Abstract
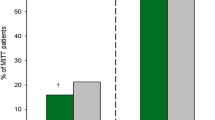
Meropenem-vaborbactam is a carbapenem and β-lactamase inhibitor combination that is newly indicated for the treatment of complicated urinary tract infections (cUTI), including adult pyelonephritis. Vaborbactam was developed due to emergence of carbapenem-resistant strains of Enterobacteriaceae. In a phase I trial, patients that received meropenem-vaborbactam 2–2 g intravenously over 3 h every 8 h, Cmax was 58.2 ± 10.8 μg/mL for meropenem and 59.0 ± 8.4 μg/mL for vaborbactam. AUC0–8 was 186 ± 33.6 μg • h/mL for meropenem and 204 ± 34.6 μg • h/mL for vaborbactam. Vss = 16.3 ± 2.6 L for meropenem and 17.6 ± 2.6 L for vaborbactam. Protein binding for vaborbactam averaged 33% in humans. Plasma clearance ranged from 10.42 ± 1.85 to 14.77 ± 2.84 L/h. One phase III trial evaluated efficacy for meropenem-vaborbactam 2–2 g intravenously every 8 h versus piperacillin-tazobactam 4–0.5 g intravenously every 8 h in complicated UTI. It found non-inferiority and statistical superiority for meropenem in overall success at the end of treatment primary end point. In another phase III trial evaluating efficacy in carbapenem-resistant Enterobacteriaceae (CRE) infections, meropenem-vaborbactam 2–2 g intravenously every 8 h was associated with decreased 28-day mortality and increased clinical cure compared with a best available therapy group.
Similar content being viewed by others
References
Bush K, Fisher JF (2011) Epidemiological expansion, structural studies, and clinical challenges of new β-lactamases from gram-negative bacteria. Annu Rev Microbiol 65:455–478. https://doi.org/10.1146/annurev-micro-090110-102911
Patel G, Bonomo RA (2013) “Stormy waters ahead”: global emergence of carbapenemases. Front Microbiol 4(48) https://doi.org/10.3389/fmicb.2013.00048
Lagacé-Wiens P, Walkty A, Karlowsky JA (2014) Ceftazidime-avibactam: an evidence-based review of its pharmacology and potential use in the treatment of gram-negative bacterial infections. Core Evid 9:13–25. https://doi.org/10.2147/CE.S40698
(2016) Carbapenem-resistant Enterobacteriaceae in Healthcare Settings. CDC. https://www.cdc.gov/hai/organisms/cre/index.html. Accessed 27 Nov 2017
Sabet M, Tarazi Z, Rubio-Aparicio D et al (2018) Activity of simulated human dosage regimens of meropenem and vaborbactam against carbapenem-resistant Enterobacteriaceae in an in vitro hollow-fiber model. Antimicrob Agents Chemother 62(2):e01969-17 https://doi.org/10.1128/AAC.01969-17
Hackel MA, Lomovskaya O, Dudley MN, Karlowsky JA, Sahm DF (2017) Evaluation of the in vitro activity of meropenem-vaborbactam against clinical isolates of KPC-positive Enterobacteriaceae. Antimicrob Agents Chemother 62(1):e01904–e01917 https://doi.org/10.1128/AAC.01904-17
Griffith DC, Rubino C, Loutit JS, Morgan EE, White D, Dudley MN (2014) A phase 1 study of the safety, tolerability, and pharmacokinetics of a single dose of the beta-lactamase inhibitor RPX7009 alone, meropenem alone, and both in combination (Carbavance) in healthy adult subjects. Abstract F-960. 54th Intersci Conf Antimicrob Agents Chemother; American Society of Microbiology, Washington D.C.
Wenzler E, Gotfried MH, Loutit JS et al (2015) Meropenem-RPX7009 concentrations in plasma, epithelial lining fluid, and alveolar macrophages of healthy adult subjects. Antimicrob Agents Chemother 59(12):7232–7239 https://doi.org/10.1128/AAC.01713-15
Griffith DC, Loutit JS, Morgan EE, Durso S, Dudley MN (2016) Phase 1 study of the safety, tolerability, and pharmacokinetics of the β-lactamase inhibitor vaborbactam (RPX7009) in healthy adult subjects. Antimicrob Agents Chemother 60(10):6326–6332 https://doi.org/10.1128/AAC.00568-16
Rubino CM, Bhavnani SM, Loutit JS, Lohse B, Dudley MN, Griffith DC (2018) Single-dose pharmacokinetics and safety of meropenem-vaborbactam in subjects with chronic renal impairment. Antimicrob Agents Chemother 62(3):e02103-17 https://doi.org/10.1128/AAC.02103-17
Kaye KS, Bhowmick T, Metallidis S et al (2018) Effect of meropenem-vaborbactam vs piperacillin-tazobactam on clinical cure or improvement and microbial eradication in complicated urinary tract infection: the TANGO I randomized clinical trial. JAMA 319(8):788–799 https://doi.org/10.1001/jama.2018.0438
Kaye KS, Vazquez J, Mathers A et al (2017) Meropenem-vaborbactam (VABOMERE) vs. best available therapy for CRE infections: TANGO II randomized, controlled phase 3 study results abstract. IDWeek, San Diego, p 1862
Weiss WJ, Pulse ME, Nguyen P et al (2017) Activity of meropenem-vaborbactam against carbapenem-resistant Enterobacteriaceae in a murine model of pyelonephritis. Antimicrob Agents Chemother 62(1):e01439-17 https://doi.org/10.1128/AAC.01439-17
Sabet M, Tarazi Z, Nolan T et al (2017) Activity of meropenem-vaborbactam in mouse models of infection due to KPC-producing carbapenem-resistant Enterobacteriaceae (CRE). Antimicrob Agents Chemother 62(1). https://doi.org/10.1128/AAC.01446-17
Hecker SJ, Reddy KR, Totrov M et al (2015) Discovery of a cyclic boronic acid β-lactamase inhibitor (RPX7009) with utility vs class A serine carbapenemases. J Med Chem 58(9):3682–3692. https://doi.org/10.1021/acs.jmedchem.5b00127
Zhanel GG, Wiebe R, Dilay L et al (2007) Comparative review of the carbapenems. Drugs 67(7):1027–1052
Bush K (2015) A resurgence of β-lactamase inhibitor combinations effective against multidrug-resistant gram-negative pathogens. Int J Antimicrob Agents 46(5):483–493. https://doi.org/10.1016/j.ijantimicag.2015.08.011
Codjoe FS, Donkor ES (2017) Carbapenem resistance: a review. Med Sci (Basel) 6(1). https://doi.org/10.3390/medsci6010001
Lomovskaya O, Sun D, Rubio-Aparicio D et al (2017) Vaborbactam: spectrum of beta-lactamase inhibition and impact of resistance mechanisms on activity in Enterobacteriaceae. Antimicrob Agents Chemother 61(11). https://doi.org/10.1128/AAC.01443-17
Lomovskaya O, Tsivkovski R (2014) The effect of Trp105 substitutions in KPC on interactions with the novel beta-lactamase inhibitor RPX7009 Abstract C-1194. 54th Interscience Conference on Antimicrobial Agents and Chemotherapy; American Society of Microbiology, Washington D.C.
Lapuebla A, Abdallah M, Olafisoye O et al (2015) Activity of meropenem combined with RPX7009, a novel β-lactamase inhibitor, against gram-negative clinical isolates in New York City. Antimicrob Agents Chemother 59(8):4856–4860 https://doi.org/10.1128/AAC.00843-15
Pfaller MA, Huband MD, Mendes RE, Flamm RK, Castanheira M (2018) In vitro activity of meropenem-vaborbactam and characterization of carbapenem resistance mechanisms among carbapenem-resistant Enterobacteriaceae from the 2015 meropenem-vaborbactam surveillance program. Int J Antimicrob Agents pii S0924-8579(18):30064–30065 https://doi.org/10.1016/j.ijantimicag.2018.02.021
Castanheira M, Huband MD, Mendes RE, Flamm RK (2017) Meropenem-vaborbactam tested against contemporary gram-negative isolates collected worldwide during 2014, including carbapenem-resistant, KPC-producing, multidrug-resistant, and extensively drug-resistant Enterobacteriaceae. Antimicrob Agents Chemother 61(9):e00567-17 https://doi.org/10.1128/AAC.00567-17
Sun D, Rubio-Aparicio D, Nelson K, Dudley MN, Lomovskaya O (2017) Meropenem-vaborbactam resistance selection, resistance prevention, and molecular mechanisms in mutants of KPC-producing Klebsiella pneumoniae. Antimicrob Agents Chemother 61(12):e01694-17 https://doi.org/10.1128/AAC.01694-17
Haidar G, Clancy CJ, Shields RK, Hao B, Cheng S, Nguyen MH (2017) Mutations in blaKPC-3 that confer ceftazidime-avibactam resistance encode novel KPC-3 variants that function as extended-spectrum β-lactamases. Antimicrob Agents Chemother 61(5):e02534-16 https://doi.org/10.1128/AAC.02534-16
MERREM® I.V. (meropenem for injection) package insert. FDA. https://www.accessdata.fda.gov/drugsatfda_docs/label/2008/050706s022lbl.pdf. Accessed 17 Nov 2017
Zhanel GG, Simor AE, Vercaigne L, Mandell L, Group CCD (1998) Imipenem and meropenem: comparison of in vitro activity, pharmacokinetics, clinical trials and adverse effects. Can J Infect Dis 9(4):215–228
VABOMERE™ (meropenem and vaborbactam) for injection. FDA. https://www.accessdata.fda.gov/drugsatfda_docs/label/2017/209776lbl.pdf. Accessed 17 Nov 2017
Wunderink R, Giamarellos-Bourboulis EJ, Rahav G et al (2017) Meropenem-vaborbactam (VABOMERE) vs. best available therapy for carbapenem-resistant Enterobacteriaceae infections in TANGO II: primary outcomes by site of infection abstract. IDWeek, San Diego, p 1867
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Key Points
• Meropenem-vaborbactam is a newly developed carbapenem and β-lactamase inhibitor indicated for complicated UTI and targets resistance mechanism KPC and KPC-like carbapenemases.
• Meropenem-vaborbactam has been found non-inferior to piperacillin-tazobactam for treatment of acute pyelonephritis in phase III study.
• Phase I and III studies report no serious adverse events with meropenem-vaborbactam, making it a safer choice over aminoglycosides, colistin, and tigecycline.
Rights and permissions
About this article
Cite this article
Lee, Y.R., Baker, N.T. Meropenem-vaborbactam: a carbapenem and beta-lactamase inhibitor with activity against carbapenem-resistant Enterobacteriaceae. Eur J Clin Microbiol Infect Dis 37, 1411–1419 (2018). https://doi.org/10.1007/s10096-018-3260-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10096-018-3260-4