Abstract
Objective
To provide new and comprehensive evidence for diagnosis and management of FOSMN syndrome.
Methods
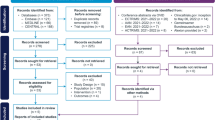
We reviewed our database to identify patients with FOSMN syndrome. Online database including PubMed, EMBASE, and OVID were also searched for relevant cases.
Results
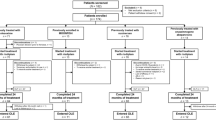
We identified a total of 71 cases, including 4 cases from our database and 67 ones from online searching. A predominance of male was observed [44 (62.0%)] with median onset age of 53 (range: 7–75) years old. The median (range) disease duration was 60 (3–552) months at the time of the visit. The initial symptoms could be sensory deficits in face (80.3%) or oral cavity (4.2%), bulbar paralysis (7.0%), dysosmia (1.4%), dysgeusia (4.2%), weakness or numbness of upper limbs (5.6%), or lower limbs (1.4%). Abnormal blink reflex was presented in 64 (90.1%) patients. CSF tests showed elevated protein level in 5 (7.0%) patients. Six (8.5%) patients had MND-related gene mutation. Five (7.0%) patients showed transient responsiveness to immunosuppressive therapy, then deteriorated relentlessly. Fourteen (19.7%) patients died, with an average survival time of around 4 years. Among them, five patients died of respiratory insufficiency.
Conclusion
The age of onset, progress of disease course, and prognosis of FOSMN syndrome could be varied significantly. The prerequisites of diagnosis were progressive and asymmetric lower motor neuron dysfunction, with sensory dysfunction which usually showed in face at the onset. Immunosuppressive therapy could be tried in some patients with suspected inflammatory clues. In general, FOSMN syndrome tended to be motor neuron disease with sensory involvement.

Similar content being viewed by others
References
Vucic S et al (2006) Facial onset sensory and motor neuronopathy (FOSMN syndrome): a novel syndrome in neurology. Brain 129(Pt 12):3384–3390
Karakis I, Vucic S, Srinivasan J (2014) Facial onset sensory and motor neuronopathy (FOSMN) of childhood onset. Muscle Nerve 50(4):614–615
Pinto W et al (2019) New findings in facial-onset sensory and motor neuronopathy (FOSMN) syndrome. Rev Neurol (Paris) 175(4):238–246
de Boer EMJ et al (2021) Facial onset sensory and motor neuronopathy: new cases, cognitive changes, and pathophysiology. Neurol Clin Pract 11(2):147–157
Liu Z et al (2022) Muscle and skin fibroblast TDP-43 expression, dynamic mutation analysis of NOTCH2NLC and C9orf72 in patients with FOSMN. Neurol Sci 43(11):6505–6510
Zheng Q et al (2016) Facial onset sensory and motor neuronopathy. Neurol Sci 37(12):1905–1909
Moher D et al (2009) Preferred reporting items for systematic reviews and meta-analyses: the PRISMA statement. PLoS Med 6(7):e1000097
Chen LX et al (2021) Taste loss as the sole presenting symptom in Chinese patient with facial onset sensory and motor neuronopathy. CNS Neurosci Ther 27(12):1610–1613
Hanawa H et al (2021) Facial onset sensory and motor neuropathy in a pain clinic outpatient: a case report. J Med Case Rep 15(1):609
Grudzińska M et al (2021) Facial onset sensory and motor neuronopathy syndrome - a rare variant of motor neurone disease. Neurol Neurochir Pol 55(3):325–327
Agirre-Beitia G et al (2020) FOSMN: A possible TDP-43 proteinopathy to consider in a patient with facial sensory symptoms. Neurol Clin Pract 10(5):e47–e50
Ohashi N et al (2020) Taste disorder in facial onset sensory and motor neuronopathy: a case report. BMC Neurol 20(1):71
Zhang W et al (2020) Facial-onset sensory motor neuronopathy syndrome is not always facial onset. J Clin Neurol 16(4):729–730
Rossor AM et al (2019) TDP43 pathology in the brain, spinal cord, and dorsal root ganglia of a patient with FOSMN. Neurology 92(9):e951–e956
Lange KS, Maier A, Leithner C (2020) Elevated CSF neurofilament light chain concentration in a patient with facial onset sensory and motor neuronopathy. Neurol Sci 41(1):217–219
Vázquez-Costa JF et al (2019) Facial onset sensory and motor neuronopathy: a motor neuron disease with an oligogenic origin? Amyotroph Lateral Scler Frontotemporal Degener 20(3–4):172–175
Zhang Q et al (2019) Facial onset motor and sensory neuronopathy syndrome with a novel TARDBP mutation. Neurologist 24(1):22–25
Truini A et al (2015) Differential trigeminal myelinated and unmyelinated nerve fiber involvement in FOSMN syndrome. Neurology 84(5):540–542
Dalla Bella E et al (2014) Heterozygous D90A-SOD1 mutation in a patient with facial onset sensory motor neuronopathy (FOSMN) syndrome: a bridge to amyotrophic lateral sclerosis. J Neurol Neurosurg Psychiatry 85(9):1009–1011
Ziso B et al (2015) Facial onset sensory and motor neuronopathy: further evidence for a TDP-43 proteinopathy. Case Rep Neurol 7(1):95–100
Broad R, Leigh PN (2015) Recognising facial onset sensory motor neuronopathy syndrome: insight from six new cases. Pract Neurol 15(4):293–297
Cruccu G et al (2014) Trigeminal isolated sensory neuropathy (TISN) and FOSMN syndrome: despite a dissimilar disease course do they share common pathophysiological mechanisms? BMC Neurol 14:248
Sonoda K et al (2013) TAR DNA-binding protein 43 pathology in a case clinically diagnosed with facial-onset sensory and motor neuronopathy syndrome: an autopsied case report and a review of the literature. J Neurol Sci 332(1–2):148–153
Dobrev D et al (2012) Facial onset sensorimotor neuronopathy syndrome: a case series. J Clin Neuromuscul Dis 14(1):7–10
Vucic S et al (2012) FOSMN syndrome: novel insight into disease pathophysiology. Neurology 79(1):73–79
Fluchere F et al (2011) Clinical features and follow-up of four new cases of facial-onset sensory and motor neuronopathy. Muscle Nerve 43(1):136–140
Isoardo G, Troni W (2008) Sporadic bulbospinal muscle atrophy with facial-onset sensory neuropathy. Muscle Nerve 37(5):659–662
Hokonohara T et al (2008) Facial onset sensory and motor neuronopathy (FOSMN) syndrome responding to immunotherapies. J Neurol Sci 275(1–2):157–158
Hardiman O et al (2017) Amyotrophic lateral sclerosis. Nat Rev Dis Primers 3:17085
Corcia P et al (2021) Split-hand and split-limb phenomena in amyotrophic lateral sclerosis: pathophysiology, electrophysiology and clinical manifestations. J Neurol Neurosurg Psychiatry 92(10):1126–1130
Mondelli M et al (2013) Sensory nerve action potential amplitude is rarely reduced in lumbosacral radiculopathy due to herniated disc. Clin Neurophysiol 124(2):405–409
Siedler G, Sommer C, Üçeyler N (2020) Pain-related evoked potentials in patients with large, mixed, and small fiber neuropathy. Clin Neurophysiol 131(3):635–641
Shefner JM et al (2020) A proposal for new diagnostic criteria for ALS. Clin Neurophysiol 131(8):1975–1978
Hammad M et al (2007) Clinical, electrophysiologic, and pathologic evidence for sensory abnormalities in ALS. Neurology 69(24):2236–2242
Menon P et al (2020) Cortical hyperexcitability evolves with disease progression in ALS. Ann Clin Transl Neurol 7(5):733–741
Mélé N et al (2018) Motor neuron disease of paraneoplastic origin: a rare but treatable condition. J Neurol 265(7):1590–1599
Thwaites G et al (2009) British Infection Society guidelines for the diagnosis and treatment of tuberculosis of the central nervous system in adults and children. J Infect 59(3):167–187
Al-Orainey IO et al (1987) Brucella meningitis. J Infect 14(2):141–145
Corcia P et al (2008) Causes of death in a post-mortem series of ALS patients. Amyotroph Lateral Scler 9(1):59–62
Ajroud-Driss S, Siddique T (2015) Sporadic and hereditary amyotrophic lateral sclerosis (ALS). Biochim Biophys Acta 1852(4):679–684
Acknowledgements
Authors would like to thank all the authors of the original articles.
Funding
National High Level Hospital Clinical Research Funding (2022-PUMCH-B-017), CAMS Innovation Fund for Medical Sciences (CIFMS 2021-I2M-1–003), Beijing Natural Science Foundation (7202158).
Author information
Authors and Affiliations
Contributions
Nan Hu: data analysis and writing of the main manuscript; Lei Zhang: data collection; Xunzhe Yang: data collection; Hanhui Fu: data collection; Liying Cui: design of the project; Mingsheng Liu: design of the project and revision of the main manuscript. All authors have read and approved the manuscript.
Corresponding author
Ethics declarations
Ethical approval
The study was approved by the Ethics Committee of the PUMCH (Peking Union Medical College Hospital).
Conflict of interest
None.
Informed consent statement
All included patients were provided written, informed consent.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Hu, N., Zhang, L., Yang, X. et al. Facial onset sensory and motor neuronopathy (FOSMN syndrome): Cases series and systematic review. Neurol Sci 44, 1969–1978 (2023). https://doi.org/10.1007/s10072-023-06703-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10072-023-06703-1