Abstract
Background
Vincristine (VCR), which is a key component of chemotherapy, is important for survival. VCR is associated with a well-known side effect, including neurotoxicity.
Aims
The aim of this study was to evaluate the features of vincristine-induced peripheral neuropathy (VIPN) and the effectiveness of pyridoxine plus pyridostigmine therapy in children with acute lymphoblastic leukemia.
Methods
The WHO and NCI CTCAE neurotoxicity scorings were used to evaluate VIPN at diagnosis, in the first month, and after the third month of the treatment. The clinical features of 23 patients having acute lymphoblastic leukemia with VIPN during the period of July 2013–February 2016 were prospectively evaluated.
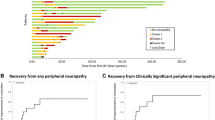
Results
The mean age was 72.8 ± 51.6 months, and 26.1%, 56.5%, and 17.4% were in standard, moderate, and high-risk groups, respectively. Neuropathy frequently occurred at induction (82.6%) and reinduction (17.4%) of the protocol. Drop foot (82.6%), leg pain (82.6%), and difficulty in walking (82.6%) were observed. The mean total cumulative dose of neuropathy occurrence was 5.6 ± 2.03 mg/m2. Our study showed that both the WHO and NCI CTCAE scorings were significantly improved via pyridoxine plus pyridostigmine therapy.
Conclusion
The WHO and NCI CTCAE scorings may be used for evaluating neuropathy at diagnosis and follow-up of neurotoxicity with treatment. Pyridoxine plus pyridostigmine therapy may be an effective option in the treatment of VIPN.
Similar content being viewed by others
References
Gurney JG, Severson RK, Davis S, Robison LL (1995) Incidence of cancer in children in the United States: sex-, race-, and 1-year age-specific rates by histologic type. Cancer 75:2186–2195
Gurney JG, Bondy ML (2006) Epidemiology of childhood cancer. In: Pizzo PA, Poplack DG (eds) Priciples and Practice of Pediatric Oncology, 5th edn. Lippincott Williams and Wilkins, Philadelphia, pp 2–14
Quasthoff S, Hartung HP (2002) Chemotherapy induced peripheral neuropathy. J Neurol 249:9–17
Toopchizade V, Hosseini M (2010) Electrophysiological signs of neuropathy caused by vincristine. Medical Journal of Tabriz University of Medical Sciences 31:19–25
Tuxen MK, Hansen SW (1994) Complication of treatment, Neurotoxicity secondary to antineoplastic drugs. Cancer Treat Rev 20:191–214
Hildebrandt G, Holler E, Woenkhaus M, Quarch G, Reichle A, Schalke B, Andreesen R (2000) Acute deterioration of Charcot–Marie–Tooth disease 1A (CMT 1A) following 2 mg of vincristine chemotherapy. Ann Oncol 11:743–747
Mols F, Beijers T, Lemmens V, van den Hurk CJ, Vreugdenhil G, van de Poll-Franse LV (2013) Chemotherapy-induced neuropathy and its association with quality of life among 2- to 11-year colorectal cancer survivors: results from the population based PROFILES registry. J Clin Oncol 31:2699–2707
Loprinzi CL, Qin R, Dakhil SR, Fehrenbacher L, Flynn KA, Atherton P, Seisler D, Qamar R, Lewis GC, Grothey A (2014) Phase III randomized, placebo-controlled, double-blind study of intravenous calcium and magnesium to prevent oxaliplatin induced sensory neurotoxicity (N08CB/Alliance). J Clin Oncol 32:997–1005
Hershman DL, Lacchetti C, Dworkin RH, Lavoie Smith EM, Bleeker J, Cavaletti G, Chauhan C, Gavin P, Lavino A, Lustberg MB, Paice J, Schneider B, Smith ML, Smith T, Terstriep S, Wagner-Johnston N, Bak K, Loprinzi CL, American Society of Clinical Oncology (2014) Prevention and management of chemotherapy-induced peripheral neuropathy in survivors of adult cancers: American Society of Clinical Oncology clinical practice guideline. J Clin Oncol 32:1941–1967
Ozyurek H, Turker H, Akbalik M, Bayrak AO, Ince H, Duru F (2007) Pyridoxine and pyridostigmine treatment in vincristine-induced neuropathy. Pediatr Hematol Oncol 24:447–452
Castellanos AM, Fields WS (1986) Grading of neurotoxicity in cancer therapy. J Clin Oncol 4:1277–1278
Basch E, Reeve BB, Mitchell SA et al (2014). Development of the National Cancer Institute’s patient-reported outcomes version of the common terminology criteria for adverse events (PRO-CTCAE). J Nalt Cancer Inst 106
Dueck AC, Mendoza TR, Mitchell SA, Reeve BB, Castro KM, Rogak LJ, Atkinson TM, Bennett AV, Denicoff AM, O'Mara AM, Li Y, Clauser SB, Bryant DM, Bearden JD 3rd, Gillis TA, Harness JK, Siegel RD, Paul DB, Cleeland CS, Schrag D, Sloan JA, Abernethy AP, Bruner DW, Minasian LM, Basch E, National Cancer Institute PRO-CTCAE Study Group (2015) Validity and reliability of the US National Cancer Institute’s patient-reported outcomes version of the common terminology criteria for adverse events (PRO-CTCAE). JAMA Oncol 1:1051–1059
Ocean AJ, Vhadat LT (2004) Chemotherapy-induced peripheral neuropathy: pathogenesis and emerging therapies. Support Care Cancer 12:619–625
Bay A, Yilmaz C, Yilmaz N, Oner AF (2006) Vincristine induced cranial polyneuropathy. Indian J Pediatr 73:531–533
McGuire S, Gospe S, Dahl G (1989) Acute vincristine neurotoxicity in the presence of motor and sensory neuropathy type 1. Med Pediatr Oncol 17:520–523
Talebian A, Goudarzi RM, Mohammadzadeh M, Mirzadeh AS (2014) Vincristine-Induced Cranial Neuropathy. Iran J Child Neurol 8:66–68
Ngamphaiboon N, Sweeney R, Wetzler M, Wang ES (2010) Pyridoxine treatment of vincristine-induced cranial polyneuropathy in an adult patient with acute lymphocytic leukemia: case report and review of the literature. Leuk Res 34:e194–e196
Müller L, Kramm CM, Tenenbaum T, Wessalowski R, Göbel U (2004) Treatment of vincristine-induced bilateral ptosis with pyridoxine and pyridostigmine. Pediatr Blood Cancer 42:287–288
Cassidy J, Paul J, Soukop M, Habeshaw T, Reed NS, Parkin D, Kaye SB (1998) Clinical trials of nimodipine as a potential neuroprotector in ovarian cancer patients treated with cisplatin. Cancer Chemother Pharmacol 41:161–166
Pace BA, Savarese A, Picardo M (2003) Neuroprotective effect of vitamin E supplementation in patients treated with cisplatin chemotherapy. J Clin Oncol 21:927–931
Liu M-Y, Huang X-E (2015) Effects of analgecine on oxaliplatin-ınduced neurotoxicity in patients with gastrointestinal cancer. Asian Pac J Cancer Prev 16:4465–4468
Haim N, Epelbaum R, Ben-Shahar M, Yarnitsky D, Simri W, Robinson E (1994) Full dose vincristine (without 2-mg dose limit) in the treatment of lymphomas. Cancer 73:2515–2519
Postma TJ, Benard BA, Huijgens PC, Ossenkoppele GJ, Heimans JJ (1993) Long-term effects of vincristine on the peripheral nervous system. J Neuro-Oncol 15:23–27
Ramchandren S, Leonard M, Mody RJ, Donohue JE, Moyer J, Hutchinson R, Gurney JG (2009) Peripheral neuropathy in survivors of childhood acute lymphoblastic leukemia. J Peripher Nerv Syst 14:184–189
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflicts of interest.
Ethical approval
Ethics committee approval was obtained from Dr. Behçet Uz Children's hospital on 24.12.2015 (protocol number:2015/51).
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Aydin Köker, S., Gözmen, S., Demirağ, B. et al. Effect of pyridoxine plus pyridostigmine treatment on vincristine-induced peripheral neuropathy in pediatric patients with acute lymphoblastic leukemia: a single-center experience. Neurol Sci 42, 3681–3686 (2021). https://doi.org/10.1007/s10072-020-04970-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10072-020-04970-w