Abstract
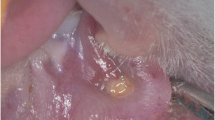
Cancer treatment with chemotherapy or radiotherapy is associated with some side effects including in the oral cavity. One of the more significant oral complications is oral mucositis (OM) which induces severe pain and limits fundamental life behaviors such as eating, drinking, and talking. Although advancements in cancer treatment improved the survival rate, severe OM and opportunistic infection affect treatment adversely. Therefore, the control of OM is important for oral health quality of life and prognosis. Low-level laser therapy (LLLT) and photodynamic therapy (PT) are noninvasive methods that reduce inflammation and pain during wound healing. The aim of this study is to evaluate immunohistochemical and histological examination of the OM region of the PT comparing LLLT. In this study, 24 Sprague-Dawley rats were divided into three groups as control, LLLT, and PT groups. All groups received 5-fluorouracil intraperitoneally and a linear trauma to the mouth pouch with a needle. After the formation of OM in the mouth, the control group had no treatment; the LLLT group was administered LLLT, and the PT group had LLLT after indocyanine green application. Then all groups were sacrificed, and histological analyses and protein level detection of basic fibroblast growth factor (bFGF), transforming growth factor (TGF-β), and platelet-derived growth factor (PDGF-BB) were evaluated in all groups. PT was determined to be more statistically significantly than LLLT with bFGF and PDGF-BB. However, regarding TGF-β, no statistically significant difference was observed between the groups. Within the limitations of this study, indocyanine green may accelerate the LLLT effect. However, further studies on this subject are required.





Similar content being viewed by others
References
Villa A, Sonis ST (2015) Mucositis: pathobiology and management. Curr Opin Oncol 27(3):159–164. https://doi.org/10.1097/CCO.0000000000000180
Gautam AP, Fernandes DJ, Vidyasagar MS, Maiya GA (2012) Low level helium neon laser therapy for chemoradiotherapy induced oral mucositis in oral cancer patients - a randomized controlled trial. Oral Oncol 48(9):893–897. https://doi.org/10.1016/j.oraloncology.2012.03.008
Naidu MU, Ramana GV, Rani PU, Mohan IK, Suman A, Roy P (2004) Chemotherapy-induced and/or radiation therapy-induced oral mucositis--complicating the treatment of cancer. Neoplasia 6(5):423–431. https://doi.org/10.1593/neo.04169
Mañas A, Palacios A, Contreras J, Sánchez-Magro I, Blanco P, Fernández-Pérez C (2009) Incidence of oral mucositis, its treatment and pain management in patients receiving cancer treatment at radiation oncology departments in Spanish hospitals (MUCODOL study). Clin Transl Oncol 11(10):669–676
Nicolatou-Galitis O, Sarri T, Bowen J, Di Palma M, Kouloulias VE, Niscola P, Riesenbeck D, Stokman M, Tissing W, Yeoh E (2013) Systematic review of anti-inflammatory agents for the management of oral mucositis in cancer patients. Support Care Cancer 21(11):3179–3189
Franca CM, Franca CM, Nunez SC, Prates RA, Noborikawa E, Faria MR, Ribeiro MS (2009) Low-intensity red laser on the prevention and treatment of induced-oral mucositis in hamsters. J Photochem Photobiol B 94(1):25–31. https://doi.org/10.1016/j.jphotobiol.2008.09.006
Cruz Ede P, Campos L, Pereira Fda S, Magliano GC, Benites BM, Arana-Chavez VE, Ballester RY, Simoes A (2015) Clinical, biochemical and histological study of the effect of antimicrobial photodynamic therapy on oral mucositis induced by 5-fluorouracil in hamsters. Photodiagn Photodyn Ther 12(2):298–309. https://doi.org/10.1016/j.pdpdt.2014.12.007
Bayer S, Kazancioglu HO, Acar AH, Demirtas N, Kandas NO (2017) Comparison of laser and ozone treatments on oral mucositis in an experimental model. Lasers Med Sci 32(3):673–677. https://doi.org/10.1007/s10103-017-2166-1
Migliorati C, Hewson I, Lalla RV, Antunes HS, Estilo CL, Hodgson B, Lopes NN, Schubert MM, Bowen J, Elad S, Mucositis Study Group of the Multinational Association of Supportive Care in Cancer/International Society of Oral O (2013) Systematic review of laser and other light therapy for the management of oral mucositis in cancer patients. Support Care Cancer 21(1):333–341. https://doi.org/10.1007/s00520-012-1605-6
de Freitas LF, Hamblin MR (2016) Proposed mechanisms of photobiomodulation or low-level light therapy. IEEE J Sel Top Quantum Electron 22 (3). doi:https://doi.org/10.1109/jstqe.2016.2561201
Wang Y, Huang YY, Wang Y, Lyu P, Hamblin MR (2017) Photobiomodulation of human adipose-derived stem cells using 810nm and 980nm lasers operates via different mechanisms of action. Biochim Biophys Acta Gen Subj 1861(2):441–449. https://doi.org/10.1016/j.bbagen.2016.10.008
Wang X, Tian F, Soni SS, Gonzalez-Lima F, Liu H (2016) Interplay between up-regulation of cytochrome-c-oxidase and hemoglobin oxygenation induced by near-infrared laser. Sci Rep 6:30540. https://doi.org/10.1038/srep30540
Bjordal JM, Johnson MI, Iversen V, Aimbire F, Lopes-Martins RAB (2006) Low-level laser therapy in acute pain: a systematic review of possible mechanisms of action and clinical effects in randomized placebo-controlled trials. Photomed Laser Ther 24(2):158–168
Wang X, Reddy DD, Nalawade SS, Pal S, Gonzalez-Lima F, Liu H (2017) Impact of heat on metabolic and hemodynamic changes in transcranial infrared laser stimulation measured by broadband near-infrared spectroscopy. Neurophotonics 5(1):011004
Sonis ST (2011) Oral mucositis. Anti-Cancer Drugs 22(7):607–612. https://doi.org/10.1097/CAD.0b013e3283462086
Silva GB, Sacono NT, Othon-Leite AF, Mendonca EF, Arantes AM, Bariani C, Duarte LG, Abreu MH, Queiroz-Junior CM, Silva TA, Batista AC (2015) Effect of low-level laser therapy on inflammatory mediator release during chemotherapy-induced oral mucositis: a randomized preliminary study. Lasers Med Sci 30(1):117–126. https://doi.org/10.1007/s10103-014-1624-2
Srikanth K, Chandra RV, Reddy AA, Reddy BH, Reddy C, Naveen A (2015) Effect of a single session of antimicrobial photodynamic therapy using indocyanine green in the treatment of chronic periodontitis: a randomized controlled pilot trial. Quintessence Int 46(5):391–400. https://doi.org/10.3290/j.qi.a33532
Keshri GK, Gupta A, Yadav A, Sharma SK, Singh SB (2016) Photobiomodulation with pulsed and continuous wave near-infrared laser (810 nm, Al-Ga-as) augments dermal wound healing in immunosuppressed rats. PLoS One 11(11):e0166705. https://doi.org/10.1371/journal.pone.0166705
Akbari T, Pourhajibagher M, Hosseini F, Chiniforush N, Gholibegloo E, Khoobi M, Shahabi S, Bahador A (2017) The effect of indocyanine green loaded on a novel nano-graphene oxide for high performance of photodynamic therapy against enterococcus faecalis. Photodiagn Photodyn Ther. https://doi.org/10.1016/j.pdpdt.2017.08.017
Beltes C, Economides N, Sakkas H, Papadopoulou C, Lambrianidis T (2017) Evaluation of antimicrobial photodynamic therapy using indocyanine green and near-infrared diode laser against enterococcus faecalis in infected human root canals. Photomed Laser Surg 35(5):264–269. https://doi.org/10.1089/pho.2016.4100
Tsuda T, Kaibori M, Hishikawa H, Nakatake R, Okumura T, Ozeki E, Hara I, Morimoto Y, Yoshii K, Kon M (2017) Near-infrared fluorescence imaging and photodynamic therapy with indocyanine green lactosome has antineoplastic effects for hepatocellular carcinoma. PLoS One 12(8):e0183527. https://doi.org/10.1371/journal.pone.0183527
Sonis ST, Peterson RL, Edwards LJ, Lucey CA, Wang L, Mason L, Login G, Ymamkawa M, Moses G, Bouchard P, Hayes LL, Bedrosian C, Dorner AJ (2000) Defining mechanisms of action of interleukin-11 on the progression of radiation-induced oral mucositis in hamsters. Oral Oncol 36(4):373–381
Leitao RF, Ribeiro RA, Bellaguarda EA, Macedo FD, Silva LR, Oria RB, Vale ML, Cunha FQ, Brito GA (2007) Role of nitric oxide on pathogenesis of 5-fluorouracil induced experimental oral mucositis in hamster. Cancer Chemother Pharmacol 59(5):603–612. https://doi.org/10.1007/s00280-006-0301-y
Topaloglu N, Guney M, Yuksel S, Gulsoy M (2015) Antibacterial photodynamic therapy with 808-nm laser and indocyanine green on abrasion wound models. J Biomed Opt 20(2):28003. https://doi.org/10.1117/1.JBO.20.2.028003
Deyhimi P, Khademi H, Birang R, Akhoondzadeh M (2016) Histological evaluation of wound healing process after photodynamic therapy of rat oral mucosal ulcer. J Dent 17(1):43
Campos L, Cruz ÉP, Pereira FS, Arana-Chavez VE, Simões A (2016) Comparative study among three different phototherapy protocols to treat chemotherapy-induced oral mucositis in hamsters. J Biophotonics 9(11–12):1236–1245
Jin H-J, Lee S-H, Lee S-U, Lee H-S, Jin S-M, Kim D-H, Chae S-W, Sohn J-H (2013) Morphological and histological changes of rabbit vocal fold after steroid injection. Otolaryngol Head Neck Surg 149(2):277–283
de Sousa Farias S, Nemezio M, Corona S, Aires C, Borsatto M (2016) Effects of low-level laser therapy combined with toluidine blue on polysaccharides and biofilm of Streptococcus mutans. Lasers Med Sci 31(5):1011–1016
Souza SL, Andrade PF, Silva JS, Tristao FS, Rocha FA, Palioto DB, Grisi MF, Taba M Jr, Novaes AB Jr (2013) Effects of antimicrobial photodynamic therapy on transforming growth factor-beta1 levels in the gingival crevicular fluid. Photomed Laser Surg 31(2):65–71. https://doi.org/10.1089/pho.2012.3328
Scully C, Sonis S, Diz P (2006) Oral mucositis. Oral Dis 12(3):229–241
Gautam AP, Fernandes DJ, Vidyasagar MS, Maiya AG, Guddattu V (2015) Low level laser therapy against radiation induced oral mucositis in elderly head and neck cancer patients-a randomized placebo controlled trial. J Photochem Photobiol B 144:51–56. https://doi.org/10.1016/j.jphotobiol.2015.01.011
Keefe DM, Schubert MM, Elting LS, Sonis ST, Epstein JB, Raber-Durlacher JE, Migliorati CA, DB MG, Hutchins RD, Peterson DE, Mucositis Study Section of the Multinational Association of Supportive Care in C and the International Society for Oral O (2007) Updated clinical practice guidelines for the prevention and treatment of mucositis. Cancer 109(5):820–831. https://doi.org/10.1002/cncr.22484
Yoshino F, Yoshida A, Nakajima A, Wada-Takahashi S, Takahashi S-S, Lee MC-I (2013) Alteration of the redox state with reactive oxygen species for 5-fluorouracil-induced oral mucositis in hamsters. PLoS One 8(12):e82834
Pourzarandian A, Watanabe H, Ruwanpura SM, Aoki A, Ishikawa I (2005) Effect of low-level Er: YAG laser irradiation on cultured human gingival fibroblasts. J Periodontol 76(2):187–193
Bensadoun RJ, Nair RG (2012) Efficacy of low-level laser therapy (LLLT) in oral mucositis: what have we learned from randomized studies and meta-analyses? Photomed Laser Surg 30(4):191–192. https://doi.org/10.1089/pho.2012.9890
Kreisler M, Christoffers AB, Willershausen B, d'Hoedt B (2003) Effect of low-level GaAlAs laser irradiation on the proliferation rate of human periodontal ligament fibroblasts: an in vitro study. J Clin Periodontol 30(4):353–358
AlGhamdi KM, Kumar A, Moussa NA (2012) Low-level laser therapy: a useful technique for enhancing the proliferation of various cultured cells. Lasers Med Sci 27(1):237–249
Pereira AN, Eduardo Cde P, Matson E, Marques MM (2002) Effect of low-power laser irradiation on cell growth and procollagen synthesis of cultured fibroblasts. Lasers Surg Med 31(4):263–267. https://doi.org/10.1002/lsm.10107
Aras MH, Güngörmüş M (2010) Placebo-controlled randomized clinical trial of the effect two different low-level laser therapies (LLLT)—intraoral and extraoral—on trismus and facial swelling following surgical extraction of the lower third molar. Lasers Med Sci 25(5):641–645
Arduino PG, Cafaro A, Garrone M, Gambino A, Cabras M, Romagnoli E, Broccoletti R (2016) A randomized pilot study to assess the safety and the value of low-level laser therapy versus clonazepam in patients with burning mouth syndrome. Lasers Med Sci 31(4):811–816. https://doi.org/10.1007/s10103-016-1897-8
Dickers B, Lamard L, Peremans A, Geerts S, Lamy M, Limme M, Rompen E, De Moor R, Mahler P, Rocca J (2009) Temperature rise during photo-activated disinfection of root canals. Lasers Med Sci 24(1):81–85
de-Melo MA, Passos VF, Alves JJ, Barros EB, Santiago SL, Rodrigues LK (2011) The effect of diode laser irradiation on dentin as a preventive measure against dental erosion: an in vitro study. Lasers Med Sci 26(5):615–621
Mendoza-Garcia J, Sebastian A, Alonso-Rasgado T, Bayat A (2015) Optimization of an ex vivo wound healing model in the adult human skin: functional evaluation using photodynamic therapy. Wound Repair Regen 23(5):685–702. https://doi.org/10.1111/wrr.12325
Safavi SM, Kazemi B, Esmaeili M, Fallah A, Modarresi A, Mir M (2008) Effects of low-level He-Ne laser irradiation on the gene expression of IL-1beta, TNF-alpha, IFN-gamma, TGF-beta, bFGF, and PDGF in rat’s gingiva. Lasers Med Sci 23(3):331–335. https://doi.org/10.1007/s10103-007-0491-5
Acknowledgements
This study was funded by Bezmialem Vakif University Scientific Research Projects Unit.
Funding
This study was funded by Bezmialem Vakif University (grant number 9.2015/13).
Role of funding source was for purchasing of chemicals and animals.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Ethical approval
The experimental protocol was approved by the Institutional Review Board and Animal Use Committee of the Bezmialem Vakif University (protocol = 2017/11).
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Alinca, S.B., Saglam, E., Kandas, N.O. et al. Comparison of the efficacy of low-level laser therapy and photodynamic therapy on oral mucositis in rats. Lasers Med Sci 34, 1483–1491 (2019). https://doi.org/10.1007/s10103-019-02757-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10103-019-02757-w