Abstract
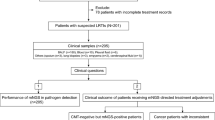
Timely diagnosis of tuberculosis (TB), caused by Mycobacterium tuberculosis (MTB), is only achieved for ~58% cases. An improved, accurate, time- and cost-effective method for bacteriological confirmation of MTB is necessary. We evaluated Mycotube, a new variant of Lowenstein–Jensen (LJ) culture medium, by comparing it with Mycobacterium Growth Indicator Tube (MGIT) 960 (gold standard), local LJ, and bioMérieux LJ-T in terms of isolation rate and time-to-growth. Pulmonary and extra-pulmonary samples from treatment-naïve suspects (n = 207) were decontaminated by the N-acetyl-l-cysteine-sodium hydroxide method and used to inoculate the four media. Subjective and objective parameters were used for evaluation. Mycotube yielded 140 positive results, compared to 162, 69, and 141 from MGIT, local LJ, and LJ-T, respectively. Of these, 139 (67%) were true-positive results and 1 (0.5%) was false-positive. The mean time-to-growth detection was 17.4 days for Mycotube, compared to 14.5, 28.1, and 16.5 days for MGIT, local LJ, and LJ-T, respectively. The mean time-to-growth for local LJ significantly differed from that for MGIT, but not those for LJ-T and Mycotube. No contamination was observed. Mycotube had a sensitivity of 85.8% and a specificity of 97.8% as compared to MGIT. Mycotube offers good results, comparable with those observed for conventional LJ. It requires only basic laboratory infrastructure. The overall cost of the test should be nearly three times lower than that of MGIT. Mycotube helps with TB diagnosis and generates pure isolates for drug susceptibility testing.




Similar content being viewed by others
References
World Health Organization (WHO) (2016) Global tuberculosis report 2016. WHO, Geneva, Switzerland
Pablos-Méndez A, Sterling TR, Frieden TR (1996) The relationship between delayed or incomplete treatment and all-cause mortality in patients with tuberculosis. JAMA 276:1223–1228
Zumla A, Davies PDO (2016) Progress towards achieving global tuberculosis control: so near, yet so far. Int J Tuberc Lung Dis 20:285–286
Kent PT, Kubica GP; Department of Health and Human Services, Public Health Service, Centers for Disease Control (1985) Public health mycobacteriology: a guide for the level III laboratory. Centers for Disease Control and Prevention (CDC), Atlanta
Rodrigues C, Shenai S, Sadani M et al (2009) Evaluation of the Bactec MGIT 960 TB system for recovery and identification of Mycobacterium tuberculosis complex in a high volume tertiary care centre. Indian J Med Microbiol 27:217–221
Brum CB, Ramos DF, de Souza Abilleira F et al (2016) The BACTEC MGIT™ 320 system as a laboratory tool to diagnose tuberculosis in a Brazilian hospital with a high prevalence of HIV infection. Rev Soc Bras Med Trop 49:112–114
Palomino JC (2005) Nonconventional and new methods in the diagnosis of tuberculosis: feasibility and applicability in the field. Eur Respir J 26:339–350
Joloba ML, Johnson JL, Feng PJ et al (2014) What is the most reliable solid culture medium for tuberculosis treatment trials? Tuberculosis (Edinb) 94:311–316
Oberhelman RA, Soto-Castellares G, Gilman RH et al (2010) Diagnostic approaches for paediatric tuberculosis by use of different specimen types, culture methods, and PCR: a prospective case–control study. Lancet Infect Dis 10:612–620
Bates M, Zumla A (2016) The development, evaluation and performance of molecular diagnostics for detection of Mycobacterium tuberculosis. Expert Rev Mol Diagn 16:307–322
Centers for Disease Control and Prevention (CDC) (2013) Core curriculum on tuberculosis: what the clinician should know. CDC, Atlanta
Asmar S, Chatellier S, Mirande C et al (2016) A chlorhexidine agar plate culture medium protocol to complement standard broth culture of Mycobacterium tuberculosis. Front Microbiol 7:30
Asmar S, Chatellier S, Mirande C et al (2015) A novel solid medium for culturing Mycobacterium tuberculosis isolates from clinical specimens. J Clin Microbiol 53:2566–2569
Harausz E, Lusiba JK, Nsereko M et al (2015) Comparison of MGIT and Myco/F lytic liquid-based blood culture systems for recovery of Mycobacterium tuberculosis from pleural fluid. J Clin Microbiol 53:1391–1394
Rageade F, Picot N, Blanc-Michaud A et al (2014) Performance of solid and liquid culture media for the detection of Mycobacterium tuberculosis in clinical materials: meta-analysis of recent studies. Eur J Clin Microbiol Infect Dis 33:867–870
Chihota VN, Grant AD, Fielding K et al (2010) Liquid vs. solid culture for tuberculosis: performance and cost in a resource-constrained setting. Int J Tuberc Lung Dis 14:1024–1031
da Silva Rezende Moreira A, Huf G, da Silva Vieira MAM et al (2015) Liquid vs solid culture medium to evaluate proportion and time to change in management of suspects of tuberculosis—a pragmatic randomized trial in secondary and tertiary health care units in Brazil. PLoS One 10:e0127588
American Thoracic Society (2000) Diagnostic standards and classification of tuberculosis in adults and children. Am J Respir Crit Care Med 161:1376–1395
Acknowledgments
The authors would like to thank the staff of the Mycobacteriology section of the Department of Microbiology of the hospital for their support, as well as the bioMérieux team for their funding support and mentorship.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Funding
This study was financially supported by bioMérieux India.
Conflict of interest
Five of the authors (SC, NB, AvB, NS, and BB) are employees of bioMérieux, a company designing, developing, and selling diagnostic tests in the field of infectious diseases. The opinions expressed in this manuscript are those of the authors, which may, in some cases, be different from formal company policy.
Ethical approval
The study design and corresponding documents were approved by the Institutional Ethics Committee of PD Hinduja Hospital and Medical Research Centre, Mumbai,
Informed consent
Informed consent was obtained from all individual participants included in the study.
Rights and permissions
About this article
Cite this article
Nambiar, R., Chatellier, S., Bereksi, N. et al. Evaluation of Mycotube, a modified version of Lowenstein–Jensen (LJ) medium, for efficient recovery of Mycobacterium tuberculosis (MTB). Eur J Clin Microbiol Infect Dis 36, 1981–1988 (2017). https://doi.org/10.1007/s10096-017-3052-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10096-017-3052-2