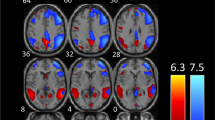
Abstract
In humans, spatial integration develops slowly, continuing through childhood into adolescence. On the assumption that this protracted course depends on the formation of networks with slowly developing top-down connections, we compared effective connectivity in the visual cortex between 13 children (age 7–13) and 14 adults (age 21–42) using a passive perceptual task. The subjects were scanned while viewing bilateral gratings, which either obeyed Gestalt grouping rules [colinear gratings (CG)] or violated them [non-colinear gratings (NG)]. The regions of interest for dynamic causal modeling were determined from activations in functional MRI contrasts stimuli > background and CG > NG. They were symmetrically located in V1 and V3v areas of both hemispheres. We studied a common model, which contained reciprocal intrinsic and modulatory connections between these regions. An analysis of effective connectivity showed that top-down modulatory effects generated at an extrastriate level and interhemispheric modulatory effects between primary visual areas (all inhibitory) are significantly weaker in children than in adults, suggesting that the formation of feedback and interhemispheric effective connections continues into adolescence. These results are consistent with a model in which spatial integration at an extrastriate level results in top-down messages to the primary visual areas, where they are supplemented by lateral (interhemispheric) messages, making perceptual encoding more efficient and less redundant. Abnormal formation of top-down inhibitory connections can lead to the reduction of habituation observed in migraine patients.




Similar content being viewed by others
References
Burkhalter A (1993) Development of forward and feedback connections between areas V1 and V2 of human visual cortex. Cereb Cortex 3:476–487
Burkhalter A, Bernardo KL, Charles V (1993) Development of local circuits in human visual cortex. J Neurosci 13:1916–1931
Stevens MC, Kiehl KA, Pearlson GD, Calhoun VD (2007) Functional neural networks underlying response inhibition in adolescents and adults. Behav Brain Res 181:12–22
Hwang K, Velanova K, Luna B (2010) Strengthening of top-down frontal cognitive control networks underlying the development of inhibitory control: a functional magnetic resonance imaging effective connectivity study. J Neurosci 30:15535–15545
Filippi M, Rocca MA (2008) Headache and migraine. Neurol Sci 29(Suppl 3):336–338
Chiapparini L, Ferraro S, Grazzi L, Bussone G (2010) Neuroimaging in chronic migraine. Neurol Sci 31(Suppl 1):S19–S22
Abu-Arefeh I, Russell G (1994) Prevalence of headache and migraine in school children. BMJ 309:765–769
Rao RP, Ballard DH (1999) Predictive coding in the visual cortex: a functional interpretation of some extra-classical receptive-field effects. Nat Neurosci 2:79–87
Friston KJ, Harrison L, Penny W (2003) Dynamic causal modelling. Neuroimage 19:1273–1302
Knyazeva MG, Fornari E, Meuli R, Innocenti G, Maeder P (2006) Imaging of a synchronous neuronal assembly in the human visual brain. Neuroimage 29:593–604
Knyazeva MG, Fornari E, Meuli R, Maeder P (2006) Interhemispheric integration at different spatial scales: the evidence from EEG coherence and FMRI. J Neurophysiol 96:259–275
Kovacs I, Kozma P, Feher A, Benedek G (1999) Late maturation of visual spatial integration in humans. Proc Natl Acad Sci USA 96:12204–12209
Atkinson J (2000) The developing visual brain. Oxford University Press, Oxford, p 211
Norcia A (2003) Development of spatial selectivity and response timing in humans. In: Werner JS, Chalupa LM (eds) The visual neurosciences. MIT Press, Cambridge, pp 1603–1615
Fornari E, Knyazeva MG, Meuli R, Maeder P (2007) Myelination shapes functional activity in the developing brain. Neuroimage 38:511–518
Braunitzer G, Rokszin A, Kobor J, Nagy A, Sztriha L, Benedek G (2011) Development of visual contour integration in children with migraine without aura. Cephalalgia 31:1048–1056
Friston K, Kiebel S (2009) Predictive coding under the free-energy principle. Philos Trans R Soc Lond B Biol Sci 364:1211–1221
Murray SO, Schrater P, Kersten D (2004) Perceptual grouping and the interactions between visual cortical areas. Neural Netw 17:695–705
Braddick OJ, O’Brien JM, Wattam-Bell J, Atkinson J, Hartley T, Turner R (2001) Brain areas sensitive to coherent visual motion. Perception 30:61–72
Ban H, Yamamoto H, Fukunaga M, Nakagoshi A, Umeda M, Tanaka C, Ejima Y (2006) Toward a common circle: interhemispheric contextual modulation in human early visual areas. J Neurosci 26:8804–8809
Cardin V, Friston KJ, Zeki S (2011) Top-down modulations in the visual form pathway revealed with dynamic causal modeling. Cereb Cortex 21:550–562
Rytsar R, Fornari E, Frackowiak RS, Ghika JA, Knyazeva MG (2011) Inhibition in early Alzheimer’s disease: an fMRI-based study of effective connectivity. Neuroimage 57:1131–1139
Knyazeva MG (2013) Splenium of corpus callosum: patterns of interhemispheric interaction in children and adults. Neural Plast 2013:639430
Ashburner J, Friston KJ (2000) Voxel-based morphometry-the methods. Neuroimage 11:805–821
Krasnow B, Tamm L, Greicius MD, Yang TT, Glover GH, Reiss AL, Menon V (2003) Comparison of fMRI activation at 3 and 1.5 T during perceptual, cognitive, and affective processing. Neuroimage 18:813–826
Buxton RB, Wong EC, Frank LR (1998) Dynamics of blood flow and oxygenation changes during brain activation: the balloon model. Magn Reson Med 39:855–864
Seghier ML, Zeidman P, Neufeld NH, Leff AP, Price C (2010) Identifying abnormal connectivity in patients using dynamic causal modeling of fMRI responses. Front Syst Neurosci 4:142
Stephan KE, Friston KJ (2010) Analyzing effective connectivity with functional magnetic resonance imaging. Wiley Interdiscip Rev Cogn Sci 1:446–459
Friston KJ, Li B, Daunizeau J, Stephan KE (2011) Network discovery with DCM. Neuroimage 56:1202–1221
Simion F, Regolin L, Bulf H (2008) A predisposition for biological motion in the newborn baby. Proc Natl Acad Sci USA 105:809–813
Spelke ES (1990) Principles of object perception. Cogn Sci 14:29–56
Humphrey GK, Humphrey DE, Muir DW, Dodwell PC (1986) Pattern perception in infants: effects of structure and transformation. J Exp Child Psychol 41:128–148
Lewis TL, Ellemberg D, Maurer D, Dirks M, Wilkinson F, Wilson HR (2004) A window on the normal development of sensitivity to global form in Glass patterns. Perception 33:409–418
Hadad BS, Kimchi R (2006) Developmental trends in utilizing perceptual closure for grouping of shape: effects of spatial proximity and collinearity. Percept Psychophys 68:1264–1273
Hadad BS, Maurer D, Lewis TL (2011) Long trajectory for the development of sensitivity to global and biological motion. Dev Sci 14:1330–1339
Mondloch CJ, Geldart S, Maurer D, de Schonen S (2003) Developmental changes in the processing of hierarchical shapes continue into adolescence. J Exp Child Psychol 84:20–40
Scherf KS, Behrmann M, Kimchi R, Luna B (2009) Emergence of global shape processing continues through adolescence. Child Dev 80:162–177
Farber DA, Knyazeva MG (1991) Electrophysiological correlates of interhemispheric interaction in ontogenesis. In: Ramaekers G, Njiokiktjien C (eds) Pediatric behavioural neurology. Suyi Publications, Amsterdam, pp 86–99
Innocenti GM, Kiper DC, Knyazeva MG, Deonna TW (1999) On nature and limits of cortical developmental plasticity after an early lesion, in a child. Restor Neurol Neurosci 15:219–227
Knyazeva MG, Innocenti GM (2001) EEG coherence studies in the normal brain and after early-onset cortical pathologies. Brain Res Brain Res Rev 36:119–128
Knyazeva MG, Maeder P, Kiper DC, Deonna T, Innocenti GM (2002) Vision after early-onset lesions of the occipital cortex: II. Physiological studies. Neural Plast 9:27–40
Bullier J, Hupe JM, James AC, Girard P (2001) The role of feedback connections in shaping the responses of visual cortical neurons. Prog Brain Res 134:193–204
Gilbert CD, Sigman M (2007) Brain states: top-down influences in sensory processing. Neuron 54:677–696
Altmann CF, Bulthoff HH, Kourtzi Z (2003) Perceptual organization of local elements into global shapes in the human visual cortex. Curr Biol 13:342–349
Kourtzi Z, Tolias AS, Altmann CF, Augath M, Logothetis NK (2003) Integration of local features into global shapes: monkey and human FMRI studies. Neuron 37:333–346
Coppola G, Pierelli F, Schoenen J (2009) Habituation and migraine. Neurobiol Learn Mem 92:249–259
Schoenen J (2006) Neurophysiological features of the migrainous brain. Neurol Sci 27(Suppl 2):S77–S81
Pro S, Tarantino S, Capuano A, Vigevano F, Valeriani M (2014) Primary headache pathophysiology in children: the contribution of clinical neurophysiology. Clin Neurophysiol 125:6–12
Clark A (2013) Whatever next? Predictive brains, situated agents, and the future of cognitive science. Behav Brain Sci 36:181–204
Carmeli C, Lopez-Aguado L, Schmidt KE, De Feo O, Innocenti GM (2007) A novel interhemispheric interaction: modulation of neuronal cooperativity in the visual areas. PLoS One 2:e1287
Bocci T, Caleo M, Giorli E, Barloscio D, Maffei L, Rossi S, Sartucci F (2011) Transcallosal inhibition dampens neural responses to high contrast stimuli in human visual cortex. Neuroscience 187:43–51
Genc E, Bergmann J, Tong F, Blake R, Singer W, Kohler A (2011) Callosal connections of primary visual cortex predict the spatial spreading of binocular rivalry across the visual hemifields. Front Hum Neurosci 5:161
Lehky SR (1988) An astable multivibrator model of binocular rivalry. Perception 17:215–228
Oelkers-Ax R, Bender S, Just U, Pfuller U, Parzer P, Resch F, Weisbrod M (2004) Pattern-reversal visual-evoked potentials in children with migraine and other primary headache: evidence for maturation disorder? Pain 108:267–275
Conner IP, Sharma S, Lemieux SK, Mendola JD (2004) Retinotopic organization in children measured with fMRI. J Vis 4:509–523
Dougherty RF, Ben-Shachar M, Bammer R, Brewer AA, Wandell BA (2005) Functional organization of human occipital-callosal fiber tracts. Proc Natl Acad Sci USA 102:7350–7355
Saenz M, Fine I (2010) Topographic organization of V1 projections through the corpus callosum in humans. Neuroimage 52:1224–1229
Aboitiz F, Scheibel AB, Fisher RS, Zaidel E (1992) Fiber composition of the human corpus callosum. Brain Res 598:143–153
Innocenti GM, Price DJ (2005) Exuberance in the development of cortical networks. Nat Rev Neurosci 6:955–965
Pinto JG, Hornby KR, Jones DG, Murphy KM (2010) Developmental changes in GABAergic mechanisms in human visual cortex across the lifespan. Front Cell Neurosci 4:16
Eickhoff SB, Stephan KE, Mohlberg H, Grefkes C, Fink GR, Amunts K, Zilles K (2005) A new SPM toolbox for combining probabilistic cytoarchitectonic maps and functional imaging data. Neuroimage 25:1325–1335
Acknowledgments
This work was supported by the Centre d’Imagerie BioMédicale (CIBM) of the University of Lausanne (UNIL), the Swiss Federal Institute of Technology Lausanne (EPFL), the University of Geneva (UniGe), the Centre Hospitalier Universitaire Vaudois (CHUV), the Hôpitaux Universitaires de Genève (HUG) and the Leenaards and the Jeantet Foundations.
Conflict of interest
All the authors certify that there is no actual or potential conflict of interest in relation to this article.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Fornari, E., Rytsar, R. & Knyazeva, M.G. Development of spatial integration depends on top-down and interhemispheric connections that can be perturbed in migraine: a DCM analysis. Neurol Sci 35 (Suppl 1), 215–224 (2014). https://doi.org/10.1007/s10072-014-1777-6
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10072-014-1777-6