Abstract
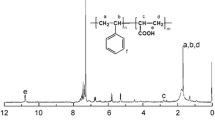
In our present work, a novel controlled radical polymerization system is developed based on xanthene-9-thione (XT). It was found that the radical polymerization of styrene (St) became controlled in the presence of a small amount of XT. At the early stage of the polymerization, the polymerization rate was relatively low and the as-formed polystyrene (PS) had low number-average molecular weight (Mn) and narrow polydispersity (Ð). After XT was consumed, the polymerization rate increased dramatically and the Mn of PS increased gradually with polymerization proceeding. When the polymerization of St was carried out with a proper molar ratio of initiator to XT and at an appropriate temperature, shortened slow polymerization stage and good control over Mn could be achieved. To further improve the regulating ability of XT, a series of substituent groups (-CF3, -CH(CH3)2, -N(CH3)2) were introduced onto the xanthene ring of XT, and the effects of these derivatives on the polymerization of St were investigated in detail. UV-Vis spectroscopy was carried out to monitor the concentration of XT during the polymerization and the chemical structure of the as-formed PS was fully characterized by 1HNMR and ESI-MS analysis. A possible mechanism involving the formation and evolution of the cross-termination products was proposed to interpret the observed polymerization behavior.
Similar content being viewed by others
References
Mishra, A. K.; Choi, C.; Maiti, S.; Seo, Y.; Lee, K. S.; Kim, E.; Kim, J. K. Sequential synthesis of well-defined poly(vinyl acetate)-block-polystyrene and poly(vinyl alcohol)-blockpolystyrene copolymers using difunctional chloroamidexanthate iniferter. Polymer 2018, 139, 68–75.
Kutcherlapati, S. R.; Koyilapu, R.; Jana, T. Poly(N-vinyl imidazole) grafted silica nanofillers: Synthesis by RAFT polymerization and nanocomposites with polybenzimidazole. J. Polym. Sci., Part A: Polym. Chem. 2018, 56(4), 365–375.
Díaz-Silvestre, S.; Saldívar-Guerra E.; Rivera-Vallejo, C.; Thomas, C. S.; Cabello-Romero, J.; Guerrero-Santos, R.; Jiménez-Regalado, E. Synthesis of associative block copolymers electrolytes via RAFT polymerization. Polym. Bull. 2018, 75(3), 891–907.
Semsarzadeh, M. A.; Sabzevari, A. Highly effective organometallic-mediated radical polymerization of vinyl acetate using alumina-supported Co(acac)2 catalyst: A case study of adsorption and polymerization. J. Appl. Polym. Sci. 2018, 135(13), 46057.
Bensabeh, N.; Ronda, J. C.; Galià, M.; Cádiz, V.; Lligadas, G.; Percec, V. SET-LRP of the hydrophobic biobased menthyl acrylate. Biomacromolecules 2018, 19(4), 1256–1268.
Matyjaszewski, K.; Tsarevsky, N. V. Macromolecular engineering by atom transfer radical polymerization. J. Am. Chem. Soc. 2014, 136(18), 6513.
Fu, X.; Yuan, Y.; Chen, X.; Xiao, Y.; Wang, J.; Zhou, C.; Lei, J. Use of short isobornyl methacrylate building blocks to improve the heat and oil resistance of thermoplastic elastomers via RAFT emulsion polymerization. J. Appl. Polym. Sci. 2017, 134(40), 45379.
Hawker, C. J.; Bosman, A. W.; Harth, E. New polymer synthesis by nitroxide mediated living radical polymerizations. Chem. Rev. 2001, 101(12), 3661.
Matyjaszewski, K.; Xia, J. Atom transfer radical polymerization. Chem. Rev. 2001, 101(9), 866–868.
Hill, M. R.; Carmean, R. N.; Sumerlin, B. S. Expanding the scope of raft polymerization: recent advances and new horizons. Macromolecules 2015, 48(16), 5459–5469.
Li, Q. L.; Li, L.; Wang, H. S.; Wang, R.; Wang, W.; Jiang, Y. J.; Tian, Q.; Liu, J. P. The doubly thermo-responsive triblock copolymer nanoparticles prepared through seeded RAFT polymerization. Chinese J. Polym. Sci. 2017, 35(1), 66–77.
Goto A.; Sato K.; Tsujii, Y.; Fukuda, T.; Moad G.; Rizzardo, E.; Thang, S. H. Mechanism and kinetics of RAFT-based living radical polymerizations of styrene and methyl methacrylate. Macromolecules 2001, 34(3), 402–408.
Poller, L.; Thomson, J. M. Determining the effect of side reactions on product distributions in RAFT polymerization by MALDI-TOF MS.Polym. Chem. 2015, 6(30), 5437–5450.
Ranieri, K.; Delaittre, G.; Barner-kowollik, C.; Thomas, J. Direct access to dithiobenzoate RAFT agent fragmentation rate coefficients by ESR spin-trapping. Macromol. Rapid Commun. 2014, 35(23), 2023.
Mayadunne, R. T. A.; Rizzardo, E.; Chiefari, J.; Chong, Y. K.; Moad, G.; Thang, S. H. Living radical polymerization with reversible addition-fragmentation chain transfer (RAFT polymerization) using dithiocarbamates as chain transfer agents. Macromolecules 1999, 32, 6977–6980.
Chiefari, J.; Mayadunne, R. T. A.; Moad, C. L.; Moad, G.; Rizzardo, E.; Postma, A.; Skidmore, M. A.; Thang, S. H. Thiocarbonylthio compounds (SC(Z)S-R) in free radical polymerization with reversible addition-fragmentation chain transfer (RAFT polymerization). effect of the activating group Z. Macromolecules 2003, 36(7), 2273–2283.
Moad, G.; Chiefari J, Mayadunne, R. T. A.; Moad, C. L.; Postma, A.; Rizzardo, E.; Thang, S. H. Initiating free radical polymerization. Macromol. Symp. 2002, 182, 65–80.
Moad, G.; Chiefari, J.; Chong, Y. K.; Krstina, J.; Mayadunne, R. T. A.; Postma, A.; Rizzardo, E.; Thang S. H. Living free radical polymerization with reversible addition-fragmentation chain transfer (the life of RAFT). Polym. Int. 2000, 49(9), 993–1001.
Barner-Kowollik, C.; Quinn, J. F.; Morsley, D. R.; Davis, T. P. Modeling the reversible addition-fragmentation chain transfer process in cumyl dithiobenzoate-mediated styrene homopolymerizations: Assessing rate coefficients for the addition–fragmentation equilibrium. J. Polym. Sci., Part A: Polym. Chem. 2001, 39(9), 1353–1365.
Monteiro, M. J.; Brouwer, H. D. Intermediate radical termination as the mechanism for retardation in reversible addition-fragmentation chain transfer polymerisation. Macromolecules 2001, 34(3), 349–352.
Feldermann, A.; Coote, M. L.; Stenzel, M. H.; Davis, T. P.; Barner-Kowollik, C. Consistent experimental and theoretical evidence for long-lived intermediate radicals in living free radical polymerization. J. Am. Chem. Soc. 2004, 126(48), 15915–15923.
Toy, A. A.; Chaffey-Millar, H.; Davis, T. P.; Stenzel, M. H.; Izgorodina, E. I.; Coote, M. L.; Barner-Kowollik, C. Thioketone spin traps as mediating agents for free radical polymerization processes. Chem. Commun. 2006, 8(8), 835–837.
Junkers, T.; Stenzel, M. H.; Davis, T. P.; Barner-Kowollik, C. Thioketone-mediated polymerization of butyl acrylate: controlling free-radical polymerization via a dormant radical species. Macromol. Rapid Commun. 2010, 28(6), 746–753.
Zheng, X.; Yue, M.; Yang, P.; Li, Q.; Yang, W. Cycloketyl radical mediated living polymerization. Polym. Chem. 2012, 3(8), 1982–1986.
Huang, X.; Wang, L.; Yang, W. Preparation of core-shell particles by surface-initiated cycloketyl radical mediated living polymerization. Polym. Chem. 2015, 6(37), 6664–6670.
Yao, C.; Wang, L.; Yang, W. Cycloketyl radical mediated suspension polymerization of styrene. RSC Adv. 2016, 6(74), 69743.
Wertz, S.; Leifert, D.; Studer, A. Cross dehydrogenative coupling via base-promoted homolytic aromatic substitution (BHAS): synthesis of fluorenones and xanthones. Org. Lett. 2013, 15(4), 928–931.
Hadjipavlou, C.; Kostakis, I. K.; Pouli, N.; Marakos, P.; Pratsinis, H.; Kletsas, D. Synthesis and antiproliferative activity of substituted benzopyranoisoindoles: a new class of cytotoxic compounds. Bioorg. Med. Chem. Lett. 2006, 16(18), 4822–4825.
Lakouraj, M. M.; Mohseni, S. M. Synthesis, characterization, and biological activities of organosoluble and thermally stable xanthone-based polyamides. J. Mater. Sci. 2015, 26(3), 234–244.
Nakatake, D.; Yokote, Y.; Matsushima, Y.; Yazaki, R.; Ohshima, T. A highly stable but highly reactive zinc catalyst for transesterification supported by a bis(imidazole) ligand. Green Chem. 2016, 18(6), 1524–1530.
Günzler, F.; Junkers, T.; Barner-Kowollik, C. Studying the mechanism of thioketone-mediated polymerization via electrospray ionization mass spectrometry. J. Polym. Sci., Part A: Polym. Chem. 2010, 47(7), 1864–1876.
Rodríguez-Sanchez, I.; Glossman-Mitnik, D.; Zaragoza-Contreras, E. A. Theoretical evaluation of the order of reactivity of transfer agents utilized in RAFT polymerization: group Z. J. Mol. Model. 2010, 16(1), 95–105.
Beaudoin, E.; Bertin, D.; Gigmes, D.; Marque, S. R. A.; Siri, D.; Tordo, P. Alkoxyamine C-ON bond homolysis: stereoelectronic effects. Eur. J. Org. Chem. 2006, 7, 1755–1768.
Zubenko, D.; Tsentalovich, Y.; Lebedeva, N.; Kirilyuk, I.; Roshchupkina, G.; Zhurko, I.; Reznikov, V.; Marque, S. R. A.; Bagryanskaya, E. Laser flash photolysis and CIDNP studies of steric effects on coupling rate constants of imidazolidine nitroxide with carbon-centered radicals, methyl isobutyrate-2-yl and tert-butyl propionate-2-yl. J. Org. Chem 2006, 71(16), 6044–6052.
Marchand, J.; Autissier, L.; Guillaneuf, Y.; Couturier, J. L.; Gigmes, D.; Bertin, D. SG1 nitroxide analogues: a comparative study. Aust. J. Chem. 2010, 63, 1237–1244.
Nicolasa, J.; Guillaneuf, Y.; Lefay, C.; Bertin, D.; Gigmes, D.; Charleux, B. Nitroxide-mediated polymerization. Prog. Polym. Sci. 2013, 38, 63–235.
Acknowledgments
This work was financially supported by the National Natural Science Foundation of China (Nos. 21404004, and 21474006) and the Innovation and Promotion Project of Beijing University of Chemical Technology.
Author information
Authors and Affiliations
Corresponding authors
Electronic supplementary material
Rights and permissions
About this article
Cite this article
Yu, HY., Wang, J., Shao, JW. et al. Controlled Radical Polymerization of Styrene Mediated by Xanthene-9-thione and Its Derivatives. Chin J Polym Sci 36, 1303–1311 (2018). https://doi.org/10.1007/s10118-018-2153-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10118-018-2153-4