Abstract
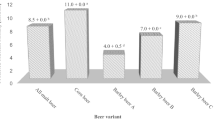
Pressure-assisted thermal processing (PATP) inactivation kinetics of Bacillus amyloliquefaciens spores in deionized water (DIW) were evaluated for egg patty mince (EPM) and green pea puree (GPP). Recovery of PATP-injured spores during storage was determined. The number of B. amyloliquefaciens spores in DIW was reduced more than 6 log when treated at 121°C and 700 MPa, including a come-up time reduction (3.32 log MPN/g) and a pressure holding time reduction (3.55 log MPN/g). Treatments at 700 MPa in combination with 105°C for 16 min, 115°C for 5 min, or 121°C for 3 min, decreased B. amyloliquefaciens spore populations in EPM to levels undetectable using an enrichment procedure. No significant (p>0.05) recovery of PATP-injured spores was observed in EPM and GPP during storage for 8 weeks, compared with controls. These results provide useful information for enhancing microbial lethality of PATP-resistant bacterial spores in low-acid foods.
Similar content being viewed by others
References
Rajan S, Ahn J, Balasubramaniam VM, Yousef AE. Combined pressure-thermal inactivation kinetics of Bacillus amyloliquefaciens spores in egg patty mince. J. Food Prot. 69: 853–860 (2006)
Ratphitagsanti W, Ahn J, Balasubramaniam VM, Yousef AE. Influence of pressurization rate and pressure pulsing on the inactivation of Bacillus amyloliquefaciens spores during pressureassisted thermal processing. J. Food Prot. 72: 775–782 (2009)
Matser AM, Krebbers B, van den Berg RW, Bartels PV. Advantages of high pressure sterilisation on quality of food products. Trends Food Sci. Technol. 15: 79–85 (2004)
Busta FF, Suslow TV, Parish ME, Beuchat LR, Farber JM, Garrett EH, Harris LJ. The use of indicators and surrogate microorganisms for the evaluation of pathogens in fresh and fresh-cut produce. Compr. Rev. Food Sci Food Saf. 2: 179–185 (2003)
Margosch D, Gazle MG, Ehrmann MA, Vogel RF. Pressure inactivation of Bacillus endospores. Appl. Environ. Microbiol. 70: 7321–7328 (2004)
San Martin MF, Barbosa-Canovas GV, Swanson BG. Food processing by high hydrostatic pressure. Crit. Rev. Food Sci. Nutr. 46: 627–645 (2002)
Gao Y-L. Efficacy of high hydrostatic pressure and mild heat to reduce Geobacillus stearothermophilus as 1.1923 spores in model food systems. J. Food Saf. 30: 124–140 (2010)
Panagou EZ, Tassou CC, Manitsa C, Mallidis C. Modelling the effect of high pressure on the inactivation kinetics of a pressureresistant strain of Pediococcus damnosus in phosphate buffer and gilt-head seabream (Sparus aurata). J. Appl. Microbiol. 102: 1499–1507 (2007)
Buzrul S, Alpas H, Bozoglu F. Use of Weibull frequency distribution model to describe the inactivation of Alicyclobacillus acidoterrestris by high pressure at different temperatures. Food Res. Int. 38: 151–157 (2005)
Ahn J, Balasubramaniam VM, Yousef AE. Inactivation kinetics of selected aerobic and anaerobic bacterial spores by pressure-assisted thermal processing. Int. J. Food Microbiol. 113: 321–329 (2007)
Whiting RC, Sackitey S, Calderone S, Morely K, Phillips JG. Model for the survival of Staphylococcus aureus in nongrowth environments. Int. J. Food Microbiol. 31: 231–243 (1996)
Ahn J, Balasubramaniam VM. Effects of inoculum level and pressure pulse on the inactivation of Clostridium sporogenes spores by pressure-assisted thermal processing. J. Microbiol. Biotechnol. 17: 616–623 (2007)
Ross T. Indices for performance evaluation of predictive models in food microbiology. J. Appl. Bacteriol. 81: 501–508 (1996)
Garthright WE, Blodgett RJ. FDA’s preferred MPN methods for standard, large or unusual tests, with a spreadsheet. Food Microbiol. 20: 439–445 (2003)
Hartmann C, Delgado A, Szymczyk J. Convective and diffusive transport effects in a high pressure induced inactivation process of packed food. J. Food Eng. 59: 33–44 (2003)
Palou E, Lopez-Malo A, Barbosa-Cánovas GV, Welti-Chanes J, Davidson PM, Swanson BG. High hydrostatic pressure come-up time and yeast viability. J. Food Prot. 61: 1657–1660 (1998)
Palou E, Lopez-Malo A, Barbosa-Canovas GV, Welti-Chanes J, Swanson BG. Kinetic analysis of Zygosaccharomyces bailii inactivation by high hydrostatic pressure. LWT-Food Sci. Technol. 30: 703–708 (1997)
Garcia-Gonzalez L, Geeraerd AH, Elst K, van Ginneken L, van Impe JF, Devlieghere F. Influence of type of microorganism, food ingredients and food properties on high-pressure carbon dioxide inactivation of microorganisms. Int. J. Food Microbiol. 129: 253–263 (2009)
Solomon EB, Hoover DG. Inactivation of Campylobacter jejuni by high pressure hydrostatic pressure. Lett. Appl. Microbiol. 38: 505–509 (2004)
Garriga M, Grebol N, Aymerich MT, Monfort JM, Hugas M. Microbial inactivation after high-pressure processing at 600 MPa in commercial meat products over its shelf life. Innov. Food Sci. Emerg. Technol. 5: 451–457 (2004)
Smelt JPPM, Hellemons JC, Wouters PC, van Gerwen SJC. Physiological and mathematical aspects in setting criteria for decontamination of foods by physical means. Int. J. Food Microbiol. 78: 57–77 (2002)
Mozhaev VV, Heremans K, Frank J, Masson P, Balny C. Exploiting the effects of high hydrostatic-pressure in biotechnological applications. Trends Biotechnol. 12: 493–501 (1994)
Garcia-Graells C, Masschalck B, Michiels CW. Inactivation of Escherichia coli in milk by high-hydrostatic-pressure treatment in combination with antimicrobial peptides. J. Food Prot. 62: 1248–1254 (1999)
Ananta E, Heinz V, Schluter O, Knorr D. Kinetic studies on highpressure inactivation of Bacillus stearothermophilus spores suspended in food matrices. Innov. Food Sci. Emerg. Technol. 2: 261–272 (2001)
Corradini MG, Peleg M. Demonstration of the applicability of the Weibull-log-logistic survival model to the isothermal and nonisothermal inactivation of Escherichia coli K-12 MG1655. J. Food Prot. 67: 2617–2621 (2004)
Peleg M, Normand MD. Calculating microbial survival parameters and predicting survival curves from non-isothermal inactivation data. Crit. Rev. Food Sci. Nutr. 44: 409–418 (2004)
Xiong R, Xie G, Edmondson AE, Sheard MA. A mathematical model for bacterial inactivation. Int. J. Food Microbiol. 46: 45–55 (1999)
Kilimann KV, Hartmann C, Delgado A, Vogel RF, Ganzle MG. Combined high pressure and temperature induced lethal and sublethal injury of Lactococcus lactis-Application of multivariate statistical analysis. Int. J. Food Microbiol. 109: 25–33 (2006)
Garcia-Graells C, van Opstal I, Vanmuysen SCM, Michiels CW. The lactoperoxidase system increases efficacy of high-pressure inactivation of foodborne bacteria. Int. J. Food Microbiol. 81: 211–221 (2003)
Fujii K, Ohtani A, Watanabe J, Ohgoshi H, Fujii T, Honma K. High-pressure inactivation of Bacillus cereus spores in the presence of argon. Int. J. Food Microbiol. 72: 239–242 (2002)
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Ahn, J., Balasubramaniam, B.V.M. Inactivation kinetics and injury recovery of Bacillus amyloliquefaciens spores in low-acid foods during pressure-assisted thermal processing. Food Sci Biotechnol 23, 1851–1857 (2014). https://doi.org/10.1007/s10068-014-0253-7
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10068-014-0253-7