Abstract
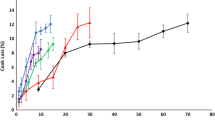
Changes in white shrimp (Litopenaeus vannamei) protein during thermal denaturation were studied using Raman spectroscopy and isotopic H/D exchange. Denaturation of shrimp protein began after heating for 10 min at 50°C. A decrease in the percentage of α-helices accompanied by an increase in the percentage of β-sheets occurred while the total percentage of disordered structures increased. With extension of the exchange time, the relative intensity of the O-D bond increased, accompanied by a higher relative O-D bond intensity for heated shrimp, compared with unheated shrimp. H/D exchange revealed a higher rate of deuteration kinetics in heated shrimp than for unheated shrimp, especially during the first 2 h, consistent with water loss from denatured white shrimp protein. Physical property changes in muscle tissue can be caused by changes in hydrogen bonding and hydrophobicity during thermal processes.
Similar content being viewed by others
References
Niamnuy S, Devahastin S, Soponronnarit S. Changes in protein compositions and their effects on physical changes of shrimp during boiling in salt solution. Food Chem. 108: 165–175 (2008)
Tapaneyasin R, Devahastin S, Tansakul A. Drying methods and quality of shrimp dried in a jet-spouted bed dryer. J. Food Process Eng. 28: 35–52 (2005)
Chalida N, Soraya, K, Sa kamon D. Artificial neural network modeling of physicochemical changes of shrimp during boiling. LWT-Food Sci. Technol. 45: 110–116 (2011)
Michalczyk M, Surowka K. Changes in protein fractions of rainbow trout (Oncorhynchus mykiss) gravads during production and storage. Food Chem. 104: 1006–1013 (2007)
Sriket P, Benjakul S, Visessanguan W, Kijroongrojana K. Comparative studies on chemical composition and thermal properties of black tiger shrimp (Penaeus monodon) and white shrimp (Penaeus vannamei) meats. Food Chem. 103: 1199–1207 (2007)
Herrero AM, Carmona P, Cofrades S, Jimnez-Colmenero F. Raman spectroscopic determination of structural changes in meat batters upon soy protein addition and heat treatment. Food Res. Int. 41: 765–772 (2008)
Herrero AM. Raman spectroscopy for monitoring protein structure in muscle food systems. Crit. Rev. Food Sci. 48: 512–523 (2008)
Herrero AM. Raman spectroscopy a promising technique for quality assessment of meat and fish: A review. Food Chem. 107: 1642–1651 (2008)
Herrero AM, Carmona P, García ML, Solas MT, Careche M. Ultrastructural changes and structure and mobility of myowater in frozen-stored hake (Merluccius merluccius L.) muscle: Relationship with functionality and texture. J. Agr. Food Chem. 53: 2558–2566 (2005)
Niamnuy C, Devahastin S, Soponronnarit S. Quality changes of shrimp during boiling in salt solution. J. Food Sci. 72: S289–S297 (2007)
Ignacio SG, Pedro C, Pilar M, Javier B, Isabel SA, Arantxa RC, Mercedes C. Protein and water structural changes in fish surimi during gelation as revealed by isotopic H/D exchange and Raman spectroscopy. Food Chem. 106: 56–64 (2008)
Alix AJP, Pedanou G, Berjot M. Determination of the quantitative secondary structure of proteins by using some parameters of the Raman amide I band. J. Mol. Struct. 174: 159–164 (1988)
Herrero AM, Carmona P, Careche M. Raman spectroscopic study of structural changes in hake (Merluccius merluccius L.) muscle proteins during frozen storage. J. Agr. Food Chem. 52: 2147–2153 (2004)
Offer G, Trinick J. A unifying hypothesis for the mechanism of changes in the water-holding capacity of meat. J. Sci. Food Agr. 34: 1018–1019 (1983)
Offer G, Knight P. The structural basis of water-holding in meat. pp. 53–173. In: Developments in Meat Science-4. Lawrie R (ed). Elsevier Applied Science, New York, NY, USA (1988)
Straadt IK, Rasmussen M, Anderson HJ, Bertram HC. Aging-induced changes in microstructure and water distribution in fresh and cooked pork in relation to water-holding capacity and cooking loss-A combined confocal laser scanning microscopy (CLSM) and low-field nuclear magnetic resonance relaxation study. Meat Sci. 75: 687–695 (2007)
Frushour BG, Koening JL. Raman spectroscopy of proteins. Vol. 2, pp. 35–97. In: Advances in Infrared and Raman Spectroscopy. Clark RJH, Hester RE (eds). Heyden, London, UK (1975)
Nishinari K, Zhang H, Ikeda S. Hydrocolloid gels of polysaccharides and proteins. Curr. Opin. Colloid In. 5: 195–201 (2000)
Niwa E. Chemistry of surimi gelation. pp. 389–427. In: Surimi Technology. Lanier TC, Lee C (eds). Marcel Dekker, New York, NY, USA (1992)
Bouraoui M, Nakai S, Li-Chan E. In situ investigation of protein structure in Pacific whiting surimi and gels using Raman spectroscopy. Food Res. Int. 30: 65–72 (1997)
Carmona P, Molina M, Rodríguez-Casado A. Raman study of the thermal behaviour and conformational stability of basic pancreatic trypsin inhibitor. Eur. Biophys. J. 32: 137–143 (2003)
Nazlin KH, Guillermo A, Shuryo N, Eunice CYL. Raman spectral analysis in the C-H stretching region of proteins and amino acids for Investigation of hydrophobic interactions. J. Agr. Food Chem. 47: 924–933 (1999)
Maeda Y, Kitano H. The structure of water in polymer systems as revealed by Raman spectroscopy. Spectrochim. Acta. 51: 2433–2446 (1995)
Arteaga GE. Assessment of protein surface hydrophobicity by spectroscopic methods and its relation to emulsifying properties of proteins. PhD thesis, University of British Columbia, Vancouver, Canada (1994)
Lafleur M, Pigeon M, Pézolet M, Caillé JP. Raman spectrum of interstitial water in biological systems. J. Phys. Chem. 93: 1522–1526 (1989)
Leikin S, Parsegian VA, Yang WH, Walrafen E. Raman spectral evidence for hydration forces between collagen triple helices. P. Natl. Acad. Sci. US. 94: 11312–11317 (1997)
Careche M, Herrero AM, Rodriguez-Casado A, Del Mazo ML, Carmona P. Structural changes of hake (Merluccius merluccius L.) fillets: Effects of freezing and frozen storage. J. Agr. Food Chem. 47: 952–959 (1999)
Keyi W. Introduction of protein. Science Press, Beijing, China. pp. 120–121 (2007)
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Gao, R., Feng, X., Li, W. et al. Changes in properties of white shrimp (Litopenaeus vannamei) protein during thermal denaturation. Food Sci Biotechnol 25, 21–26 (2016). https://doi.org/10.1007/s10068-016-0003-0
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10068-016-0003-0