Chen Wang, Yuan Lu, Marwa Nabil Sayed Abdelaziz, Chen-Yu Lin, Mohamed El-Telbany, Su Zar Thi Lwin, and Catherine Hofilena Damaso performed visualization and investigation. All the authors have read and approved the final version of the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Johno, D., Zhang, Y., Mohammadi, T.N. et al. Characterization of selected phages for biocontrol of food-spoilage pseudomonads. Int Microbiol (2024). https://doi.org/10.1007/s10123-023-00479-2
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s10123-023-00479-2