Abstract
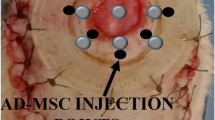
Herein, we report the influence of administering different protocols of preconditioned diabetic adipose-derived mesenchymal stem cells (ADSs) with photobiomodulation in vitro, and photobiomodulation in vivo on the number of mast cells (MCs), their degranulation, and wound strength in the maturation step of a Methicillin-resistant Staphylococcus aureus (MRSA)-infectious wound model in rats with type one diabetes. An MRSA-infectious wound model was generated on diabetic animals, and they were arbitrarily assigned into five groups (G). G1 were control rats. In G2, diabetic ADS were engrafted into the wounds. In G3, diabetic ADS were engrafted into the wound, and the wound was exposed to photobiomodulation (890 nm, 890 ± 10 nm, 80 Hz, 0.2 J/cm2) in vivo. In G4, preconditioned diabetic ADS with photobiomodulation (630 and 810 nm; each 3 times with 1.2 J/cm2) in vitro were engrafted into the wound. In G5, preconditioned diabetic ADS with photobiomodulation were engrafted into the wound, and the wound was exposed to photobiomodulation in vivo. The results showed that, the maximum force in all treatment groups was remarkably greater compared to the control group (all, p = 0.000). Maximum force in G4 and G5 were superior than that other treated groups (both p = 0.000). Moreover, G3, G4, and G5 showed remarkable decreases in completely released MC granules and total numbers of MC compared to G1 and G2 (all, p = 0.000). We concluded that diabetic rats in group 5 showed significantly better results in terms of accelerated wound healing and MC count of an ischemic infected delayed healing wound model.





Similar content being viewed by others
References
Jneid J, Lavigne J, La Scola B, Cassir N (2017) The diabetic foot microbiota: a review. Hum Microbiome J 5:1–6
Liu C, Ponsero AJ, Armstrong DG, Lipsky BA, Hurwitz BL (2020) The dynamic wound microbiome. BMC Med 18:1–12
Baltzis D, Eleftheriadou I, Veves A (2014) Pathogenesis and treatment of impaired wound healing in diabetes mellitus: new insights. Adv Ther 31:817–836
Komi DEA, Khomtchouk K, Santa Maria PL (2020) A review of the contribution of mast cells in wound healing: involved molecular and cellular mechanisms. Clin Rev Allergy Immunol 58(3):298–312
Johnzon C-F, Rönnberg E, Pejler G (2016) The role of mast cells in bacterial infection. Am J Pathol 186:4–14
Tellechea A, Leal EC, Kafanas A, Auster ME, Kuchibhotla S, Ostrovsky Y, Tecilazich F, Baltzis D, Zheng Y, Carvalho E, Zabolotny JM, Weng Z, Petra A, Patel A, Panagiotidou S, Pradhan-Nabzdyk L, Theoharides TC, Veves A (2016) Mast cells regulate wound healing in diabetes. Diabetes 65:2006–2019
Dong J, Chen L, Zhang Y, Jayaswal N, Mezghani I, Zhang W, Veves A (2020) Mast cells in diabetes and diabetic wound healing. Adv Ther 37:4519–4537
Kosaric N, Kiwanuka H, Gurtner GC (2019) Stem cell therapies for wound healing. Expert Opin Biol Ther 19:575–585
Patel S, Srivastava S, Singh MR, Singh D (2019) Mechanistic insight into diabetic wounds: Pathogenesis, molecular targets and treatment strategies to pace wound healing, Biomedicine & pharmacotherapy. Biomed Pharmacother 112:108615
Ahmadi H, Amini A, FadaeiFathabady F, Mostafavinia A, Zare F, Ebrahimpour-Malekshah R, Ghalibaf MN, Abrisham M, Rezaei F, Albright R, Ghoreishi SK, Chien S, Bayat M (2020) Transplantation of photobiomodulation-preconditioned diabetic stem cells accelerates ischemic wound healing in diabetic rats. Stem cell Res Ther 11(1):494
Ebrahimpour-Malekshah R, Amini A, Zare F, Mostafavinia A, Davoody S, Deravi N, Rahmanian M, Hashemi SM, Habibi M, Ghoreishi SK, Chien S, Shafikhani S, Ahmadi H, Bayat S, Bayat M (2020) Combined therapy of photobiomodulation and adipose-derived stem cells synergistically improve healing in an ischemic, infected and delayed healing wound model in rats with type 1 diabetes mellitus. BMJ Open Diabetes Res Care 8(1):e001033. https://doi.org/10.1136/bmjdrc-2019-001033
Moradi A, Zare F, Mostafavinia A, Safaju S, Shahbazi A, Habibi M, Abdollahifar MA, Hashemi SM, Amini A, Ghoreishi SK, Chien S, Hamblin MR, Kouhkheil R, Bayat M (2020) Photobiomodulation plus adipose-derived stem cells improve healing of ischemic infected wounds in type 2 diabetic rats. Sci Rep 10:1206
Şener LT, Albeniz I (2015) Challenge of mesenchymal stem cells against diabetic foot ulcer. Curr Stem Cell Res Ther 10:530–534
Tsuji W, Rubin JP, Marra KG (2014) Adipose-derived stem cells: implications in tissue regeneration. World J Stem Cells 6:312
Álvaro-Afonso FJ, Sanz-Corbalán I, Lázaro-Martínez JL, Kakagia D, Papanas N (2020) Adipose-derived mesenchymal stem cells in the treatment of diabetic foot ulcers: a review of preclinical and clinical studies. Angiology 71:853–863
Chehelcheraghi F, Bayat M, Chien S (2020) Effect of mesenchymal stem cells and chicken embryo extract on flap viability and mast cells in rat skin flaps. J Invest Surgery 33(2):123–133
Chehelcheraghi F, Chien S, Bayat M (2019) Mesenchymal stem cells improve survival in ischemic diabetic random skin flap via increased angiogenesis and VEGF expression. J Cell Biochem 120:17491–17499
Yadav A, Gupta A (2017) Noninvasive red and near-infrared wavelength-induced photobiomodulation: promoting impaired cutaneous wound healing. Photodermatol Photoimmunol Photomed 33:4–13
Pinto H, Goñi Oliver P, Sánchez-VizcaínoMengual E (2021) The effect of photobiomodulation on human mesenchymal cells: a literature review. Aesthetic Plast Surg 45(4):1826–1842
Fathabadie FF, Bayat M, Amini A, Bayat M, Rezaie F (2013) Effects of pulsed infra-red low level-laser irradiation on mast cells number and degranulation in open skin wound healing of healthy and streptozotocin-induced diabetic rats. J Cosmet Laser Ther : Off Pub Euro Soc Laser Dermatol 15:294–304
Bagheri M, Amini A, Abdollahifar MA, Ghoreishi SK, Piryaei A, Pouriran R, Chien S, Dadras S, Rezaei F, Bayat M (2018) Effects of photobiomodulation on degranulation and number of mast cells and wound strength in skin wound healing of streptozotocin-induced diabetic rats. Photomed Laser Surg 36:415–423
Kouhkheil R, Fridoni M, Abdollhifar MA, Amini A, Bayat S, Ghoreishi SK, Chien S, Kazemi M, Bayat M (2019) Impact of photobiomodulation and condition medium on mast cell counts, degranulation, and wound strength in infected skin wound healing of diabetic rats. Photobiomodul Photomed Laser Surg 37:706–714
Soleimani H, Amini A, Abdollahifar MA, Norouzian M, Kouhkheil R, Mostafavinia A, Ghoreishi SK, Bayat S, Chien S, Bayat M (2021) Combined effects of photobiomodulation and curcumin on mast cells and wound strength in wound healing of streptozotocin-induced diabetes in rats. Lasers Med Sci 36:375–386
Amini A, Pouriran R, Abdollahifar M-A, Abbaszadeh HA, Ghoreishi SK, Chien S, Bayat M (2018) Stereological and molecular studies on the combined effects of photobiomodulation and human bone marrow mesenchymal stem cell conditioned medium on wound healing in diabetic rats. J Photochem Photobiol, B 182:42–51
Babaei S, Bayat M, Nouruzian M, Bayat M (2013) Pentoxifylline improves cutaneous wound healing in streptozotocin-induced diabetic rats. Eur J Pharmacol 700:165–172
Falanga V (2005) Wound healing and its impairment in the diabetic foot. The Lancet 366:1736–1743
Xiao J, Li J, Cai L, Chakrabarti S, Li X (2014) Cytokines and diabetes research. J Diabetes Res 2014:920613. https://doi.org/10.1155/2014/920613. Epub 2014 Jan 16
Rapala K, Laato M, Niinikoski J, Kujari H, Söder O, Mauviel A, Pujol J-P (1991) Tumor necrosis factor alpha inhibits wound healing in the rat. Eur Surg Res 23:261–268
Tellechea A, Bai S, Dangwal S, Theocharidis G, Nagai M, Koerner S, Cheong JE, Bhasin S, Shih TY, Zheng Y, Zhao W, Zhang C, Li X, Kounas K, Panagiotidou S, Theoharides T, Mooney D, Bhasin M, Sun L, Veves A (2020) Topical application of a mast cell stabilizer improves impaired diabetic wound healing. J Invest Dermatol 140(4):901-911 e911
Ding X, Rath P, Angelo R, Stringfellow T, Flanders E, Dinh S, Gomez-Orellana I, Robinson JR (2004) Oral absorption enhancement of cromolyn sodium through noncovalent complexation. Pharm Res 21:2196–2206
Cairns H, Fitzmaurice C, Hunter D, Johnson P, King J, Lee T, Lord G, Minshull R, Cox J (1972) Synthesis and structure-activity relations of disodium cromoglycate and some related compounds. J Med Chem 15:583–589
Edwards AM, Bibawy D, Matthews S, Tongue N, Arshad SH, LødrupCarlsen K, Øymar K, Pollock I, Clifford R, Thomas A (2015) Long-term use of a 4% sodium cromoglicate cutaneous emulsion in the treatment of moderate to severe atopic dermatitis in children. J Dermatolog Treat 26(6):541–547
Nazari M, Ni NC, Lüdke A, Li SH, Guo J, Weisel RD, Li RK (2016) Mast cells promote proliferation and migration and inhibit differentiation of mesenchymal stem cells through PDGF. J Mol Cell Cardiol 94:32–42
Yun IS, Jeon YR, Lee WJ, Lee JW, Rah DK, Tark KC, Lew DH et al (2012) Effect of human adipose derived stem cells on scar formation and remodeling in a pig model: a pilot study. Dermatolo Surg 38(10):1678–1688
Houreld N (2019) Healing effects of photobiomodulation on diabetic wounds. Appl Sci 9:5114
Gruber BL, Marchese MJ, Kew R (1995) Angiogenic factors stimulate mast-cell migration. Blood 86(7):2488–2493
Hiromatsu Y, Toda S (2003) Mast cells and angiogenesis. Microsc Res Tech 60:64–69
Theoharides TC, Alysandratos KD, Angelidou A, Delivanis DA, Sismanopoulos N, Zhang B, Asadi S, Vasiadi M, Weng Z, Miniati A (2012) Mast cells and inflammation. Biochim Biophys Acta 1822(1):21–33
Bayat M, Chien S, Chehelcheraghi F (2021) Co-localization of Flt1 and tryptase of mast cells in skin wound of rats with type I diabetes: Initial studies. Acta Histochem 123(2):151680
do Valle IB, Prazeres PHDM, Mesquita RA, Silva TA, de Castro Oliveira HM, Castro PR, Freitas IDP, Oliveira SR, Gomes NA, de Oliveira RF (2020) Photobiomodulation drives pericyte mobilization towards skin regeneration. Sci Rep 10(1):15
Pereira MCM, de Pinho CB, Medrado ARP, de Araújo Andrade Z, de Almeida Reis SR (2010) Influence of 670 nm low-level laser therapy on mast cells and vascular response of cutaneous injuries. J Photochem Photobiol B 98(3):188–192
Silveira L, Prates R, Novelli M, Marigo H, Garrocho A, Amorim J, Sousa G, Pinotti M, Ribeiro M (2008) Investigation of mast cells in human gingiva following low-intensity laser irradiation. Photomed Laser Surg 26:315–321
Sawasaki I, Geraldo-Martins VR, Ribeiro MS, Marques MM (2009) Effect of low-intensity laser therapy on mast cell degranulation in human oral mucosa. Lasers Med Sci 24:113–116
Khoshvaghti A, Zibamanzarmofrad M, Bayat M (2011) Effect of low-level treatment with an 80-Hz pulsed infrared diode laser on mast-cell numbers and degranulation in a rat model of third-degree burn. Photomed Laser Surg 29:597–604
Funding
The present study was financially supported by the Research Department of the School of Medicine at Shahid Beheshti University of Medical Sciences, Tehran, Iran (Grant no. 20266). This article was extracted from the thesis written by Mr. Houssein Ahmadi (Registration No.: IR. SBMU. MSP.REC.1399.105).
Author information
Authors and Affiliations
Contributions
Mohammad Bayat and Abdollah Amini researched the data and wrote the manuscript. Houssein Ahmadi, Robabeh Asadi, and Roohollah Ebrahimpour-Malekshah performed the methods; Rouhallah Gazor, Sasha H. Shafikhani, Fatemehalsadat Rezaei, Latif Gachkar, and Sufan Chien presented their comments; Atarodalsadat Mostafavinia and Seyed Kamran Ghoreishi performed statistical tests.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Ahmadi, H., Bayat, M., Amini, A. et al. Impact of preconditioned diabetic stem cells and photobiomodulation on quantity and degranulation of mast cells in a delayed healing wound simulation in type one diabetic rats. Lasers Med Sci 37, 1593–1604 (2022). https://doi.org/10.1007/s10103-021-03408-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10103-021-03408-9