Abstract
Background
In patients with systemic lupus erythematosus (SLE), a higher frequency of atherosclerotic lesions is associated with poor prognosis. Hydroxychloroquine (HCQ) has been reported to improve the lifespan and the prognosis of dyslipidaemia in patients with SLE, but the mechanism is unclear. We investigated the effect of supplemental HCQ treatment on the levels of serum cytokines associated with atherosclerosis in patients with stable SLE.
Methods
Patients with SLE who received supplemental HCQ and maintained low disease activity between January 2016 and September 2020 were included in this study. Disease activity was assessed using Safety of Estrogens in Lupus National Assessment-SLE Disease Activity Index, Cutaneous Lupus Erythematous Disease Area and Severity Index, and Lupus Low Disease Activity State. Serum complement titres, anti-dsDNA antibodies, and serum cytokines (adiponectin, resistin, and leptin) were analyzed before and after HCQ treatment.
Results
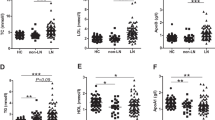
Forty-one patients (4 males and 37 females, mean age 41.3 ± 13.2 years) were included. Serum adiponectin levels were significantly increased after 3 months of HCQ treatment compared to baseline, and serum resistin levels were significantly reduced. The change in serum resistin level after HCQ administration was correlated with a significant reduction in serum TNF-α, interleukin (IL)-6, IL-8, and IL-1RA levels.
Conclusions
Supplemental HCQ treatment in patients with SLE improved adipokine levels. HCQ may improve prognosis by controlling disease activity in SLE and reducing risk factors for atherosclerosis.
Key Points • Hydroxychloroquine has been reported to improve the prognosis of dyslipidaemia in patients with SLE, but the underlying mechanism is unclear. • In this study, hydroxychloroquine improved adipokine levels in patients with SLE, implicating adipokines as a potential mechanism underlying the benefit of hydroxychloroquine on dyslipidaemia. • Supplemental hydroxychloroquine should be considered in patients with SLE harboring lipid abnormalities and risk factors for atherosclerosis. |

Similar content being viewed by others
Data availability
The dataset supporting the conclusions of this article is available upon reasonable request.
References
Murphy G, Lisnevskaia L, Isenberg D (2013) Systemic lupus erythematosus and other autoimmune rheumatic diseases: challenges to treatment. Lancet 382:809–818
Fava A, Petri M (2019) Systemic lupus erythematosus: diagnosis and clinical management. J Autoimmun 96:1–13
Mosca M, Tani C, Aringer M, Bombardieri S, Boumpas D, Brey R et al (2010) European League Against Rheumatism recommendations for monitoring patients with systemic lupus erythematosus in clinical practice and in observational studies. Ann Rheum Dis 69:1269–1274
Fanouriakis A, Kostopoulou M, Alunno A, Aringer M, Bajema I, Boletis JN et al (2019) 2019 update of the EULAR recommendations for the management of systemic lupus erythematosus. Ann Rheum Dis 78:736–745
Durcan L, Winegar DA, Connelly MA, Otvos JD, Magder LS, Petri M (2016) Longitudinal evaluation of lipoprotein parameters in systemic lupus erythematosus reveals adverse changes with disease activity and prednisone and more favorable profiles with hydroxychloroquine therapy. J Rheumatol 43:745–750
Katz G, Smilowitz NR, Blazer A, Clancy R, Buyon JP, Berger JS (2019) Systemic lupus erythematosus and increased prevalence of atherosclerotic cardiovascular disease in hospitalized patients. Mayo Clin Proc 94:1436–1443
Babary H, Liu X, Ayatollahi Y, Chen XP, Doo L, Uppaluru LK et al (2018) Favorable effects of hydroxychloroquine on serum low density lipid in patients with systemic lupus erythematosus: a systematic review and meta-analysis. Int J Rheum Dis 21:84–92
Yang DH, Leong PY, Sia SK, Wang YH, Wei JC (2019) Long-term hydroxychloroquine therapy and risk of coronary artery disease in patients with systemic lupus erythematosus. J Clin Med 8:796
Rempenault C, Combe B, Barnetche T, Gaujoux-Viala C, Lukas C, Morel J et al (2018) Metabolic and cardiovascular benefits of hydroxychloroquine in patients with rheumatoid arthritis: a systematic review and meta-analysis. Ann Rheum Dis 77:98–103
Wasko MCM, McClure CK, Kelsey SF, Huber K, Orchard T, Toledo FGS (2015) Antidiabetogenic effects of hydroxychloroquine on insulin sensitivity and beta cell function: a randomised trial. Diabetologia 58:2336–2343
Toledo FGS, Miller RG, Helbling NL, Zhang Y, DeLany JP (2021) The effects of hydroxychloroquine on insulin sensitivity, insulin clearance and inflammation in insulin-resistant adults: a randomized trial. Diabetes Obes Metab 23:1252–1261
Petri M (1996) Thrombosis and systemic lupus erythematosus: the Hopkins Lupus Cohort perspective. Scand J Rheumatol 25:191–193
Ruiz-Irastorza G, Egurbide M-V, Pijoan J-I, Garmendia M, Villar I, Martinez-Berriotxoa A, Erdozain J-G, Aguirre C (2006) Effect of antimalarials on thrombosis and survival in patients with systemic lupus erythematosus. Lupus 15:577–583
Mok CC, Tse SM, Chan KL, Ho LY (2018) Effect of immunosuppressive therapies on survival of systemic lupus erythematosus: a propensity score analysis of a longitudinal cohort. Lupus 27:722–727
Petri M, Orbai AM, Alarcón GS, Gordon C, Merrill JT, Fortin PR et al (2012) Derivation and validation of the Systemic Lupus International Collaborating Clinics classification criteria for systemic lupus erythematosus. Arthritis Rheum 64:2677–2686
Bertsias GK, Tektonidou M, Amoura Z, Aringer M, Bajema I, Berden JH et al (2012) Joint European League Against Rheumatism and European Renal Association-European Dialysis and Transplant Association (EULAR/ERA-EDTA) recommendations for the management of adult and paediatric lupus nephritis. Ann Rheum Dis 71:1771–1782
Petri M, Kim MY, Kalunian KC, Grossman J, Hahn BH, Sammaritano LR et al (2005) Combined oral contraceptives in women with systemic lupus erythematosus. N Engl J Med 353:2550–2558
Franklyn K, Lau CS, Navarra SV, Louthrenoo W, Lateef A, Hamijoyo L et al (2016) Definition and initial validation of a Lupus Low Disease Activity State (LLDAS). Ann Rheum Dis 75:1615–1621
Albrecht J, Taylor L, Berlin JA, Dulay S, Ang G, Fakharzadeh S et al (2005) The CLASI (Cutaneous Lupus Erythematosus Disease Area and Severity Index): an outcome instrument for cutaneous lupus erythematosus. J Invest Dermatol 125:889–894
Li X, Zhang D, Vatner DF, Goedeke L, Hirabara SM, Zhang Y et al (2020) Mechanisms by which adiponectin reverses high fat diet-induced insulin resistance in mice. Proc Natl Acad Sci U S A 117:32584–32593
Ahlstrom P, Rai E, Chakma S, Cho HH, Rengasamy P, Sweeney G (2017) Adiponectin improves insulin sensitivity via activation of autophagic flux. J Mol Endocrinol 59:339–350
Qiao X, Zhou ZC, Niu R, Su YT, Sun Y, Liu HL et al (2019) Hydroxychloroquine improves obesity-associated insulin resistance and hepatic steatosis by regulating lipid metabolism. Front Pharmacol 10:855
Gamez-Nava JI, Diaz-Rizo V, Perez-Guerrero EE, Muñoz-Valle JF, Saldaña-Cruz AM, Fajardo-Robledo NS et al (2020) Assessment of serum macrophage migration inhibitory factor (MIF), adiponectin, and other adipokines as potential markers of proteinuria and renal dysfunction in lupus nephritis: a cross-sectional study. Biomark Res 8:55
Hutcheson J, Ye Y, Han J, Arriens C, Saxena R, Li QZ et al (2015) Resistin as a potential marker of renal disease in lupus nephritis: an adipokine marker in lupus nephritis. Clin Exp Immunol 179:435–443
Chen H, Shi B, Feng X, Kong W, Chen W, Geng L et al (2015) Leptin and neutrophil-activating peptide 2 promote Mesenchymal stem cell senescence through activation of the phosphatidylinositol 3-kinase/Akt pathway in patients with systemic lupus Erythematosus. Arthritis Rheumatol 67:2383–2393
Diaz-Rizo V, Bonilla-Lara D, Gonzalez-Lopez L, Sanchez-Mosco D, Fajardo-Robledo NS, Perez-Guerrero EE et al (2017) Serum levels of adiponectin and leptin as biomarkers of proteinuria in lupus nephritis. PLoS ONE 12:e0184056
Dini AA, Wang P, Ye DQ (2017) Serum adiponectin levels in patients with systemic lupus erythematosus: a meta-analysis. J Clin Rheumatol 23:361–367
Chougule D, Nadkar M, Venkataraman K, Rajadhyaksha A, Hase N, Jamale T et al (2018) Adipokine interactions promote the pathogenesis of systemic lupus erythematosus. Cytokine 111:20–27
Huang Q, Tao SS, Zhang YJ, Zhang C, Li LJ, Zhao W et al (2015) Serum resistin levels in patients with rheumatoid arthritis and systemic lupus erythematosus: a meta-analysis. Clin Rheumatol 34:1713–1720
Almehed K, d’Elia HF, Bokarewa M, Carlsten H (2008) Role of resistin as a marker of inflammation in systemic lupus erythematosus. Arthritis Res Ther 10:R15
Yan C, Yu L, Zhang XL, Shang JJ, Ren J, Fan J et al (2020) Cytokine profiling in Chinese SLE patients: correlations with renal dysfunction. J Immunol Res 2020:8146502. https://doi.org/10.1155/2020/8146502
Soliman S, Mohan C (2017) Lupus nephritis biomarkers. Clin Immunol 185:10–20
Filkova M, Haluzık M, Gay S, Senolt L (2009) The role of resistin as a regulator of inflammation: implications for various human pathologies. Clin Immunol 133:157–170
Tripathi D, Kant S, Pandey S, Ehtesham NZ (2020) Resistin in metabolism, inflammation, and disease. FEBS J 287:3141–3149
Atawia RT, Bunch KL, Toque HA, Caldwell RB, Caldwell RW (2019) Mechanisms of obesity-induced metabolic and vascular dysfunctions. Front Biosci (Landmark Ed) 24:890–934
Pang SS, Le YY (2006) Role of resistin in inflammation and inflammation-related diseases. Cell Mol Immunol 3:29–34
Acquarone E, Monacelli F, Borghi R, Nencioni A, Odetti P (2019) Resistin: a reappraisal. Mech Ageing Dev 178:46–63
Park HK, Ahima RS (2013) Resistin in rodents and humans. Diabetes Metab J 37:404–414
Vuolteenaho K, Tuure L, Nieminen R, Laasonen L, Leirisalo-Repo M, Moilanen E et al (2021) Pretreatment resistin levels are associated with erosive disease in early rheumatoid arthritis treated with disease-modifying anti-rheumatic drugs and infliximab. Scand J Rheumatol 15:1–6
Santos FMM, Telles RW, Lanna CCD, Teixeira AL, Miranda AS, Rocha NP et al (2017) Adipokines, tumor necrosis factor and its receptors in female patients with systemic lupus erythematosus. Lupus 26:10–16
Fox RI (1993) Mechanism of action of hydroxychloroquine as an antirheumatic drug. Semin Arthritis Rheum 23:82–91
Schrezenmeier E, Dörner T (2020) Mechanisms of action of hydroxychloroquine and chloroquine: implications for rheumatology. Nat Rev Rheumatol 16:155–166
Abdel-Hamid AA, Firgany AE (2016) Favorable outcomes of hydroxychloroquine in insulin resistance may be accomplished by adjustment of the endothelial dysfunction as well as the skewed balance of adipokines. Acta Histochem 118:560–573
Qatanani M, Szwergold NR, Greaves DR, Ahima RS, Lazar MA (2009) Macrophage-derived human resistin exacerbates adipose tissue inflammation and insulin resistance in mice. J Clin Invest 119:531–539
Acknowledgements
We thank Enago (https://www.enago.jp) for editing a draft of this manuscript.
Author information
Authors and Affiliations
Contributions
All authors were involved in drafting the article or revising it critically for important intellectual content, and all authors approved the final version to be submitted for publication. RW and HD planned the study and wrote the manuscript. RW conducted the study and interpreted the results together with KU, SN, HS, TK, NM, MK, TM, KS, MM, RM, and HD. RW and NM conducted statistical analysis of the data obtained in the study. HD and NK reviewed the manuscript for intellectual content. The authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Ethics approval and consent to participate
This study was approved by the ethical committee of Kagawa University (Heisei30-047) and was prospectively registered. All participants gave written informed consent prior to entering the study. The study was conducted in accordance with the Declaration of Helsinki.
Consent for publication
Not applicable.
Disclosures
None.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Author’s comment
This manuscript has been published as a preprint at https://www.researchsquare.com/article/rs-942050/v1.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Wakiya, R., Ueeda, K., Shimada, H. et al. Supplemental hydroxychloroquine therapy regulates adipokines in patients with systemic lupus erythematosus with stable disease. Clin Rheumatol 41, 3345–3353 (2022). https://doi.org/10.1007/s10067-022-06282-0
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10067-022-06282-0