Abstract
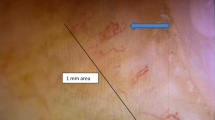
Schnitzler syndrome (SchS) is a rare acquired systemic autoinflammatory disease. The major clinical features of SchS are urticarial rash and monoclonal gammopathy, accompanied by fever, joint pain, and lymphadenopathy. There were few reports about SchS in Chinese population. Herein, we describe two patients with SchS in China and conducted a systematic literature review about SchS. Two Chinese Han patients were diagnosed as SchS in our department from 2017 to 2019. Their phenotype and genotype were carefully documented and studied. We also conducted a systematic literature review about SchS. There was one man (66 years old) and one woman (49 years old). Recurrent fever and urticarial rash occurred in both of them during the febrile attacks and normalized in asymptomatic intervals. Other manifestations included arthralgia, lymphadenopathy, and hearing loss. Hepatic cirrhosis and epilepsy were seen in the male patient. None of them had bone pain or family histories. Serum monoclonal IgM gammopathy was found in both patients. MyD88 gene mutation L258P was identified in the female patient. They were treated with tocilizumab and tripterygium wilfordii Hook F (TwHF) respectively, and both showed good response. The rarity and diversity of SchS make it difficult to be recognized. Anti-IL-6 agents may be alternative therapies when anti-IL-1 therapy is unresponsive or unavailable. Due to the case report, the effect of TwHF in the treatment of SchS should be further studied.

Similar content being viewed by others
Availability of data and materials
All data generated or analyzed during this study are included in this published article.
References
Barriere H, Schnitzler L, Moulin G, Grolleau Y (1976) A propos de cinq observations [chronic urticarial lesions and macroglobulinemia]. Apropos of 5 cases. Sem Hop 52(4):221–227
Sokumbi O, Drage LA, Peters MS (2012) Clinical and histopathologic review of Schnitzler syndrome: the Mayo Clinic experience (1972-2011). J Am Acad Dermatol 67(6):1289–1295. https://doi.org/10.1016/j.jaad.2012.04.027
Vanderschueren S, van der Veen A (2017) The Schnitzler syndrome: chronic urticaria in disguise: a single-centre report of 11 cases and a critical reappraisal of the literature. Clin Exp Rheumatol 35(1):69–73
de Koning HD (2014) Schnitzler's syndrome: lessons from 281 cases. Clin Transl Allergy 4:41. https://doi.org/10.1186/2045-7022-4-41
Simon A, Asli B, Braun-Falco M, De Koning H, Fermand JP, Grattan C et al (2013) Schnitzler's syndrome: diagnosis, treatment, and follow-up. Allergy. 68(5):562–568. https://doi.org/10.1111/all.12129
Gusdorf L, Asli B, Barbarot S, Neel A, Masseau A, Puechal X et al (2017) Schnitzler syndrome: validation and applicability of diagnostic criteria in real-life patients. Allergy. 72(2):177–182. https://doi.org/10.1111/all.13035
Rowczenio DM, Pathak S, Arostegui JI, Mensa-Vilaro A, Omoyinmi E, Brogan P, Lipsker D, Scambler T, Owen R, Trojer H, Baginska A, Gillmore JD, Wechalekar AD, Lane T, Williams R, Youngstein T, Hawkins PN, Savic S, Lachmann HJ (2018) Molecular genetic investigation, clinical features, and response to treatment in 21 patients with Schnitzler syndrome. Blood. 131(9):974–981. https://doi.org/10.1182/blood-2017-10-810366
Pathak S, Rowczenio DM, Owen RG, Doody GM, Newton DJ, Taylor C, Taylor J, Cargo C, Hawkins PN, Krause K, Lachmann HJ, Savic S (2019) Exploratory study of MYD88 L265P, rare NLRP3 variants, and clonal hematopoiesis prevalence in patients with Schnitzler syndrome. Arthritis Rheumatol 71(12):2121–2125. https://doi.org/10.1002/art.41030
de Koning HD, van Gijn ME, Stoffels M, Jongekrijg J, Zeeuwen PL, Elferink MG et al (2015) Myeloid lineage-restricted somatic mosaicism of NLRP3 mutations in patients with variant Schnitzler syndrome. J Allergy Clin Immunol 135(2):561–564. https://doi.org/10.1016/j.jaci.2014.07.050
Treon SP, Xu L, Yang G, Zhou Y, Liu X, Cao Y, Sheehy P, Manning RJ, Patterson CJ, Tripsas C, Arcaini L, Pinkus GS, Rodig SJ, Sohani AR, Harris NL, Laramie JM, Skifter DA, Lincoln SE, Hunter ZR (2012) MYD88 L265P somatic mutation in Waldenstrom's macroglobulinemia. N Engl J Med 367(9):826–833. https://doi.org/10.1056/NEJMoa1200710
Bashir M, Bettendorf B, Hariman R (2018) A rare but fascinating disorder: case collection of patients with Schnitzler syndrome. Case Rep Rheumatol 2018:7041576–7041574. https://doi.org/10.1155/2018/7041576
Lauwers A, Chouvy V, Mosnier JF, Misery L, Alexandre C (1999) A case of Schnitzler's syndrome with nodular regenerative hyperplasia of the liver. Rev Rhum Engl Ed 66(5):281–283
Czaja AJ, Carpenter HA (2004) Progressive fibrosis during corticosteroid therapy of autoimmune hepatitis. Hepatology. 39(6):1631–1638. https://doi.org/10.1002/hep.20235
Friedman SL (2008) Mechanisms of hepatic fibrogenesis. Gastroenterology. 134(6):1655–1669. https://doi.org/10.1053/j.gastro.2008.03.003
Gieling RG, Wallace K, Han YP (2009) Interleukin-1 participates in the progression from liver injury to fibrosis. Am J Physiol Gastrointest Liver Physiol 296(6):G1324–G1331. https://doi.org/10.1152/ajpgi.90564.2008
Meier RPH, Meyer J, Montanari E, Lacotte S, Balaphas A, Muller YD et al (2019) Interleukin-1 receptor antagonist modulates liver inflammation and fibrosis in mice in a model-dependent manner. Int J Mol Sci 20(6). https://doi.org/10.3390/ijms20061295
Choi J, Koh S (2008) Role of brain inflammation in epileptogenesis. Yonsei Med J 49(1):1–18. https://doi.org/10.3349/ymj.2008.49.1.1
DeSena AD, Do T, Schulert GS (2018) Systemic autoinflammation with intractable epilepsy managed with interleukin-1 blockade. J Neuroinflammation 15(1):38. https://doi.org/10.1186/s12974-018-1063-2
Schneider SW, Gaubitz M, Luger TA, Bonsmann G (2007) Prompt response of refractory Schnitzler syndrome to treatment with anakinra. J Am Acad Dermatol 56(5 Suppl):S120–S122. https://doi.org/10.1016/j.jaad.2006.05.057
Martinez-Taboada VM, Fontalba A, Blanco R, Fernandez-Luna JL (2005) Successful treatment of refractory Schnitzler syndrome with anakinra: comment on the article by Hawkins et al. Arthritis Rheum 52(7):2226–2227. https://doi.org/10.1002/art.21101
de Koning HD, Bodar EJ, van der Meer JW, Simon A (2007) Schnitzler syndrome study G. Schnitzler syndrome: beyond the case reports: review and follow-up of 94 patients with an emphasis on prognosis and treatment. Semin Arthritis Rheum 37(3):137–148. https://doi.org/10.1016/j.semarthrit.2007.04.001
de Koning HD, Schalkwijk J, van der Ven-Jongekrijg J, Stoffels M, van der Meer JW, Simon A (2013) Sustained efficacy of the monoclonal anti-interleukin-1 beta antibody canakinumab in a 9-month trial in Schnitzler's syndrome. Ann Rheum Dis 72(10):1634–1638. https://doi.org/10.1136/annrheumdis-2012-202192
Pesek R, Fox R (2014) Successful treatment of Schnitzler syndrome with canakinumab. Cutis. 94(3):E11–E12
Krause K, Tsianakas A, Wagner N, Fischer J, Weller K, Metz M, Church MK, Maurer M (2017) Efficacy and safety of canakinumab in Schnitzler syndrome: a multicenter randomized placebo-controlled study. J Allergy Clin Immunol 139(4):1311–1320. https://doi.org/10.1016/j.jaci.2016.07.041
Krause K, Feist E, Fiene M, Kallinich T, Maurer M (2012) Complete remission in 3 of 3 anti-IL-6-treated patients with Schnitzler syndrome. J Allergy Clin Immunol 129(3):848–850. https://doi.org/10.1016/j.jaci.2011.10.031
Claus J, Vanderschueren S (2019) Variable responses to tocilizumab in four patients with Schnitzler syndrome. J Clin Immunol 39(4):370–372. https://doi.org/10.1007/s10875-019-00644-1
Lv QW, Zhang W, Shi Q, Zheng WJ, Li X, Chen H, Wu QJ, Jiang WL, Li HB, Gong L, Wei W, Liu H, Liu AJ, Jin HT, Wang JX, Liu XM, Li ZB, Liu B, Shen M, Wang Q, Wu XN, Liang D, Yin YF, Fei YY, Su JM, Zhao LD, Jiang Y, Li J, Tang FL, Zhang FC, Lipsky PE, Zhang X (2015) Comparison of Tripterygium wilfordii Hook F with methotrexate in the treatment of active rheumatoid arthritis (TRIFRA): a randomised, controlled clinical trial. Ann Rheum Dis 74(6):1078–1086. https://doi.org/10.1136/annrheumdis-2013-204807
Chen BJ (2001) Triptolide, a novel immunosuppressive and anti-inflammatory agent purified from a Chinese herb Tripterygium wilfordii Hook F. Leuk Lymphoma 42(3):253–265. https://doi.org/10.3109/10428190109064582
Yu Y, Koehn CD, Yue Y, Li S, Thiele GM, Hearth-Holmes MP, Mikuls TR, O’Dell JR, Klassen LW, Zhang Z, Su K (2015) Celastrol inhibits inflammatory stimuli-induced neutrophil extracellular trap formation. Curr Mol Med 15(4):401–410. https://doi.org/10.2174/1566524015666150505160743
Liu L, Zhao H, Sun X, Zheng Q, Luo Y, Ru Y, Zhang Y, Chen X, Zhu B, Yin C, Li B, Li X (2018) Efficacy and safety of Tripterygium wilfordii hook F for chronic urticaria: a systematic review and meta-analysis. BMC Complement Altern Med 18(1):243. https://doi.org/10.1186/s12906-018-2305-7
Li R, Lu K, Wang Y, Chen M, Zhang F, Shen H, Yao D, Gong K, Zhang Z (2017) Triptolide attenuates pressure overload-induced myocardial remodeling in mice via the inhibition of NLRP3 inflammasome expression. Biochem Biophys Res Commun 485(1):69–75. https://doi.org/10.1016/j.bbrc.2017.02.021
Fangxiao M, Yifan K, Jihong Z, Yan S, Yingchao L (2019) Effect of Tripterygium wilfordii polycoride on the NOXs-ROS-NLRP3 inflammasome signaling pathway in mice with ulcerative colitis. Evid Based Complement Alternat Med 2019:9306283–9306287. https://doi.org/10.1155/2019/9306283
Pan XC, Liu Y, Cen YY, Xiong YL, Li JM, Ding YY et al (2019) Dual role of triptolide in interrupting the NLRP3 inflammasome pathway to attenuate cardiac fibrosis. Int J Mol Sci 20(2). https://doi.org/10.3390/ijms20020360
Acknowledgments
We appreciate all supports from the patients and their families.
Funding
This work was supported by the Natural Science Foundation of Beijing (grant number 7192170), the Chinese Academy of Medical Sciences (CAMS) Innovation Fund for Medical Sciences (CIFMS) (grant number 2017-I2M-3-001), and the National Key Research and Development Program of China (grant numbers 2016YFC0901500; 2016YFC0901501).
Author information
Authors and Affiliations
Contributions
Ruyu Yan, Wei Cao, Xinchao Liu, Feng Li, and Min Shen were treating physicians for the patients and had contributed suggestions for the diagnosis and management of the patients. Ruyu Yan drafted the manuscript. Min Shen designed the study and revised the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Disclosures
None.
Ethics approval
Ethical approval for this study was approved by the Institutional Review Board of Peking Union Medical College Hospital and performed according to the Declaration of Helsinki.
Consent to participate
Informed consents were obtained from both participants.
Consent for publication
Written informed consent for publication was obtained from all authors.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Yan, R., Cao, W., Liu, X. et al. A Chinese case series of Schnitzler syndrome and complete remission in one tocilizumab-treated patient. Clin Rheumatol 39, 3847–3852 (2020). https://doi.org/10.1007/s10067-020-05204-2
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10067-020-05204-2