Abstract
Pulmonary mucormycosis is rare in systemic lupus erythematosus. A 20-year-old lady with lupus nephritis and neuropsychiatric lupus was treated with injection methylprednisolone and cyclophosphamide. After few days, she developed fever, breathlessness, and hoarseness of voice. After neck and chest imaging, possibility of mucormycosis was considered which was later confirmed on microbiological test. Patient was treated with conventional amphotericin B. Literature review was done, and 8 patients with disseminated or pulmonary mucormycosis were identified with SLE. In patients with high index of suspicion, early imaging can help in diagnosis and early and aggressive management even with conventional amphotericin B can result in favorable outcome.
Key Points • Pulmonary mucormycosis in systemic lupus erythematosus is rare. • Radiological investigation can guide towards diagnosis. • Early and aggressive treatment can lead to good outcome. |
Similar content being viewed by others
Introduction
Mucormycosis is an invasive fungal infection caused by the Mucorales order of Zygomycetes, causing high burden of in-hospital mortality [1]. Although these infections are uncommon, increasing number of cases is being recognized in immunocompromised states. The common predisposing factors for mucormycosis include uncontrolled diabetes, high dose glucocorticoids, neutropenia, organ transplant recipients, iron overload states, and recently COVID-19 infection in India [2, 3].
Infections are a major cause of morbidity and mortality in SLE, although the prevalence of invasive fungal infections is low in these patients [4]. Immune dysregulation and the use of immunosuppressive drugs predispose these patients to invasive fungal infections [5]. Data on mucormycosis in SLE is sparse, and there are only 2 cases reported so far of pulmonary mucormycosis (PM) in patients with SLE.
We describe a case with active lupus and PM who was successfully managed with conventional amphotericin B. In addition, we also present the data available so far on mucormycosis in SLE.
Case report
A 20-year-old lady presented with 6 months history of low-grade fever, oral ulcers, alopecia, inflammatory polyarthralgia of small joints of hand, anaemia requiring blood transfusions, and pedal oedema which had recently progressed to anasarca. There was no history of photosensitivity or malar rash. She also had progressive proximal muscle weakness over the past 3 months. Twenty days ago, she had two episodes of generalized tonic–clonic seizures and history of irrelevant talk. Physical examination revealed normal sensorium and orientation, blood pressure of 120/80 mmHg, pallor, anasarca, ulcers over palate and buccal mucosa, diffuse hair thinning, and boggy swelling of bilateral proximal interphalangeal joints. Cardiovascular, respiratory, and abdominal examination was unremarkable. She had grade 2 power in the shoulder and pelvic girdle and grade 3 at the knee and elbows and grade 4 at the wrist and ankle joint. Deep tendon reflexes and sensory system examination were normal.
Laboratory evaluation showed haemoglobin of 9.9 g%, platelet count of 27,000/cu.mm, creatinine 1.1 mg/dl, lactate dehydrogenase 338 U/L, creatinine phosphokinase 27 U/L, Alanine and aspartate aminotransferase 58 U/L, and 20 U/L, respectively. Anti-nuclear antibody (ANA) was positive with homogenous pattern, anti-ds-DNA antibody levels were > 300 IU, and in addition, she had anti-histone, anti-nucleosome, anti-Ro, and anti-La antibodies. Her IgG and IgM anticardiolipin (aCL) antibody titres were 25 GPL (normal: 3–18 GPL) and > 100 MPL (normal: 3–18 MPL), respectively, with negative beta2-glycoprotein antibody. Her complement levels were C3: 12 mg/dl (normal: 60–120 mg/dl), and C4: 2 mg/dl (normal: 15–40 mg/dl). Urinalysis showed proteinuria 3 + with active urine sediments and 24-h urinary protein of 2.7 g. Cerebrospinal fluid examination and MRI head were normal.
A diagnosis of SLE with Lupus nephritis and neuropsychiatric lupus was made. She was treated with daily intravenous pulse of 250 mg methylprednisolone (MPS) for 3 days followed by prednisolone 1 mg/kg body weight. She also received intravenous cyclophosphamide (CYC) induction 750 mg/m2 body surface area (750 mg). With this and supportive care, her anasarca and muscle weakness improved.
A few days later, she developed breathlessness and tachycardia, with presence of coarse crepitations in right infra-scapular and infra-axillary regions. ECG showed sinus tachycardia. Chest X-ray showed new onset consolidation in right lower zone, following which antibiotics (intravenous piperacillin-tazobactam and oral azithromycin) were started and prednisolone was tapered to 0.25 mg/kg. She had no fever, cough, or expectoration. A day later, she has sudden onset of hoarseness of voice and video-laryngoscopy showed evidence of subglottic narrowing.
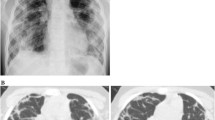
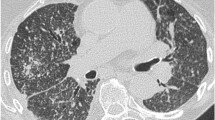
Contrast-enhanced computed tomography (CECT) scan of neck and chest revealed mass-like consolidation in the right lower lobe with central ground glass opacities intermingled with small cavities and intersecting reticular strands suggestive of Bird’s nest sign. Multiple variable-sized random nodules were seen scattered in bilateral upper lobes (right more than left), with many of them showing central cavitation and peripheral ground glass halo (Fig. 1a). No obvious bronchial wall thickening was seen. Based on these imaging findings in a background of ongoing immunosuppression, a diagnosis of invasive fungal infection (IFI) was made, mucormycosis being the most likely aetiology. The rest of the upper airway showed no abnormality. The paranasal sinuses were normal.
CECT Chest. a CECT chest at presentation. Axial section of upper chest reveals multiple nodules with central cavitation in right upper lobe (white arrowhead). Lung window and mediastinal window images at mid and lower levels reveal Bird’s nest sign (yellow arrowheads) suggestive of invasive fungal infection. b. Post treatment HRCT chest. Axial sections of right lung at upper, mid and lower levels reveal reduction in size and number of cavitary lesions in right upper lobe (white arrowhead) and resolution of the mass-like consolidation in the right lower lobe with a residual thin-walled cavity (yellow arrowheads)
With subglottic stenosis and multiple cavitary lesions in the lung, a possibility of coexisting small vessel vasculitis was also considered; however, ANCAs were negative. Hoarseness of voice subsided within 2 days of its onset with ongoing prednisolone therapy. Throat and nasal swab for COVID-19 RTPCR was negative. Laboratory assessment showed no evidence of sepsis. Bronchoalveolar lavage aspirate (BAL) on KOH wet mount preparation showed plenty of broad aseptate right angled branching fungal hyphae classical of Mucorales fungi (Fig. 2a). This was confirmed on fungal culture with a confluent growth of Rhizopus microsporus. Serological assay for Aspergillus, galactomannan, β-D-glucan, and quantitative PCR in serum or tissue sample was not done. There was no evidence of neutropenia or elevated blood sugar during the hospital stay.
With the diagnosis of PM, she was treated with conventional amphotericin B (AmB) at 1 mg/kg body weight every 24 h with monitoring of renal functions and electrolytes. The serial HRCT chest (on day 15 of amphotericin B dose and subsequently after completing 1 g of cumulative amphotericin B) showed resolution of the mass-like consolidation in the right lower lobe and a residual thin-walled cavity was seen. The satellite lesions in upper lobes significantly reduced in size and number (Fig. 1b). She remained afebrile with no breathlessness and improvement in chest findings. On discharge, she was given oral posaconazole and low dose oral prednisolone with hydroxychloroquine. Patient continued to have proteinuria without active urine sediments on follow-up. In view of bland proteinuria and recent history of PM, she was treated with angiotensin receptor blockers and tacrolimus. Presently, she is asymptomatic with only minimal residual muscle weakness in proximal muscles.
Methods
A literature review was undertaken in MEDLINE and Google scholar. Any cases of mucormycosis in SLE were sought in MEDLINE using the search string {(“mucor”[MeSH Terms] OR “mucor”[All fields] OR “mucormycosis”[MeSH Terms] OR “mucormycosis”[All fields] OR “zygomycetes”[All fields] OR “rhizopus”[MeSH Terms] or “rhizopus”[All fields]) AND (“lupus” OR “SLE”)}. A total of 46 articles were retrieved in MEDLINE and 11 is Google scholar. Articles without a full text or those not in English were excluded. A total of 8 cases of disseminated mucormycosis were identified with available clinical details. In addition, 1 case each of disseminated mucormycosis was reported as treatment-related adverse event in a trial of myeloablative stem cell transplant in SLE by Burt RK [6] and an autopsy series of SLE by Jindal B [7].
Table 1 summarizes disseminated and pulmonary mucormycosis cases reported in literature [8,9,10,11,12,13,14,15].
Discussion
The present case highlights the importance of early diagnosis using radiology and BAL in PM as that can result in favourable outcome. Though infections are an important cause of hospitalization in patients with SLE, invasive fungal infections (IFI) are uncommon in SLE. However, IFI do occur due to use of immunosuppressive therapy and have an in-hospital mortality of more than 50% [16]. In a retrospective study from South China, of 45 patients of SLE with IFI, Aspergillus (44.4%) and Cryptococcus (51.1%) were the commonly identified species. Patients with active lupus nephritis developed more IFI with higher mortality rates being noted (20%) [4]. Another study from Taiwan compared a large cohort of SLE (24,541 patients) with age-matched non-SLE diseased controls (245,410 subjects); incidence rate of IFI among patient of lupus was 20.83 per 10,000 person-years compared to 1.88 per 10,000 person-year in non-SLE diseased controls [17].
In our case, an HRCT done early in the disease course was suggestive of mass-like consolidation with central ground glass opacity giving a ‘reverse halo sign’. Also, the central low attenuation areas showed intersecting reticular lines and small cavities typical of ‘Bird’s nest sign’. Multiple randomly distributed macronodules with peripheral ‘halo sign’ and central cavitation were also seen. These findings are typically seen in invasive fungal infections. Among the common invasive fungal infections encountered in a background of immunosuppression, PM and invasive pulmonary Aspergillosis (IPA) can show these findings; however, the presence of Bird’s nest sign, reverse halo sign, absence of bronchial wall thickening, and random rather than peri-bronchial distribution of nodules were findings which favoured a diagnosis of mucormycosis more than Aspergillosis [18]. A definite diagnosis of PM was established by BAL which on microbiological assessment revealed broad aseptate right angled branching hyphae of mucormycetes [19].
The reversed halo sign on imaging is not specific for fungal infections but in severely immunocompromised patients, this sign is suggestive of invasive fungal infection; halo sign has been reported in 19–94% of patients with PM [20, 21]. Other common imaging findings in PM include areas of wedge-shaped or mass-like consolidations and nodules with cavitation. Consolidations crossing the fissures, invasion of the chest wall, and pulmonary arteries are more common with PM than IPA. Pulmonary artery pseudoaneurysms may also be seen in PM [22]. Though radiological signs are not definitive for the diagnosis, guidelines for screening of fungal infection in immunosuppressed patients recommend early HRCT in those with a clinical suspicion [23]. Even when radiology is suggestive, a definitive mycological diagnosis should be made as mucormycosis is susceptible to only AmB, posaconazole, and isavuconazole [24].
Early diagnosis and initiation of treatment are important. Correctable risk factors, mainly the control of blood glucose, tapering dose of steroids, and discontinuation of other immunosuppressive drugs should be addressed. Although conventional AmB has been the cornerstone of treatment for Mucorales [25], but over a decade, lipid formulation of AmB is recommended due to better tissue penetration and less nephrotoxicity [26]. Our patient received conventional AmB due to affordability issues and responded well to treatment; therefore, in a resource poor country, considering early treatment with conventional AmB can bring favourable outcomes. Treatment is recommended till the near normalization of radiographic abnormalities and negativity of cultures [27]. Since the patients with SLE require immunosuppressive agents for their disease to be in remission, long-term therapy is indicated in these patients with posaconazole having a potent activity against Mucorales and being available in oral preparation [28]. Using adjunctive treatment like interferon gamma and/or granulocyte macrophage colony stimulating factor, hyperbaric oxygen requires more data [29].
Conclusion
Pulmonary mucormycosis is well described in patients with uncontrolled diabetes, neutropenia, and organ-transplant recipients but it is not well reported in SLE. In patients with high index of suspicion, early imaging can guide in initial diagnosis followed by confirmation with culture or histopathology. Early and aggressive treatment even with conventional AmB can lead to good outcome.
Data availability
Data can be made available at reasonable request to the corresponding author.
Abbreviations
- AmB:
-
Amphotericin B
- aCL:
-
Anticardiolipin
- ANA:
-
Anti-nuclear antibody
- BAL:
-
Bronchoalveolar lavage aspirate
- CECT:
-
Contrast enhanced computed tomography
- CYC:
-
Cyclophosphamide
- IFI:
-
Invasive fungal infections
- IPA:
-
Invasive pulmonary aspergillosis
- MPS:
-
Methylprednisolone
- PM:
-
Pulmonary mucormycosis
- SLE:
-
Systemic lupus erythematosus
References
Prakash H, Chakrabarti A (2019) Global epidemiology of mucormycosis. J Fungi (Basel) 5(1):26
Jeong W, Keighley C, Wolfe R, Lee WL, Slavin MA, Kong DCM et al (2019) The epidemiology and clinical manifestations of mucormycosis: a systematic review and meta-analysis of case reports. Clin Microbiol Infect 25(1):26–34
Singh AK, Singh R, Joshi SR, Misra A (2021) Mucormycosis in COVID-19: a systematic review of cases reported worldwide and in India. Diabetes Metab Syndr. 15(4):102146
Lao M, Wang X, Ding M, Yang Z, Chen H, Liang L et al (2019) Invasive fungal disease in patients with systemic lupus erythematosus from Southern China: a retrospective study. Lupus 28(1):77–85
Jung JY, Yoon D, Choi Y, Kim HA, Suh CH (2019) Associated clinical factors for serious infections in patients with systemic lupus erythematosus. Sci Rep 9(1):9704
Burt RK, Traynor A, Statkute L, Barr WG, Rosa R, Schroeder J et al (2006) Nonmyeloablative hematopoietic stem cell transplantation for systemic lupus erythematosus. JAMA 295(5):527–535
Jindal B, Joshi K, Radotra BD, Banerjee AK (2000) Fatal complications of systemic lupus erythematosus–an autopsy study from north India. Indian J Pathol Microbiol 43(3):311–317
Wong KL, Tai YT, Loke SL, Woo EK, Wong WS, Chan MK et al (1986) Disseminated zygomycosis masquerading as cerebral lupus erythematosus. Am J Clin Pathol 86(4):546–549
Bloxham CA, Carr S, Ryan DW, Kesteven PJ, Bexton RS, Griffiths ID et al (1990) Disseminated zygomycosis and systemic lupus erythematosus. Intensive Care Med 16(3):201–207
Fingerote RJ, Seigel S, Atkinson MH, Lewkonia RM (1990) Disseminated zygomycosis associated with systemic lupus erythematosus. J Rheumatol 17(12):1692–1694
Coffey MJ, Fantone J, Stirling MC, Lynch JP (1992) Pseudoaneurysm of pulmonary artery in mucormycosis. Radiographic characteristics and management. Am Rev Respir Dis 145(6):1487–90
Liu MF, Chen FF, Hsiue TR, Liu CC (2000) Disseminated zygomycosis simulating cerebrovascular disease and pulmonary alveolar haemorrhage in a patient with systemic lupus erythematosus. Clin Rheumatol 19(4):311–314
Hung HC, Shen GY, Chen SC, Yeo KJ, Tsao SM, Lee MC et al (2015) Pulmonary mucormycosis in a patient with systemic lupus erythematosus: a diagnostic and treatment challenge. Case Rep Infect Dis. 2015:478789
de Clerck F, Van Ryckeghem F, Depuydt P, Benoit D, Druwé P, Hugel A et al (2014) Dual disseminated infection with Nocardia farcinica and Mucor in a patient with systemic lupus erythematosus: a case report. J Med Case Rep 20(8):376
Rangwala SD, Strickland BA, Rennert RC, Ravina K, Bakhsheshian J, Hurth K et al (2019) Ruptured mycotic aneurysm of the distal circulation in a patient with mucormycosis without direct skull base extension: case report. Oper Neurosurg (Hagerstown) 16(3):E101–E107
Martínez-Martínez MU, Herrera-Van Oostdam D, Román-Acosta S, Magaña-Aquino M, Baranda-Cándido L, Abud-Mendoza C (2012) Invasive fungal infections in patients with systemic lupus erythematosus. J Rheumatol 39(9):1814–1818
Su CF, Lai CC, LI TH, Chang YF, Lin YT, Tsai CY et al (2020) Risk of invasive fungal infection in systemic lupus erythematosus: a nationwide population based study in Taiwan. Ann Rheum Dis. 79(Suppl 1):876
Jung J, Kim MY, Lee HJ, Park YS, Lee SO, Choi SH et al (2015) Comparison of computed tomographic findings in pulmonary mucormycosis and invasive pulmonary aspergillosis. Clin Microbiol Infect 21(7):684.e11–8
Peng M, Meng H, Sun Y, Xiao Y, Zhang H, Lv K et al (2019) Clinical features of pulmonary mucormycosis in patients with different immune status. J Thorac Dis 11(12):5042–5052
Reid G, Lynch JP 3rd, Fishbein MC, Clark NM (2020) Mucormycosis. Semin Respir Crit Care Med 41(1):99–114
Agrawal R, Yeldandi A, Savas H, Parekh ND, Lombardi PJ, Hart EM. Pulmonary Mucormycosis: Risk Factors, Radiologic Findings, and Pathologic Correlation. Radiographics. 2020 May-Jun;40(3):656–666.
Hamilos G, Samonis G, Kontoyiannis DP (2011) Pulmonary mucormycosis. Semin Respir Crit Care Med 32(6):693–702
Ruhnke M, Behre G, Buchheidt D, Christopeit M, Hamprecht A, Heinz W et al (2018) Diagnosis of invasive fungal diseases in haematology and oncology: 2018 update of the recommendations of the infectious diseases working party of the German society for hematology and medical oncology (AGIHO). Mycoses 61(11):796–813
Skiada A, Lanternier F, Groll AH, Pagano L, Zimmerli S, Herbrecht R et al (2013) European Conference on Infections in Leukemia. Diagnosis and treatment of mucormycosis in patients with hematological malignancies: guidelines from the 3rd European Conference on Infections in Leukemia (ECIL 3). Haematologica 98(4):492–504
Spellberg B, Ibrahim AS (2010) Recent advances in the treatment of mucormycosis. Curr Infect Dis Rep 12(6):423–429
Stone NR, Bicanic T, Salim R, Hope W (2016) Liposomal amphotericin B (AmBisome(®)): a review of the pharmacokinetics, pharmacodynamics, clinical experience and future directions. Drugs 76(4):485–500
Fernandez JF, Maselli DJ, Simpson T, Restrepo MI (2013) Pulmonary mucormycosis: what is the best strategy for therapy? Respir Care 58(5):e60–e63
Sipsas NV, Gamaletsou MN, Anastasopoulou A, Kontoyiannis DP (2018) Therapy of mucormycosis. J Fungi (Basel) 4(3):90
Brunet K, Rammaert B. Mucormycosis treatment: recommendations, latest advances, and perspectives. J Mycol Med. 2020 Sep;30(3):101007.
Author information
Authors and Affiliations
Contributions
All authors were involved in case management. Ideation of manuscript was done by AA; drafting of manuscript was done by RK, RC, and AA. Critical revisions were done by AA and RC. Final approval of version was done by all the authors. Radiological diagnosis was done by AM and microbiological confirmation was done by RM.
Corresponding author
Ethics declarations
Ethics approval
Not required.
Consent to participate
Written consent obtained from the patient.
Consent to publish
Written consent obtained from the patient.
Disclosures
None.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Kharbanda, R., Mehndiratta, A., Chatterjee, R. et al. Pulmonary mucormycosis in systemic lupus erythematosus: successful management of a case along with review of literature. Clin Rheumatol 41, 307–312 (2022). https://doi.org/10.1007/s10067-021-05941-y
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10067-021-05941-y