Abstract
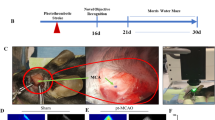
The integrity of the structural cerebral cortex is disrupted after stroke either at the macroscopic or microscopic levels. Therefore, many attempts have been gathered to circumvent stroke-associated problems in the brain tissue. The current study was aimed to design an animal model of photochemical stroke using rose bengal (RB) plus laser irradiation (L) after 10, 15, and 20 min (´) and evaluate its effect on the cerebral tissue using unbiased stereological quantitative methods and morphometric histological analysis. Photochemical stroke was induced by intraperitoneal injection of RB dye and further activation through the exposure of the right sensorimotor cortex with the green laser radiation (100 mW; 532 nm). Mice were randomly allocated into 4 groups (each in 15) as follows: control (10 μg/gbw RB), RB + 10’L, RB + 15’L, and RB + 20’L. Target irradiation site was adjusted to 2 mm lateral to the bregma. Vernier caliper morphometric evaluation, cresyl violet staining, and unbiased stereological assays including Cavalier’s principle and point counting techniques were used to monitor the pathological changes and lesion volume on days 1, 3, and 7 after the ischemia induction. Our data showed that the mean diameter of the lesion site and lesion infarct volume in the group RB + 20’L) was significantly increased relative to the other groups (P < 0.05). Notably, the lesion volume and diameter in the group RB + 15’L was larger compared with the group RB + 10’L and control mice (P < 0.05). Data showed an increased acute inflammatory response such as hyperemia and edema 3 days after ischemic induction while the intensity of acute changes and lesion volume were reduced and replaced with necrotic and chronic pathological changes including astrogliosis on day 7. It is concluded that the laser irradiation of RB-injected mice at a distinct time period could induce the magnificent degenerative effects on the cerebral cortex which is similar to the stroke condition.




Similar content being viewed by others
References
Prasad K et al (2014) Intravenous autologous bone marrow mononuclear stem cell therapy for ischemic stroke: a multicentric, randomized trial. Stroke 45(12):3618–3624
Ford G et al (2004) Stroke, dementia, and drug delivery. Br J Clin Pharmacol 57(1):15–26
Campbell BC et al (2015) Endovascular therapy for ischemic stroke with perfusion-imaging selection. N Engl J Med 372(11):1009–1018
Kolb B et al (2007) Growth factor-stimulated generation of new cortical tissue and functional recovery after stroke damage to the motor cortex of rats. J Cereb Blood Flow Metab 27(5):983–997
Sims NR, Muyderman H (2010) Mitochondria, oxidative metabolism and cell death in stroke. Biochimica et Biophysica Acta (BBA)-Molecular Basis of Disease 1802(1):80–91
Thrift AG et al (2001) Incidence of the major stroke subtypes: initial findings from the North East Melbourne stroke incidence study (NEMESIS). Stroke 32(8):1732–1738
Kalladka D, Muir KW (2014) Brain repair: cell therapy in stroke. Stem Cells Cloning 7:31
Davis SM, Donnan GA (2012) Secondary prevention after ischemic stroke or transient ischemic attack. N Engl J Med 366(20):1914–1922
Feigin VL et al (2009) Worldwide stroke incidence and early case fatality reported in 56 population-based studies: a systematic review. Lancet Neurol 8(4):355–369
Andrews EM et al (2008) Human adult bone marrow-derived somatic cell therapy results in functional recovery and axonal plasticity following stroke in the rat. Exp Neurol 211(2):588–592
Dimyan MA, Cohen LG (2011) Neuroplasticity in the context of motor rehabilitation after stroke. Nat Rev Neurol 7(2):76
Wen H et al (2008) Thigh muscle function in stroke patients revealed by velocity-encoded cine phase-contrast magnetic resonance imaging. Muscle Nerve 37(6):736–744
Lipton P (1999) Ischemic cell death in brain neurons. Physiol Rev 79(4):1431–1568
Whiteley WN et al (2014) Targeting recombinant tissue-type plasminogen activator in acute ischemic stroke based on risk of intracranial hemorrhage or poor functional outcome: an analysis of the third international stroke trial. Stroke 45(4):1000–1006
Ridker PM et al (1994) Prospective study of endogenous tissue plasminogen activator and risk of stroke. Lancet 343(8903):940–943
Zivin JA et al (1985) Tissue plasminogen activator reduces neurological damage after cerebral embolism. Science 230(4731):1289–1292
Rosenblum WI, El-Sabban F (1977) Platelet aggregation in the cerebral microcirculation: effect of aspirin and other agents. Circ Res 40(3):320–328
Labat-gest V, Tomasi S (2013, 76) Photothrombotic ischemia: a minimally invasive and reproducible photochemical cortical lesion model for mouse stroke studies. J Vis Exp:e50370
Bergeron M (2003) Inducing photochemical cortical lesions in rat brain. Curr Protoc Neurosci 23(1):9.16. 1–9.16. 18
Dietrich WD et al (1987) Photochemically induced cerebral infarction. I. Early microvascular alterations. Acta Neuropathol 72(4):315–325
Schroeter M, Jander S, Stoll G (2002) Non-invasive induction of focal cerebral ischemia in mice by photothrombosis of cortical microvessels: characterization of inflammatory responses. J Neurosci Methods 117(1):43–49
Bagheri HS et al (2018) Low-level laser irradiation at a high power intensity increased human endothelial cell exosome secretion via Wnt signaling. Lasers Med Sci 33(5):1131–1145
Watson BD et al (1985) Induction of reproducible brain infarction by photochemically initiated thrombosis. Ann Neurol 17(5):497–504
Kitagawa K et al (1998) Cerebral ischemia after bilateral carotid artery occlusion and intraluminal suture occlusion in mice: evaluation of the patency of the posterior communicating artery. J Cereb Blood Flow Metab 18(5):570–579
Lee J-K et al (2007) Photochemically induced cerebral ischemia in a mouse model. Surg Neurol 67(6):620–625 discussion 625
Boquillon M, Boquillon J, Bralet J (1992) Photochemically induced, graded cerebral infarction in the mouse by laser irradiation evolution of brain edema. J Pharmacol Toxicol Methods 27(1):1–6
Choi Y et al (2016) Human mesenchymal stem cell transplantation promote neural differentiation and behavioral performance in the photothrombotic mouse model. Biotechnol J 11(11):1397–1404
West MJ (2012) Introduction to stereology. Cold Spring Harb Protoc 2012(8):pdb.top070623
Golub VM et al (2015) Neurostereology protocol for unbiased quantification of neuronal injury and neurodegeneration. Front Aging Neurosci 7:196
Gundersen HJ et al (1988) Some new, simple and efficient stereological methods and their use in pathological research and diagnosis. Apmis 96(5):379–394
Franklin K, Paxinos G (1997) The Mouse Brain in Stereotaxic Coordinates, 1st edn. Academic Press, New York
Silva VM et al (2005) The rat ear vein model for investigating in vivo thrombogenicity of ultrafine particles (UFP). Toxicol Sci 85(2):983–989
Esfandiary E et al (2014) Novel effects of Rosa damascena extract on memory and neurogenesis in a rat model of Alzheimer's disease. J Neurosci Res 92(4):517–530
Osinubi A, Noronha C, Okanlawon A (2005) Morphometric and stereological assessment of the effects of long-term administration of quinine on the morphology of rat testis. West Afr J Med 24(3):200–205
Meinel TR et al (2020) MRI characteristics in acute ischemic stroke patients with preceding direct oral anticoagulant therapy as compared to vitamin K antagonists. BMC Neurol 20(1):86
Mărgăritescu O et al (2009) Histopathological changes in acute ischemic stroke. Rom J Morphol Embryol 50(3):327–339
Xing C et al (2012) Pathophysiologic cascades in ischemic stroke. Int J Stroke 7(5):378–385
Lakhan SE, Kirchgessner A, Hofer M (2009) Inflammatory mechanisms in ischemic stroke: therapeutic approaches. J Transl Med 7(1):97
Paciaroni M, Caso V, Agnelli G (2009) The concept of ischemic penumbra in acute stroke and therapeutic opportunities. Eur Neurol 61(6):321–330
Alaverdashvili M, Paterson PG, Bradley MP (2015) Laser system refinements to reduce variability in infarct size in the rat photothrombotic stroke model. J Neurosci Methods 247:58–66
Hao JX et al (1994) Photochemically induced spinal cord ischaemia in rats: assessment of blood flow by laser Doppler flowmetry. Acta Physiol Scand 151(2):209–215
Yu W et al (2000) Photochemically-induced ischemic injury of the rat sciatic nerve: a light- and electron microscopic study. J Peripher Nerv Syst 5(4):209–217
Lansberg MG et al (2001) Evolution of cerebral infarct volume assessed by diffusion-weighted magnetic resonance imaging. Arch Neurol 58(4):613–617
Liu NW et al (2017) Evolutional characterization of photochemically induced stroke in rats: a multimodality imaging and molecular biological study. Transl Stroke Res 8(3):244–256
Funding
This study was supported by a grant from Tabriz University of Medical Sciences.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflicts of interest.
Ethical approval
This study was confirmed by the local ethics committee of Medical Sciences University of Tabriz (IR.TBZMED.VCR.REC.1397.420).
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Shahi, M., Abedelahi, A., Mohammadnejad, D. et al. Exact location of sensorimotor cortex injury after photochemical modulation; evidence of stroke based on stereological and morphometric studies in mice. Lasers Med Sci 36, 91–98 (2021). https://doi.org/10.1007/s10103-020-03017-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10103-020-03017-y