Abstract
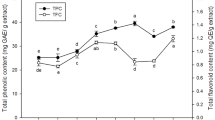
Chestnut inner shell (CIS) was fermented at 30 °C for 12 day using Monascus kaoliang, either in solid or submerged state, and alcohol extracts (70% ethanol) of the fermented CIS were examined for their antioxidant (total phenol content and diphenylpicrylhydrazyl radical scavenging activity) and in vitro cosmeceutical activities (tyrosinase and elastase inhibitory activities). Both activities were significantly increased by the M. kaoliang-fermentation, more apparently by submerged fermentation (SMF) than by solid-state fermentation (SSF). The cosmeceutical activity reached its maximum value on the 3rd day of fermentation. The residual amounts of phenolic acids and catechins in the CIS extracts were increased by the fermentation, up to 395.0 and 344.3 µg/g, respectively. More phenolic acids were produced by SMF than SSF, whereas more catechins were produced by SSF than SMF. Therefore, SMF using M. kaoliang was an efficient process for the utilization of CIS as a source of cosmeceuticals.




Similar content being viewed by others
Abbreviations
- CIS:
-
Chestnut inner shell
- DPPH:
-
2,2-Diphenyl-1-picrylhydrazyl
- GAE:
-
Gallic acid equivalent
- HPLC:
-
High-performance liquid chromatography
- PDA:
-
Potato dextrose agar
- PFTE:
-
Polytetrafluoroethylene
- SSF:
-
Solid-state fermentation
- SMF:
-
Submerged fermentation
- TPC:
-
Total phenolic content
- MEB:
-
Malt extract broth
References
Abd Razak DL, Abd Rashid NY, Jamaluddin A, Sharifudin SA, Long K. Enhancement of phenolic acid content and antioxidant activity of rice bran fermented with Rhizopus oligosporus and Monascus purpureus. Biocatalysis and Agricultural Biotechnology. 4: 33-38 (2014). https://doi.org/10.1016/j.intimp.2013.12.020
Almeida ABD, Santos NH, Lima TMD, Santana RV, Oliveira Filho JGD, Peres DS, Egea MB. Pigment bioproduction by Monascus purpureus using corn bran, a byproduct of the corn industry. Biocatalysis and Agricultural Biotechnology. 32: 101931 (2021). https://doi.org/10.1016/j.bcab.2021.101931
Bei Q, Liu Y, Wang L, Chen G, Wu Z. Improving free, conjugated, and bound phenolic fractions in fermented oats (Avena sativa L.) with Monascus anka and their antioxidant activity. Journal of Functional Foods. 32: 185-194 (2017)
Cai Z-Y, Li X-M, Liang J-P, Xiang L-P, Wang K-R, Shi Y-L, Yang R, Shi M, Ye J-H, Lu J-L, Zheng X-Q, Liang Y-R. Bioavailability of tea catechins and its improvement. Molecules. 23(9): 2346 (2018). https://doi.org/10.3390/molecules23092346
Caprarulo V, Giromini C, Rossi L. Review: Chestnut and quebracho tannins in pig nutrition: the effects on performance and intestinal health. Animal. 15: 100064 (2021). https://doi.org/10.1016/j.animal.2020.100064
Cavalcanti RMF, Jorge JA, Guimarães LHS. Characterization of Aspergillus fumigatus CAS-21 tannase with potential for propyl gallate synthesis and treatment of tannery effluent from leather industry. 3 Biotech. 8(6): 270 (2018). https://doi.org/10.1007/s13205-018-1294-z. Epub 2018 May 25. PMID: 29868308; PMCID: PMC5970104.
Chávez-González ML, Guyot S, Rodríguez-Herrera R, Prado-Barragán A, Aguilar CN. Production profiles of phenolics from fungal tannic acid biodegradation in submerged and solid-state fermentation. Process Biochemistry. 49(4): 541-546 (2014). https://doi.org/10.1016/j.procbio.2014.01.031
Cheng J, Choi B-K, Yang SH, Suh J-W. Effect of fermentation on the antioxidant activity of rice bran by Monascus pilosus KCCM60084. Journal of Applied Biological Chemistry. 59: 57-62 (2016). https://doi.org/10.3839/jabc.2016.011
Das Mohapatra PK, Mondal KC, Pati BR. Production of tannase through submerged fermentation of tannin-containing plant extracts by Bacillus licheniformis KBR6. Polish Journal of Microbiolgy. 55(4): 297-301 (2006)
Era B, Floris S, Sogos V, Porcedda C, Piras A, Medda R, Fais A, Pintus F. Anti-aging potential of extracts from washingtonia filifera seeds. Plants. 10(1): 151 (2021). https://doi.org/10.3390/plants10010151
Fang X, Du M, Liu T, Fang Q, Liao Z, Zhong Q, Chen J, Meng X, Zhou S, Wang J. Changes in the biotransformation of green tea catechins induced by different carbon and nitrogen sources in Aspergillus niger RAF106. Frontiers in Microbiology. 10: 2521 (2019). https://doi.org/10.3389/fmicb.2019.02521
Hefni ME, Amann LS, Witthöft CMJFAM. A HPLC-UV method for the quantification of phenolic acids in cereals. Food Analytical Methods, 12(12): 2802-2812 (2019)
Hu M, Yang X, Chang X. Bioactive phenolic components and potential health effects of chestnut shell: A review. Journal of Food Biochemistry. 45(4): e13696 (2021). https://doi.org/10.1111/jfbc.13696
Huang Q, Chai W-M, Ma Z-Y, Deng W-L, Wei Q-M, Song S, Zou Z-R, Peng Y-Y. Antityrosinase mechanism of ellagic acid in vitro and its effect on mouse melanoma cells. Journal of Food Biochemistry. 43(11): e12996. (2019). https://doi.org/10.1111/jfbc.12996
Huynh NT, Van Camp J, Smagghe G, Raes KJ. Improved release and metabolism of flavonoids by steered fermentation processes : a review. International Journal of Molecular Sciences. 15(11): 19369-19388 (2014). https://doi.org/10.3390/ijms151119369
Jamaluddin A, Rashid NYA, Razak DLA, Sharifudin SA, Long K. Effect of fungal fermentation on tyrosinase and elastase inhibition activity in rice bran. Agriculture and Agricultural Science Procedia. 2: 252-256 (2014)
Cheon J. What is the global production of chestnuts?. https://www.tridge.com/market-guides/posts/how-is-the-global-production-of-chestnuts. Accessed Oct 21, 2021
Kwon K-R, Alam MB, Park J-H, Kim T-H, Lee S-H. Attenuation of UVB-induced photo-aging by polyphenolic-rich spatholobus suberectus stem extract via modulation of MAPK/AP-1/MMPs signaling in human keratinocytes. Nutrients. 11(6): 1341. (2019). https://doi.org/10.3390/nu11061341
Marinova G, Batchvarov VJ. Evaluation of the methods for determination of the free radical scavenging activity by DPPH. Bulgarian Journal of Agricultural Science. 17(1): 11-24 (2011)
Maurelli L, Ionata E, La Cara F, Morana A. Chestnut shell as unexploited source of fermentable sugars: effect of different pretreatment methods on enzymatic saccharification. Applied Biochemistry and Biotechnology. 170(5): 1104-1118 (2013). https://doi.org/10.1007/s12010-013-0264-5
Mithieux SM, Weiss AS. Elastin. Advances in Protein Chemistry. 70: 437-461 (2005). https://doi.org/10.1016/s0065-3233(05)70013-9
Mohamed MS, Saleh AM, Abdel-Farid IB, El-Naggar SAJ. Growth, hydrolases and ultrastructure of Fusarium oxysporum as affected by phenolic rich extracts from several xerophytic plants. Pesticide Biochemistry and Physiology. 141: 57-64 (2017). https://doi.org/10.1016/j.pestbp.2016.11.007
Nguyen T, Garrahan MA, Nanc, SA, Seeger CE, Wong C. Assimilation of cholesterol by Monascus purpureus. Journal of Fungi 6(4): 352 (2020)
Noh J-R, Gang G-T, Kim Y-H, Yang K-J, Hwang J-H, Lee H-S, Oh W-K, Song K-S, Lee C-H. Antioxidant effects of the chestnut (Castanea crenata) inner shell extract in t-BHP-treated HepG2 cells, and CCl4- and high-fat diet-treated mice. Food and Chemical Toxicology. 48(11): 3177-3183 (2010). https://doi.org/10.1016/j.fct.2010.08.018
Pyo Y-H, Jin Y-J. Monascus-mediated fermentation improves the nutricosmetic potentials of soybeans. Food Science and Biotechnology. 25(3): 883-891 (2016). https://doi.org/10.1007/s10068-016-0145-0
Ritthibut N, Lim ST, Oh SJ. In vitro cosmeceutical activity of alcoholic extract from chestnut inner shell fermented with Aspergillus sojae. Food Science and Biotechnology. 31(4): 443-450 (2022)
Ritthibut N, Oh S-J, Lim S-T. Enhancement of bioactivity of rice bran by solid-state fermentation with Aspergillus strains. LWT-Food Science and Technology. 135: 110273 (2021)
Robledo A, Aguilera-Carbó A, Rodriguez R, Martinez JL, Garza Y, Aguilar CN. Ellagic acid production by Aspergillus niger in solid state fermentation of pomegranate residues. Journal of Industrial Microbiology and Biotechnology. 35(6): 507-513 (2008)
Rzepka Z, Buszman E, Beberok A, Wrześniok D. From tyrosine to melanin: Signaling pathways and factors regulating melanogenesis. Postepy Higieny i Medycyny Doswiadczalnej. 70: 695-708 (2016). https://doi.org/10.5604/17322693.1208033
Sabu A, Augur C, Swati C, Pandey A J. Tannase production by Lactobacillus sp. ASR-S1 under solid-state fermentation. Process Biochemistry. 41(3): 575-580. (2006). https://doi.org/10.1016/j.procbio.2005.05.011
Shoko T, Maharaj VJ, Naidoo D, Tselanyane M, Nthambeleni R, Khorombi E, Apostolides Z. Anti-aging potential of extracts from Sclerocarya birrea (A. Rich.) Hochst and its chemical profiling by UPLC-Q-TOF-MS. BMC Complementary and Alternative Medicine, 18(1): 1-14 (2018). https://doi.org/10.1186/s12906-018-2112-1
Srianta I, Zubaidah E, Estiasih T, Iuchi Y, Yamada M. Antioxidant activity of pigments derived from Monascus purpureus-fermented rice, corn, and sorghum. International Food Research Journal. 24(3): 1186 (2017)
Srinivasulu C, Ramgopal M, Ramanjaneyulu G, Anuradha CM, Suresh Kumar C. Syringic acid (SA): A review of its occurrence, biosynthesis, pharmacological and industrial importance. Biomedicine & Pharmacotherapy. 108: 547-557 (2018). https://doi.org/10.1016/j.biopha.2018.09.069
Subramaniyam R, Vimala R. Solid state and submerged fermentation for the production of bioactive substances: a comparative study. International Journal of Natural Sciences 3(3): 480-486 (2012)
Tawaha K, Alali FQ, Gharaibeh M, Mohammad M, El-Elimat T. Antioxidant activity and total phenolic content of selected Jordanian plant species. Food Chemistry. 104(4): 1372-1378 (2007). https://doi.org/10.1016/j.foodchem.2007.01.064
Tuyen PT, Xuan TD, Khang DT, Ahmad A, Quan NV, Tu Anh TT, Anh LH, Minh TN. Phenolic compositions and antioxidant properties in bark, flower, inner skin, kernel and leaf extracts of Castanea crenata Sieb. et Zucc. Antioxidants. 6(2): 31 (2017). https://doi.org/10.3390/antiox6020031
Van de Lagemaat J, Pyle DL. Solid‐state fermentation: A continuous process for fungal tannase production. Biotechnology and Bioengineering. 87(7): 924-929 (2004). https://doi.org/10.1002/bit.20206
Vendruscolo F, Bühler RMM, de Carvalho JC, de Oliveira D, Moritz DE, Schmidell W, Ninow JL. Monascus: a reality on the production and application of microbial pigments. Applied Biochemistry and Biotechnology. 178(2): 211-223 (2016). https://doi.org/10.1007/s12010-015-1880-z
Wang H, Provan GJ, Helliwell K. HPLC determination of catechins in tea leaves and tea extracts using relative response factors. Food Chemistry 81(2): 307-312 (2003). https://doi.org/10.1016/S0308-8146(02)00510-1
Wen D, Li C, Di H, Liao Y, Liu HJ. A universal HPLC method for the determination of phenolic acids in compound herbal medicines. Journal of Agricultural and Food Chemistry. 53(17): 6624-6629 (2005). https://doi.org/10.1021/jf0511291
Wen Q, Cao X, Chen Z, Xiong Z, Liu J, Cheng Z, Zheng Z, Long C, Zheng B, Huang ZJ. An overview of Monascus fermentation processes for monacolin K production. Open Chemistry. 18(1): 10-21 (2020). https://doi.org/10.1515/chem-2020-0006
Yao J, Guo GS, Ren GH, Liu YH. Production, characterization and applications of tannase. Journal of Molecular Catalysis B 101: 137-147 (2014)
Zivkovic J, Mujic I, Zekovic Z, Nikolić G, Vidovic S, Mujić A. Extraction and analysis of condensed tannins in castanea sativa mill. Journal of Central European Agriculture. 10(3): 283-288 (2009)
Acknowledgements
This research was supported by the Basic Science Research Program through the National Research Foundation of Korea (NRF) funded by the Ministry of Education [grant number NRF-2021R1A6A3A01086642].
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
The authors disclose the following: Jin, Ritthibut, Oh, and Lim declare that they have no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Jin, Yy., Ritthibut, N., Lim, ST. et al. Antioxidant and in vitro cosmeceutical activities of chestnut inner shell fermented by Monascus kaoliang. Food Sci Biotechnol 32, 813–822 (2023). https://doi.org/10.1007/s10068-022-01225-6
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10068-022-01225-6