Abstract
Background
This systematic review study aims to evaluate the cognitive function of patients with mild hyperphenylalaninemia (mHPA) without treatment.
Methods
A systematic literature search was done from the 1st of May to the 30th of July in 2021 on published studies. The search strategy was ((hyperphenylalaninemia) OR (phenylketonuria) AND (cognition)). We use PubMed, Scopus, and the Web of Science databases. Studies which reported their findings regarding the cognitive function of patients with mHPA (screening serum phenylalanine > 120 and < 600 μmol/L) were included and reviewed.
Results
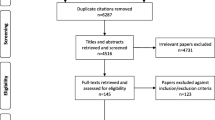
From initially retrieved 2805 studies, finally, 15 studies (10 on untreated patients with Phe levels below 360 μmol/L, 7 on untreated patients with Phe levels between 360 and 600 μmol/L, four 120–600 μmol/L) were selected. Most of the studies used the Wechsler Intelligence Scale for IQ evaluation, two (2/15) of them used the Stanford–Binet test and two used both tests. Four studies have reported a worse cognitive outcome compared to the control group, and in one study, relative defects in attention and working memory were reported. Other studies have reported normal IQ levels and no significant cognitive defects.
Conclusion
It is suggested that Phe levels between 120 and 360 μmol/L are generally safe. Some studies showed that untreated patients with higher levels might show some degrees of cognitive impairment. In conclusion, current knowledge is insufficient to state that treatment is not required for HPA patients to preserve their cognitive status, especially in patients with Phe levels of 360–600 μmol/L. Further studies with a larger sample size and standardized cognitive function evaluation tools are needed.

Similar content being viewed by others
Availability of data and materials
Not applicable.
References
Campistol J, Gassio R, Artuch R, Vilaseca MA, PFu Unit, (2011) Neurocognitive function in mild hyperphenylalaninemia. Dev Med Child Neurol 53(5):405–408
Evinc SG, Pektas E, Foto-Ozdemir D, Yildiz Y, Karaboncuk y, Bilginer-Gurbuz b et al (2018) Cognitive and behavioral impairment in mild hyperphenylalaninemia. Turk J Pediatr 60(6):617–624
Garcia MBG, Conde-Guzon P, Martin CA, Conde-Guzon MJ, Zuniga RV (2017) Neuropsychological assessment among children and adolescents with phenylketonuria and hyperphenylalaninemia and its relationship with plasma phenylalanine levels. Archivos Argentinos De Pediatria 115(3):267–273
Lichter-Konecki U, Vockley J (2019) Phenylketonuria: current treatments and future developments. Drugs 79(5):495–500
Hillert A, Anikster Y, Belanger-Quintana A, Burlina A, Burton BK, Carducci C et al (2020) The genetic landscape and epidemiology of phenylketonuria. Am J Hum Genet 107(2):234–250
Blau N, Shen N, Carducci C (2014) Molecular genetics and diagnosis of phenylketonuria: state of the art. Expert Rev Mol Diagn 14(6):655–671
van Spronsen FJ (2011) Mild hyperphenylalaninemia: to treat or not to treat. J Inherit Metab Dis 34(3):651–656
de la Parra A, Garcia MI, Waisbren SE, Cornejo V, Raimann E (2015) Cognitive functioning in mild hyperphenylalaninemia. Mol Genet Metab Rep 5:72–75
Hanley W (2011) Non-PKU mild hyperphenylalaninemia (MHP)—The dilemma. Mol Genet Metab 104(1–2):23–26
Burgard P, Ullrich K, Ballhausen D, Hennermann JB, Hollak CE, Langeveld M et al (2017) Issues with European guidelines for phenylketonuria. Lancet Diabetes Endocrinol 5(9):681–683
Van Wegberg A, MacDonald A, Ahring K, Bélanger-Quintana A, Blau N, Bosch A et al (2017) The complete European guidelines on phenylketonuria: diagnosis and treatment. Orphanet J Rare Dis 12(1):1–56
Mitchell JJ, Trakadis YJ, Scriver CR (2011) Phenylalanine hydroxylase deficiency. Genet Med: Off J Am Coll Med Genet 13(8):697–707
Moher D, Liberati A, Tetzlaff J, Altman DG (2010) Preferred reporting items for systematic reviews and meta-analyses: the PRISMA statement. Int J Surg 8(5):336–341
Bajalan Z, Moafi F, MoradiBaglooei M, Alimoradi Z (2019) Mental health and primary dysmenorrhea: a systematic review. J Psychosom Obstet Gynecol 40(3):185–194
Von Elm E, Altman DG, Egger M, Pocock SJ, Gøtzsche PC, Vandenbroucke JP et al (2014) The Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) Statement: guidelines for reporting observational studies. Int J Surg 12(12):1495–1499
Levy H, Shih V, Karolkewicz V, French W, Carr J, Cass V, et al (1971) Persistent Mild Hyperphenylalaninemia in the Untreated State. A Prospective Study. N Engl J Med 285(8):424–9
Waisbren S, Schnell R, Levy H (1984) Intelligence and personality characteristics in adults with untreated atypical phenylketonuria and mild hyperphenylalaninemia. J Pediatr 105(6):955–958
Weglage J, Ullrich K, Pietsch M, Fünders B, Güttler F, Harms E (1997) Intellectual, neurologic, and neuropsychologic outcome in untreated subjects with nonphenylketonuria hyperphenylalaninemia. Pediatr Res 42(3):378–384
Diamond A, Prevor MB, Callender G, Druin DP (1997) Prefrontal cortex cognitive deficits in children treated early and continuously for PKU. Monogr Soc Res Child Dev i-206.
Weglage J, Pietsch M, Feldmann R, Koch H-G, Zschocke J, Hoffmann G et al (2001) Normal clinical outcome in untreated subjects with mild hyperphenylalaninemia. Pediatr Res 49(4):532–536
Lou Smith M, Saltzman J, Klim P, Hanley WB, Feigenbaum A, Clarke JT (2000) Neuropsychological function in mild hyperphenylalaninemia. Am J Ment Retard 105(2):69–80
Jahja R, van Spronsen FJ, De Sonneville LM, van der Meere JJ, Bosch AM, Hollak CE et al (2017) Long-term follow-up of cognition and mental health in adult phenylketonuria: a PKU-COBESO study. Behav Genet 47(5):486–497
Couce ML, Castineiras DE, Boveda MD, Bana A, Cocho JA, Iglesias AJ et al (2011) Evaluation and long-term follow-up of infants with inborn errors of metabolism identified in an expanded screening programme. Mol Genet Metab 104(4):470–475
Netley C, Hanley W, Rudner H (1984) Phenylketonuria and its variants: observations on intellectual functioning. Can Med Assoc J 131(7):751
Gassió R, Artuch R, Vilaseca MA, Fusté E, Boix C, Sans A et al (2005) Cognitive functions in classic phenylketonuria and mild hyperphenylalaninaemia: experience in a paediatric population. Dev Med Child Neurol 47(7):443–448
Costello PM, Beasley MG, Tillotson SL, Smith I (1994) Intelligence in mild atypical phenylketonuria. Eur J Pediatr 153(4):260–263
Lang MJ, Koch R, Fishier K, Baker R (1989) Nonphenylketonuric hyperphenylalaninemia. Am J Dis Child 143(12):1464–1466
Groselj U, Tansek MZ, Podkrajsek KT, Battelino T (2014) Mutation analysis in mild hyperphenylalaninemia (HPA) patients-whom and what to screen? Clin Biochem 47(3):236
Weglage J, Schmidt E, Fünders B, Pietsch M, Ullrich K (1996) Sustained attention in untreated non-PKU-hyperphenylalaninemia. J Clin Exp Neuropsychol 18(3):343–348
Weglage J, Ullrich K, Pietsch M, Fünders B, Zass R, Koch H (1996) Untreated non-phenylketonuric-hyperphenylalaninaemia: intellectual and neurological outcome. Eur J Pediatr 155(1):S26–S28
Berman PW, Graham FK, Eichman PL, Waisman HA (1961) Psychologic and neurologic status of diet-treated phenylketonuric children and their sibligs. Pediatrics 28:924–934
Koch R, Güttler F, Guldberg P, Rouse B (1998) Mild hyperphenylalaninemia and heterozygosity of the phenylalanine hydroxylase gene. Mol Genet Metab 63(2):148–150
Levy H, Waisbren S, Lobbregt D, Allred E, Leviton A, Koch R et al (1996) Maternal non-phenylketonuric mild hyperphenylalaninemia. Eur J Pediatr 155:S20–S25
Viall S, Ayyub O, Rasberry M, Lyons K, Mew NA (2017) “Mild” hyperphenylalaninemia? A case series of seven treated patients following newborn screening. Mol Genet Metab 122(4):153–155
Funding
This study was funded by Isfahan University of medical sciences with the code number 55836.
Author information
Authors and Affiliations
Contributions
R. C. and F. Z. contributed to the design of the study and literature search and data collection. S. H. contributes to design and concepts, reading the final articles, manuscript revision, and approving the final submitted version. N. R. contributed to the concepts, data collection, manuscript revision, reading the final articles, and final approval. M. H. contributes to the concepts, reading the final articles, and manuscript revision. All authors have read and approved the final version of the manuscript.
Corresponding authors
Ethics declarations
Ethics approval and consent to participate
This study was accepted by Isfahan University of medical sciences' Ethics Committee with the code number of IR.MUI.MED.REC.1400.685.
Consent for publication
Not applicable.
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Rostampour, N., Chegini, R., Hovsepian, S. et al. Cognitive function in untreated subjects with mild hyperphenylalaninemia: a systematic review. Neurol Sci 43, 5593–5603 (2022). https://doi.org/10.1007/s10072-022-06194-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10072-022-06194-6