Abstract
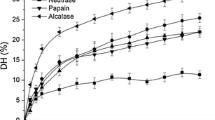
Antioxidant activities and functional properties of tea seed protein hydrolysates (TSPH) prepared using alcalase with different (10, 20, 30 and 40%) values of the degree of hydrolysis (DH) were investigated. The effect of hydrolysis time on antioxidant activity was also investigated. As the hydrolysis time was extended, the DPPH radical scavenging activity increased and finally reached a plateau, the copper chelating capacity decreased, and the superoxide radical scavenging and iron chelating activities increased initially, then subsequently slowed. The solubility, foaming properties, and emulsification properties of TSPH were affected by pH and DH. As the DH value increased, the DPPH radical scavenging activity and the reducing power increased and the copper chelating capacity decreased. TSPH at 20 and 30% DH values exhibited higher superoxide radical scavenging and stronger iron chelating activities respectively, than TSPH at other DH values. The DH value of TSPH affected the antioxidant activity and functional properties.
Similar content being viewed by others
References
Lee CP, Shih PH, Hsu CL, Yen GC. Hepatoprotection of tea seed oil (Camellia oleifera Abel.) against CCl4-induced oxidative damage in rats. Food Chem. Toxicol. 45: 888–895 (2007)
Lee CP, Yen GC. Antioxidant activity and bioactive compounds of tea seed (Camellia oleifera Abel.) oil. J. Agr. Food. Chem. 54: 779–784 (2006)
He SN, Gu Y. The Comprehensive utilization of camellia fruits. Vol. 37, pp. 104–107. In: American Camellia Yearbook. American Camellia Society, Fort Valley, GA, USA (1982)
Gong J, Li Z, Zhong H, Wan Q. Optimazation of enzymatic hydrolysis conditions of oil-tea-seed meal using response surface methodology. J. Chin. Cereal. Oil Assoc. 7: 80–89 (2010)
Moure A, Sineiro J, Domínguez H, Parajó JC. Functionality of oilseed protein products: A review. Food Res. Int. 39: 945–963 (2006)
Mannheim A, Cheryan M. Enzyme-modified proteins from corn gluten meal: Preparation and functional properties. J. Am. Oil Chem. Soc. 69: 1163–1169 (1992)
Kristinsson HG, Rasco BA. Biochemical and functional properties of Atlantic salmon (Salmo salar) muscle proteins hydrolyzed with different alkaline proteases. J. Agr. Food. Chem. 48: 657–666 (2000)
Sarmadi BH, Ismail A. Antioxidative peptides from food proteins: A review. Peptides 31: 1949–1956 (2010)
Klompong V, Benjakul S, Kantachote D, Shahidi F. Antioxidative activity and functional properties of protein hydrolysate of yellow stripe trevally (Selaroides leptolepis) as influenced by the degree of hydrolysis and enzyme type. Food Chem. 102: 1317–1327 (2007)
Kristinsson HG, Rasco BA. Fish protein hydrolysates: Production, biochemical, and functional properties. Crit. Rev. Food Sci. Nutr. 40: 43–81 (2000)
Naqash SY, Nazeer R. Antioxidant and functional properties of protein hydrolysates from pink perch (Nemipterus japonicus) muscle. J. Food Sci. Technol. 50: 972–978 (2013)
Liu Q, Kong B, Xiong YL, Xia X. Antioxidant activity and functional properties of porcine plasma protein hydrolysate as influenced by the degree of hydrolysis. Food Chem. 118: 403–410 (2010)
Zhang J, Zhang H, Wang L, Guo X, Wang X, Yao H. Isolation and identification of antioxidative peptides from rice endosperm protein enzymatic hydrolysate by consecutive chromatography and MALDI-TOF/TOF MS/MS. Food Chem. 119: 226–234 (2010)
Pan M, Jiang TS, Pan JL. Antioxidant activities of rapeseed protein hydrolysates. Food Bioprocess. Tech. 4: 1144–1152 (2011)
Benjakul S, Morrissey MT. Protein hydrolysates from Pacific whiting solid wastes. J. Agr. Food Chem. 45: 3423–3430 (1997)
Wu H-C, Chen H-M, Shiau C-Y. Free amino acids and peptides as related to antioxidant properties in protein hydrolysates of mackerel (Scomber austriasicus). Food Res. Int. 36: 949–957 (2003)
Zhang T, Li Y, Miao M, Jiang B. Purification and characterisation of a new antioxidant peptide from chickpea (Cicer arietium L.) protein hydrolysates. Food Chem. 128: 28–33 (2011)
Oyaizu M. Studies on products of browning reaction: Antioxidative activity of products of browning reaction. Jpn. J. Nutr. 44: 307–315 (1986)
Decker EA, Welch B. Role of ferritin as a lipid oxidation catalyst in muscle food. J. Agr. Food. Chem. 38: 674–677 (1990)
Saiga A, Tanabe S, Nishimura T. Antioxidant activity of peptides obtained from porcine myofibrillar proteins by protease treatment. J. Agr. Food Chem. 51: 3661–3667 (2003)
Castellani O, Martinet V, David BE, Guerin-Dubiard C, Anton M. Egg yolk phosvitin: preparation of metal-free purified protein by fast protein liquid chromatography using aqueous solvents. J. Chromatogr. B 791: 273–284 (2003)
Lowry OH, Rosebrough NJ, Farr AL, Randall RJ. Protein measurement with the Folin phenol reagent. J. Biol. Chem. 193: 265–275 (1951)
Shahidi F, Han XQ, Synowiecki J. Production and characteristics of protein hydrolysates from capelin (Mallotus villosus). Food Chem. 53: 285–293 (1995)
Sathe S, Salunkhe D. Functional properties of the Great Northern bean (Phaseolus vulgaris L.) proteins: Emulsion, foaming, viscosity, and gelation properties. J. Food Sci. 46: 71–81 (1981)
Pearce KN, Kinsella JE. Emulsifying properties of proteins: Evaluation of a turbidimetric technique. J. Agr. Food Chem. 26: 716–723 (1978)
Jamdar S, Rajalakshmi V, Pednekar M, Juan F, Yardi V, Sharma A. Influence of degree of hydrolysis on functional properties, antioxidant activity and ACE inhibitory activity of peanut protein hydrolysate. Food Chem. 121: 178–184 (2010)
Kristinsson HG, Rasco BA. Biochemical and functional properties of Atlantic salmon (Salmo salar) muscle proteins hydrolyzed with different alkaline proteases. J. Agr. Food Chem. 48: 657–666 (2000)
Li B, Chen F, Wang X, Ji B, Wu Y. Isolation and identification of antioxidative peptides from porcine collagen hydrolysate by consecutive chromatography and electrospray ionization-mass spectrometry. Food Chem. 102: 1135–1143 (2007)
Gbogouri G, Linder M, Fanni J, Parmentier M. Influence of hydrolysis degree on the functional properties of salmon byproducts hydrolysates. J. Food Sci. 69: C615–C622 (2004)
Je JY, Qian ZJ, Kim SK. Antioxidant peptide isolated from muscle protein of bullfrog, Rana catesbeiana Shaw. J. Med. Food 10: 401–407 (2007)
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Li, X., Shen, S., Deng, J. et al. Antioxidant activities and functional properties of tea seed protein hydrolysates (Camellia oleifera Abel.) influenced by the degree of enzymatic hydrolysis. Food Sci Biotechnol 23, 2075–2082 (2014). https://doi.org/10.1007/s10068-014-0282-2
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10068-014-0282-2