Abstract
Objectives
COVID-19 pandemic has already had a tremendous impact on the process of human society; the survival of mankind and the healthy living environment deterioration with the influence will last for many years. This meta-analysis aims to assess the risk of COVID-19 in patients with rheumatic diseases.
Methods
PubMed, Web of Science, Embase, China National Knowledge Infrastructure (CNKI), and Chinese Biomedical Database (CBM) were systematically searched with no language restriction up to July 5, 2021. The pooled rates were synthesized by fixed effect model or random effect model depending on heterogeneity.
Results
A total of 83 articles were included in this meta-analysis. The incidence of COVID-19 in patient with rheumatic diseases was 0.0190 (95% CI: 0.0136-0.0252), and the hospitalization rate, intensive care unit admission rate, mechanical ventilation rate, and case fatality rate of patients with rheumatic diseases infected with COVID-19 were 0.4396 (95% CI: 0.3899-0.4898), 0.0635 (95% CI: 0.0453-0.0836), 0.0461 (95% CI: 0.0330-0.0609), and 0.0346 (95% CI: 0.0218-0.0493), respectively.
Conclusions
Our research shows that patients with rheumatic diseases have great risk of COVID-19. Differences in COVID-19 incidence, hospitalization rates, and mortality rates in regions were statistically significant. We still need to pay attention to the risk of COVID-19 in patients with rheumatic diseases.
Key Points
• Although the risk of COVID-19 in patients with rheumatic diseases has been discussed in previous meta-analysis, their research directions were inconsistent, and few studies focus on prevalence or serious outcomes of COVID-19 in patient with rheumatic diseases, while the quality of these articles was variable.
• The incidence of COVID-19 and serious clinical outcomes in patients with rheumatic diseases were still high along with differential risks in most regions.
• The use of glucocorticoids and conventional synthetic disease-modifying antirheumatic drugs did not affect the hospitalization rate and mortality in rheumatism patients with COVID-19.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Coronavirus disease 2019 (COVID-19) caused by novel coronavirus has been a destructive worldwide medical crisis. As of August 5, 2021, the global death toll resulted from COVID-19 has reached 4.2 million [1]. The main clinical manifestations of the disease include dry cough, fever, dyspnea, fatigue, and acute lung injury [2,3,4]. Patients with comorbidities like hypertension and lung diseases were associated with increased risk of severe pneumonia, which eventually leads to acute respiratory distress syndrome (ARDS) and respiratory failure [2, 5, 6]. In December, 2020, WHO officially notified the main mutations of COVID-19 since its emergence [7] and confirmed that as the epidemic continues, more mutations will be discovered. At present, four worrying mutant viruses dominate the global epidemic, especially the rapidly spreading “Delta” mutant virus. The Delta strain, was firstly discovered in India, has 15 gene mutations, and partially avoids the neutralizing antibodies in the human body and easily invades human cells. Moreover, after entering the human body, the viral loads of Delta infections were greater compared to infections during initial epidemic wave in early 2020, resulting in faster virus replication ability and stronger infectivity, which is currently the main epidemic strain worldwide [8].
Rheumatic diseases characterized with the pathological invasion of bones, muscles, blood vessels, and related soft tissues or connective tissues, most of which are autoimmune diseases, and the underlying inflammatory reactions could cause various tissues and organ damage, which seriously affect normal physiological function. Common rheumatic diseases, such as rheumatoid arthritis, ankylosing spondylitis, systemic lupus erythematosus, psoriatic arthritis, systemic sclerosis, gout, and Sjogren’s syndrome, were the main cause of physical disability and decline in life quality of these patients and social burden to health care systems. Evidence shows that, in China, the prevalence of rheumatoid arthritis, ankylosing spondylitis, systemic lupus erythematosus, psoriatic arthritis, and gout are approximately 0.28%, 0.29%, 0.03%, 0.27%, and 0.09% [9, 10]. Autoimmune diseases affect 3-5% of the global populations [11, 12], representing tens of millions of patients with impaired immune systems worldwide. Rheumatic diseases have the characteristics of slow onset, long course of disease, and genetic predisposition. Incidence of rheumatoid arthritis has been declined in recent decades [13,14,15]. Compared with the general populations, RDs patients are still facing greater risk of viral and bacterial infection as well as complications [16,17,18]. The possibility that immunosuppression due to biologics and DMARDs drug treatment could increase the sensitivity of COVID-19 infection was the reason for patients with rheumatic disease discontinued treatment these days [19,20,21,22].
It is necessary to explore the risk of COVID-19 among patient with RDs, and we conducted this study to determine the risk of COVID-19 infection and its clinical outcome by integrating the epidemiological data. We analyzed the impact of individual risk factor associated with the prevalence and clinical outcomes of RDs like age, gender, living habit, comorbidities, and medications.
Materials and methods
This research was conducted based on the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) [23] and the Meta-analysis of Observational Studies in Epidemiology (MOOSE) guidelines [24].
Literature search
According to PICOs scheme, the aim of this meta-analysis was to describe the risk of COVID-19 infection, hospitalization, and clinical treatment outcomes (death, intensive care unit admission, mechanical ventilation) in RDs patients.
The electronic databases of PubMed, Web of Science, Embase, Cochrane Library, China National Knowledge Infrastructure (CNKI), and Chinese Biomedical Database (CBM) were systematically searched up to July 5, 2021. The keywords and search strategy were listed as follows: (“rheumatic disease” or “autoimmune disease” or “connective tissue disease” or “musculoskeletal disease” or “ankylosing spondylarthritis” or “rheumatoid arthritis” or “systemic lupus erythematosus” or “vasculitis” or “psoriatic arthritis” or “systemic sclerosis” or “Sjogren’s syndrome”) and “COVID-19” and (“infection” or “prevalence” or “mortality” or “death” or “need to be hospitalized” or “need for ICU/mechanical ventilation”). We also manually searched references of the relevant articles.
Inclusion and exclusion criteria
The commonly used data contained the total number of RDs observation cohort, COVID-19 confirmed cases, or number of hospitalizations or death. Articles with no data available were excluded. The Global Rheumatology Community, which created remarkable achievement, calls on all rheumatology community to conduct timely communication, collect data, and plan and disseminate accurate and comprehensive knowledge to promote rheumatism care in the COVID-19 pandemic. Articles using public data from the COVID-19 Global Rheumatology Alliance [25] were excluded for result comparison and data overlap reduction. Correspondences, letters, comments, and replies were excluded for stable conclusion.
Data extraction
Each of the enrolled article was extracted by two researchers independently for the following information: author’s name, area of research, rheumatic disease clinical diagnosis, demographic characteristics for study population (age, gender, body mass index (BMI), comorbidities), study time, disease duration, and outcome indicators including morbidity, inpatient number, deaths, intensive care unit (ICU) admission, number of persons subjected to mechanical ventilation, and medications. Characteristics about the patient’s age and the duration of the underlying disease are uniformly adjusted by showing in mean with standard deviation (SD) [26, 27]. Some research articles collecting data from multiple countries were grouped into cooperation research. Drugs for patients with rheumatic diseases were divided into the following categories: nonsteroidal anti-inflammatory drugs (NSAIDs), conventional synthetic disease-modifying antirheumatic drugs (csDMARDs), biologic or targeted synthetic DMARDs (b/tsDMARDs), and glucocorticoids (GC).
Methodological quality assessment
The methodological quality assessment of each literature was assessed based on the standardized Joanna Briggs Institute meta-analysis of Statistics Assessment and Review Instrument (JBI MAStARI) critical appraisal tools for descriptive or case series studies. The two reviewers calculated the overall score of each study based on the numbers of answers of “yes,” ranging from 0 to 9. Studies with score among 0 to 3 were categorized as low quality, 4 to 6 as medium quality, and 7 to 9 as high quality. All the discrepancies from the two reviewers about the results of study selection, data extraction, and quality assessment were resolved by discussing with consensus.
Statistical analysis
We calculated the combined rate and 95% confidence interval (CI) to estimate the incidence of COVID-19, hospitalization rate, and mortality in patient with rheumatic diseases by Freeman-Tukey double arcsine transform [28]. The I2 test and Cochran’s Q statistic were used to assess the statistic heterogeneity between studies [29]. When I2 > 50% or P < 0.05, the heterogeneity is considered as statistically significant. If heterogeneity existed, the pooled rate and 95% CI were estimated by random effects model, and, if not, assessed by fixed effect model [30, 31]. The Z test was used to evaluate the statistical significance of the pooled rate, and P < 0.05 was considered significant. In order to explore the potential sources of heterogeneity, subgroup analysis was performed according to study area. Meta-regression was adopted to detect the source of heterogeneity when data about potential variables were provided by more than 10 articles and cumulative meta-analysis based on sample size was used to test the stability of results. All analysis and statistics are done in R.
Result
Study characteristic
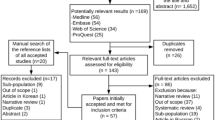
Searched on online databases, Fig. 1 shows the retrieval process of this meta-analysis. Overall, candidate publications were initially screened (PubMed (n = 3133), Web of Science (n = 1093), CNKI database (n = 322), CBM database (n = 50), Embase(n = 503)). After removal of duplicates, a total of 3841 full-test literatures were assessed for eligibility using title and abstract, and then we reviewed the bibliography abstract and full text in necessary. Eventually, 83 eligible studies were enrolled in the current meta-analysis from Asia, Europe, North America, and South America. The main characteristics of each article are presented in Table S1.
Methodological quality assessment, evaluated by JBI MAStARI critical appraisal tools, showed that the methodological qualities of all literatures were considered acceptable (Tables S2 and S3).
Prevalence of COVID-19 in rheumatic diseases
The meta-analysis of 44 observational studies with the number of 181,133 individuals showed that the prevalence of COVID-19 was 0.0190 (95% CI:0.0136-0.0252). Analysis of subgroups according to the region of study conduction showed the prevalence of COVID-19 was ranged from 0.0115 to 0.0499 (Fig. 2). Researches from North America showed the prevalence of COVID-19 in patients with RDs was 0.0499 (95% CI: 0.0246-0.0830). Heterogeneity was significant in overall study (I2 = 98.5%) which probably due to the difference in research scale. No publication bias was detected by Begg’s and Egger’s tests (Fig. S1 Egger’s: P = 0.0635; Begg’s: P = 0.1886). In addition, no significant variation of heterogeneity was detected when each study was omitted (Fig. S2).
Meta-regression was then conducted to explore the source of heterogeneity, data about potential variables were provided by more than 10 articles, and thus the factors tested by the univariate model were methodological quality, age, gender, smoking status, and comorbidities. However, no any significant heterogeneity was detected in meta-regression analysis for each factor (Table 1). It can be considered that age, gender, smoke, and comorbidities did not contribute to the risk of COVID-19 in patients with rheumatic diseases (P > 0.05). We also conducted cumulative meta-analysis according to sample sizes, and a total of 44 studies were included in this analysis. In general, the incidence rate was relatively stable, without big reversal (Fig. S3).
Hospitalization rate of COVID-19 in rheumatic diseases
A meta-analysis based on hospitalization data provided by 55 studies with 16,451 cases of coronavirus disease displayed that the total hospitalization rate was 0.4396 (95% CI: 0.3899-0.4898) (Fig. 2). Analysis of subgroups showed the hospitalization rates of COVID-19 in rheumatic diseases ranged from 0.3293 to 0.4848, with the highest hospitalization rate in Asia. There was considerable heterogeneity in both the overall (I2 = 94.3%) and subgroup analysis. The asymmetric funnel plot suggested there was small study effect or publication bias (Fig. S4 Egger’s: P = 0.0099; Begg’s: P = 0.6162). Sensitivity analysis did not find fluctuations in the stability of the result (Fig. S5).
The meta regression tested by the univariate model found that gender and comorbidities (hypertension, cardiovascular disease, and kidney disease) of confirmed cases were the source of heterogeneity (Table 1). Meta regression analysis showed that higher proportion of male sex, hypertension, and cardiovascular or kidney disease patients (male: regression coefficient: 0.5429, 95% CI: 0.1359-0.9498, P < 0.05; hypertension: regression coefficient: 0.3727, 95% CI: 0.0299-0.7154, P < 0.05; cardiovascular disease: regression coefficient: 0.9316, 95% CI: 0.1618-1.7014, P < 0.05) was associated with higher risk of hospitalization due to COVID-19. In this study, we collected the medication usage of rheumatic disease patient cohort and rheumatic disease patients with COVID-19-confirmed cohort. However, the medication data, classified by drug name in most articles and described without specific situation of single use or combined, cannot be obtained in overlapping and non-interference. Therefore, meta-regression on drug treatment was performed in a small volume. GC and csDMARD were not related to hospitalization rate (P > 0.05). Cumulative meta-analysis performed according to sample size with 55 articles showed stable result (Fig. S6).
Risk of death in rheumatic diseases with COVID-19
Meta-analysis of the mortality rate based on 57 studies showed the mortality rate caused by COVID-19 was 0.0346 (95% CI: 0.0218-0.0493) for rheumatism population (Fig. 2). We observed differences in both the mortality rate caused by coronavirus disease among different regions (P < 0.05). The mortality rates of Asia, Europe, North America, and South America were 0.0000 (95% CI: 0.0000-0.0202), 0.0539 (95% CI: 0.0295-0.0828), 0.0477 (95% CI: 0.0345-0.0627), and 0.0838 (95% CI: 0.0563-0.1161); Begg’s (P = 0.6346) and Egger’s (P = 0.0222) test suggested publication bias, and the funnel plot was not asymmetric of death risk in cohort of rheumatic patients (Fig. S7). In addition, sensitivity analysis was performed to assess the stability and liability by assessing whether the mortality rate was stable when any single study was excluded, and no significant variation of heterogeneity was detected when each study was omitted (Fig. S8).
Overall heterogeneity in above analyses was considerable (I2 = 80.0%) but limited in most subgroups. Meta regression still conducted to analyze sources of heterogeneity in mortality rate The average age of confirmed case comorbidity (cardiovascular disease) was the source of heterogeneity in calculation of mortality in rheumatism cohort (Table 1). Older age (regression coefficient: 0.0045, 95% CI: 0.0007-0.0083, P < 0.05) and a higher proportion of cardiovascular disease (regression coefficient: 0.5579, 95% CI: 0.1999-0.9159, P < 0.05) were associated with higher mortality rate due to COVID-19. Meanwhile, GC and csDMARD were not found to have an impact on the mortality of confirmed patients (P > 0.05). Cumulative meta-analysis showed the mortality rate caused by COVID-19 was stable (Fig. S9).
Clinical measures to patients with COVID-19 and rheumatic diseases
Data collection was also conducted on the number of ICU admission and mechanical ventilation from 33 and 26 articles, respectively. The ICU admission rate and mechanical ventilation rate caused by COVID-19 were 0.0635 (95% CI: 0.0453-0.0836) and 0.0461 (95% CI: 0.0330-0.0609). Patients from Asia and North America had high ICU admission rate and mechanical ventilation rate relatively (Fig. 3). However, the results of subgroup analysis showed no statistically significant difference in mechanical ventilation rate across different areas (P > 0.05). Meta regression showed that no variable contributed to mechanical ventilation rate caused by COVID-19, while methodological quality could be a source of heterogeneity about ICU admission in patients with COVID-19 and rheumatic diseases (Table 2).
Data from WHO and the COVID-19 GRA
Under the premise that the interfering factors such as age, gender, and comorbidities do not match, the case fatality rate of this meta-analysis is 0.0346 (95% CI:0.0218-0.0493) between WHO reported (2.13%) and the COVID-19 GRA reported (5.60%). But the hospitalization rate is higher than European Alliance of Associations for Rheumatology (EULAR) COVID-19 Registry monthly report [32] (28%) and the COVID-19 GRA (30.87%).
Discussion
At present, the meta-analysis has reported the morbidity, hospitalization rate, death risk of total cohort of patients with rheumatic diseases and inpatients, and serious clinical treatment rates (ICU admission rate and mechanical ventilation rate) of patients with RD, 0.0190 (95% CI: 0.0136-0.0252); 0.4396 (95% CI: 0.3899-0.4898); 0.0346 (95% CI: 0.0218-0.0493); 0.1418 (95% CI: 0.1161-0.1690); 0.0635 (95% CI: 0.0453-0.0836); and 0.0461 (95% CI: 0.0330-0.0609), correspondingly.
Since the COVID-19 spread, whether patients with rheumatic diseases have an additional risk of COVID-19-related morbidity or mortality was a concern for many rheumatologists. Fifteen months after the pandemic, the published data consistently showed that the main drivers of poor clinical outcomes were age, male sex and cardiovascular disease, and other comorbidities in general population as well as patients with rheumatic diseases [33].
We found that male sex and comorbidities like hypertension, cardiovascular disease, and kidney disease were associated with an increased risk of hospitalization caused by COVID-19; older age or cardiovascular disease was associated with an increased risk of death. At the same time, the drug use of csDMARD or GC has not been found to be related to the risk of hospitalization or death from COVID-19 in confirmed cases. For other factors potentially affecting COVID-19 prognosis in RD patients, conclusions could not be drawn due to limited data. Our results are not completely consistent with published studies, for example, older age was no longer a significant risk factor for hospitalization, which was similar to the results of Wang Q et al. [34] and Montero et al. [35] but contradictory with some other articles [36,37,38]. Although the regional differences exist, we conclude that the prevalence, hospitalization, and mortality rates caused by COVID-19 in patients with rheumatic diseases have remained at a high level. Xu’s [39] research also reported clinical results of patients with COVID-19 and rheumatic diseases. It suggests high ICU admission rate and case fatality rate, which were 0.09 (95% CI 0.05–0.15) and 0.07 (95% CI 0.03–0.11).
Akiyama [36] published a meta-analysis on the prevalence and clinical outcome of COVID-19 in patients with autoimmune diseases in October 2020. They believed that GC and csDMARDS were risk factors for hospitalization and death for rheumatism patient during the COVID-19 pandemic. It was also analyzed the use status of b/tsDMARDS-csDMARDS combination or b/tsDMARDS monotherapy, which displayed the hospitalization risk and mortality, has changed in the final even if drug dose did not stratify. In particular, anti-tumor necrosis factor (TNFα) therapy has a protective effect, related to the reduction of hospitalization risk and mortality. A scoping review identified that the use of glucocorticoids, JAK inhibitors (especially high-dose), TNF inhibitors (TNFi), and anti-IL-17 agents may be associated with an increased frequency of respiratory viral events [40]. Both csDMARDs and bDMARDs linked with a higher risk of serious infections [41]. These drugs target key molecules involved in the immune response against infectious antigens and therefore may increase susceptibility to viruses and bacteria [42]. It has been confirmed in multiple studies, in which medium-dose and above (> 5mg) glucocorticoids (GC) have increased risk of hospitalization in rheumatic patients who develop COVID-19 [43, 44]. However, studies have also reported no influence from anti-rheumatic drugs on risk of severe COVID-19 outcomes, although the accuracy of data for partial drugs was unstable [45, 46]. In more details, it has been suggested that no substantial risk was detected csDMARDS [42] as we show.
Interpretation of different results should be cautious. In the development of the COVID-19 epidemic in different periods, many changes fed back on changes in clinical data. The emergence and spread of virus variants like Delta, the improvement of prevention measures, the refinement of drug types and dosages in medical research, and the changes in the supply and demand relationship of medical resources may cause contradictions in researches. A unified viewpoint in the past will accept the challenge from new ones. Viral genomes in Europe and America are different from those in Asia, which suggests that the racial difference in the prognosis of patients with COVID-19 may be a reason for study heterogeneity [47, 48]. Our article conducted subgroup analysis based on the area, which showed differentiation in the prevalence and clinical characteristics of COVID-19 in rheumatism group. Hypertension was generally considered to be related to the prognosis of severe COVID-19 [2, 6, 49], and a new study denies it [50]. The incomplete coincidence of conclusions between studies may also be due to the following: the lag and deviation in voluntary data reporting and the lack of detailed information on the countries or continents from which these data are sourced. It is worth noting that medical load explosion at the beginning of the epidemic may cause confirmed cases reported to be smaller than real world data, due to testing measures that are unreachable.
Our study excluded communications, replies, comments, or letters and vigorously avoided data overlap. This research also has limitations. The meta-analysis of observational studies on the prevalence and hospitalization rates of COVID-19 was quite heterogeneous possibly due to differences in the scale of the study, inclusion of different diseases, and study location. Therefore, we conducted a subgroup analysis and performed meta-regression to assess the impact of each potential risk factor on individual outcomes. Meta-regression on drug treatment performed with a small number of articles resulted in analysis which is not stable enough. Most of the cases in the study came from Europe, particularly, the high proportion of articles from Italy. There is no doubt with certain study population crossover. On the other side, there was one report from South America, which showed high hospitalization rate and mortality rate. This is due to unbalances in epidemic trends and reporting efficiency within different regions.
In summary, we believed that there were differences in the risk and clinical outcome of COVID-19 in different regions for rheumatic diseases. Male, older, and underlying comorbidities are risk factors for hospitalization or death. In our analysis on glucocorticoid or csDMARDs use, without categorization of the drugs in their doses, we found no adverse effects on hospitalization and clinical outcomes. Larger studies can explain more risk factors to protect vulnerable people.
Abbreviations
- COVID-19:
-
coronavirus disease 2019
- DMARDs:
-
disease modifying anti-rheumatic drugs
- PsA:
-
psoriatic arthritis
- AS:
-
ankylosing spondylitis
- RA:
-
rheumatoid arthritis
- SLE:
-
systemic lupus erythematosus
- SSc:
-
systemic sclerosis
- SS:
-
Sjogren’s syndrome
- AD:
-
autoimmune diseases
- RDs:
-
rheumatic diseases
- NSAIDs:
-
nonsteroidal anti-inflammatory drugs
- csDMARDs:
-
conventional synthetic disease-modifying antirheumatic drugs
- b/tsDMARDs:
-
biologic or targeted synthetic DMARDs
- GC:
-
glucocorticoids
- TNFα:
-
anti-tumor necrosis factor
- TNFi:
-
TNF inhibitors
- CI:
-
confidence interval
- ICU:
-
intensive care unit
- BMI:
-
body mass index
- PCR:
-
polymerase chain reaction
References
Coronavirus disease (COVID-19) pandemic view dashboard.: World Health Organization. Available at: https://covid19.who.int/. Accessed July 5, 2021
Wu Z, McGoogan JM (2020) Characteristics of and important lessons from the coronavirus disease 2019 (COVID-19) outbreak in China: summary of a report of 72314 cases from the Chinese Center for Disease Control and Prevention %J. JAMA 323(13):1239–1242
Huang C, Wang Y, Li X, Ren L, Zhao J, Hu Y et al (2020) Clinical features of patients infected with 2019 novel coronavirus in Wuhan, China. J Lancet 395(10223):497–506
Carsana L, Sonzogni A, Nasr A, Rossi RS, Pellegrinelli A, Zerbi P et al (2020) Pulmonary post-mortem findings in a series of COVID-19 cases from northern Italy: a two-centre descriptive study %J. Lancet Infect Dis 20(10):1135–1140
Liang W, Guan W, Chen R, Wang W, Li J, Xu K et al (2020) Cancer patients in SARS-CoV-2 infection: a nationwide analysis in China %J. Lancet Oncol 21(3):335–337
Chen N, Zhou M, Dong X, Qu J, Gong F, Han Y et al (2020) Epidemiological and clinical characteristics of 99 cases of 2019 novel coronavirus pneumonia in Wuhan, China: a descriptive study %J. Lancet 395(10223):507–513
Timeline: WHO's COVID-19 response. Available at: https://www.who.int/emergencies/diseases/novel-coronavirus-2019/interactive-timeline#event-231. Accessed August 5, 2021
Li B, Deng A, Li K, Hu Y, Li Z, Xiong Q, et al. (2021) Viral infection and transmission in a large, well-traced outbreak caused by the SARS-CoV-2 Delta variant. medRxiv
Zhao J, Huang C, Huang H, Pan JK, Zeng LF, Luo MH et al (2020) Prevalence of ankylosing spondylitis in a Chinese population: a systematic review and meta-analysis. Rheumatol Int 40(6):859–872
Li R, Sun J, Ren LM, Wang HY, Liu WH, Zhang XW et al (2012) Epidemiology of eight common rheumatic diseases in China: a large-scale cross-sectional survey in Beijing. Rheumatology (Oxford) 51(4):721–729
Eaton WW, Rose NR, Kalaydjian A, Pedersen MG, Mortensen PB (2007) Epidemiology of autoimmune diseases in Denmark %J. J Autoimmun 29(1):1–9
Cooper GS, Stroehla BC (2003) The epidemiology of autoimmune diseases %J. Autoimmun Rev 2(3):119–125
Shichikawa K, Inoue K, Hirota S, Maeda A, Ota H, Kimura M et al (1999) Changes in the incidence and prevalence of rheumatoid arthritis in Kamitonda, Wakayama, Japan, 1965-1996. Ann Rheum Dis 58(12):751–756
Symmons D, Turner G, Webb R, Asten P, Barrett E, Lunt M et al (2002) The prevalence of rheumatoid arthritis in the United Kingdom: new estimates for a new century. Rheumatology (Oxford) 41(7):793–800
Helmick CG, Felson DT, Lawrence RC, Gabriel S, Hirsch R, Kwoh CK, et al (2008) Estimates of the prevalence of arthritis and other rheumatic conditions in the United States. Part I. Arthritis Rheum 58(1):15–25
Listing J, Gerhold K, Zink A (2013) The risk of infections associated with rheumatoid arthritis, with its comorbidity and treatment %J. Rheumatology (Oxford) 52(1):53–61
Di Franco M, Lucchino B, Spaziante M, Iannuccelli C, Valesini G, Iaiani G (2017) Lung Infections in Systemic Rheumatic Disease: Focus on Opportunistic Infections. Int J Mol Sci 18(2):293
Hsu CY, Ko CH, Wang JL, Hsu TC, Lin CY (2019) Comparing the burdens of opportunistic infections among patients with systemic rheumatic diseases: a nationally representative cohort study %J. Arthritis Res Ther 21(1):211
Ciurea A, Papagiannoulis E, Burki K, von Loga I, Micheroli R, Moller B et al (2021) Impact of the COVID-19 pandemic on the disease course of patients with inflammatory rheumatic diseases: results from the Swiss Clinical Quality Management cohort %J. Ann Rheum Dis 80(2):238–241
Roux CH, Brocq O, Gerald F, Pradier C, Bailly L (2020) Impact of home confinement during the COVID-19 pandemic on medication use and disease activity in spondyloarthritis Patients %J. Arthritis Rheum 72(10):1771–1772
Costantino F, Bahier L, Tarancon LC, Leboime A, Vidal F, Bessalah L et al (2021) COVID-19 in French patients with chronic inflammatory rheumatic diseases: clinical features, risk factors and treatment adherence %J. Joint Bone Spine 88(1):105095
Murray K, Quinn S, Turk M, O'Rourke A, Molloy E, O'Neill L et al (2021) COVID-19 and rheumatic musculoskeletal disease patients: infection rates, attitudes and medication adherence in an Irish population %J. Rheumatology (Oxford) 60(2):902–906
Moher D, Liberati A, Tetzlaff J, Altman DG (2009) Preferred reporting items for systematic reviews and meta-analyses: the PRISMA statement %J. BMJ 339:b2535
Stroup DF, Berlin JA, Morton SC, Olkin I, Williamson GD, Rennie D et al (2000) Meta-analysis of observational studies in epidemiology: a proposal for reporting. Meta-analysis Of Observational Studies in Epidemiology (MOOSE) group %J. JAMA 283(15):2008–2012
Data from the COVID-19 Global Rheumatology Alliance Global Registry. The COVID-19 Global Rheumatology Alliance Global Registry; 2021. Available at: https://rheum-covid.org/. Accessed August 5, 2021
Luo D, Wan X, Liu J, Tong T (2018) Optimally estimating the sample mean from the sample size, median, mid-range, and/or mid-quartile range %J. Stat Methods Med Res 27(6):1785–1805
Wan X, Wang W, Liu J, Tong T (2014) Estimating the sample mean and standard deviation from the sample size, median, range and/or interquartile range %J. BMC Med Res Methodol 14:135
Freeman MF, Tukey JW (1950) Transformations related to the angular and the square root. Ann Math Stat 21:607–611
Higgins JP, Thompson SG (2002) Quantifying heterogeneity in a meta-analysis %J. Stat Med 21(11):1539–1558
Higgins JP, Thompson SG, Deeks JJ, Altman DG (2003) Measuring inconsistency in meta-analyses %J. BMJ 327(7414):557–560
Borenstein M, Hedges LV, Higgins JP, Rothstein HR (2010) A basic introduction to fixed-effect and random-effects models for meta-analysis %J. Res Synth Methods 1(2):97–111
EULAR COVID-19 Registry Report (2021). European Alliance of Associations for Rheumatology COVID-19 Registry. Available at: https://www.eular.org/eular_covid_19_registry.cfm. Accessed August 5, 2021
Schreiber K, Hendricks O (2021) First data on COVID-19 morbidity and mortality in patients with rheumatic disease from South Korea. Lancet Rheumatol 3(10):e673–e6e5
Wang Q, Liu J, Shao R, Han X, Su C, Lu W (2021) Risk and clinical outcomes of COVID-19 in patients with rheumatic diseases compared with the general population: a systematic review and meta-analysis. Rheumatol Int 41(5):851–861
Montero F, Martinez-Barrio J, Serrano-Benavente B, Gonzalez T, Rivera J, Molina Collada J et al (2020) Coronavirus disease 2019 (COVID-19) in autoimmune and inflammatory conditions: clinical characteristics of poor outcomes. Rheumatol Int 40(10):1593–1598
Akiyama S, Hamdeh S, Micic D, Sakuraba A (2020) Prevalence and clinical outcomes of COVID-19 in patients with autoimmune diseases: a systematic review and meta-analysis. Ann Rheum Dis 80:384–391
Gamboa-Cardenas RV, Barzola-Cerron S, Toledo-Neira D, Reategui-Sokolova C, Pimentel-Quiroz V, Zevallos-Miranda F et al (2021) Predictors of hospitalization for COVID-19 in patients with autoimmune rheumatic diseases: results from a community cohort follow-up. Clin Rheumatol 40(11):4725–4734
Bakasis AD, Mavragani CP, Boki KA, Tzioufas AG, Vlachoyiannopoulos PG, Stergiou IE et al (2021) COVID-19 infection among autoimmune rheumatic disease patients: data from an observational study and literature review. J Autoimmun 123:102687
Xu C, Yi Z, Cai R, Chen R, Thong BY, Mu R (2021) Clinical outcomes of COVID-19 in patients with rheumatic diseases: a systematic review and meta-analysis of global data. Autoimmun Rev 20(4):102778
Kilian A, Chock YP, Huang IJ, Graef ER, Upton LA, Khilnani A et al (2020) Acute respiratory viral adverse events during use of antirheumatic disease therapies: a scoping review. Semin Arthritis Rheum 50(5):1191–1201
Curtis JR, Xi J, Patkar N, Xie A, Saag KG, Martin C (2007) Drug-specific and time-dependent risks of bacterial infection among patients with rheumatoid arthritis who were exposed to tumor necrosis factor alpha antagonists. Arthritis Rheum 56(12):4226–4227
Ruscitti P, Conforti A, Cipriani P, Giacomelli R, Tasso M, Costa L et al (2021) Pathogenic implications, incidence, and outcomes of COVID-19 in autoimmune inflammatory joint diseases and autoinflammatory disorders. Adv Rheumatol 61(1):45
Hasseli R, Mueller-Ladner U, Hoyer BF, Krause A, Lorenz HM, Pfeil A, et al (2021) Older age, comorbidity, glucocorticoid use and disease activity are risk factors for COVID-19 hospitalisation in patients with inflammatory rheumatic and musculoskeletal diseases. RMD Open 7(1):e001464
Gianfrancesco M, Hyrich KL, Al-Adely S, Carmona L, Danila MI, Gossec L et al (2020) Characteristics associated with hospitalisation for COVID-19 in people with rheumatic disease: data from the COVID-19 Global Rheumatology Alliance physician-reported registry. Ann Rheum Dis 79(7):859–866
Bower H, Frisell T, Di Giuseppe D, Delcoigne B, Ahlenius GM, Baecklund E, et al (2021) Impact of the COVID-19 pandemic on morbidity and mortality in patients with inflammatory joint diseases and in the general population: a nationwide Swedish cohort study. Ann Rheum Dis 80(8):1086–1093
Santos CS, Morales CM, Alvarez ED, Castro CA, Robles AL, Sandoval TP (2020) Determinants of COVID-19 disease severity in patients with underlying rheumatic disease. Clin Rheumatol 39(9):2789–2796
Forster P, Forster L, Renfrew C, Forster M (2020) Phylogenetic network analysis of SARS-CoV-2 genomes. Proc Natl Acad Sci U S A 117(17):9241–9243
Murugan C, Ramamoorthy S, Kuppuswamy G, Murugan RK, Sivalingam Y, Sundaramurthy A (2021) COVID-19: A review of newly formed viral clades, pathophysiology, therapeutic strategies and current vaccination tasks. Int J Biol Macromol 193(Pt B):1165–1200
Nikoloski Z, Alqunaibet AM, Alfawaz RA, Almudarra SS, Herbst CH, El-Saharty S et al (2021) Covid-19 and non-communicable diseases: evidence from a systematic literature review. BMC Public Health 21(1):1068
Zhong L, Wu Y, Gao J, Zhang J, Xie Q, He H et al (2021) Effects of hypertension on the outcomes of COVID-19: a multicentre retrospective cohort study. Ann Med 53(1):770–776
Acknowledgements
We really appreciate the efforts of all the researchers whose articles were included in this study.
Funding
This study was supported by grants from the National Natural Science Foundation of China (81273169, 81573218, 81773514) and the funds for academic and technical leaders in Anhui province (2017D140).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Ethics approval
Not required.
Consent to participate
Not required.
Consent for publication
This article has been carefully prepared, unpublished, and does not consider applying for other journals. All the authors meet the Uniform Requirements for Manuscripts Submitted to Biomedical Journals criteria for authorship and approved the manuscript.
Disclosures
None.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information

Fig. S1
Funnel plots for meta-analysis regarding the prevalence of COVID-19 in patients with rheumatic diseases (PNG 152 kb)

Fig. S2
Sensitivity analysis for prevalence of COVID-19 in patients with rheumatic diseases. (PNG 2050 kb)

Fig. S3
Cumulative meta-analysis regarding the prevalence of COVID-19 in patients with rheumatic diseases (by sample size) (PNG 1600 kb)

Fig. S4
Funnel plots for meta-analysis regarding the hospitalization rate of COVID-19 in patients with rheumatic diseases (PNG 147 kb)

Fig. S5
Sensitivity analysis for hospitalization rate of COVID-19 in patients with rheumatic diseases (PNG 2630 kb)

Fig. S6
Cumulative meta-analysis regarding hospitalization rate of COVID-19 in patients with rheumatic diseases (by sample size) (PNG 3040 kb)

Fig. S7
Funnel plots for meta-analysis regarding mortality rate of COVID-19 in patients with rheumatic diseases (PNG 145 kb)

Fig. S8
Sensitivity analysis for mortality rate of COVID-19 in patients with rheumatic diseases (PNG 2670 kb)

Fig. S9
Cumulative meta-analysis regarding mortality rate of COVID-19 in patients with rheumatic diseases (by sample size) (PNG 2940 kb)
Table S1
Characteristics of studies included in the meta-analysis((a) Epidemiological information; (b) Medication information) (DOCX 267 kb)
Table S2
JBI MAStARI critical appraisal tools (DOCX 16 kb)
Table S3
Methodological quality assessment (DOCX 49 kb)
Rights and permissions
About this article
Cite this article
Wang, F., Ma, Y., Xu, S. et al. Prevalence and risk of COVID-19 in patients with rheumatic diseases: a systematic review and meta-analysis. Clin Rheumatol 41, 2213–2223 (2022). https://doi.org/10.1007/s10067-022-06087-1
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10067-022-06087-1