Abstract
Background
Rheumatoid arthritis (RA) is an inflammatory disease that leads to altered body composition. The loss of lean mass with a preservation or increase in fat mass has been termed rheumatoid cachexia (RC), to contrast with classic cachexia, which is characterized by severe weight loss. There are limited data on the prevalence and progression of cachexia in RA over time, as well as on associated factors. Our aim was to determine the prevalence of cachexia and to determine associations with potential factors.
Methods
This prospective cohort study recruited consecutively patients diagnosed with RA and followed for 1 year. The assessments were performed: clinical features, body composition, and physical function. RC and classic cachexia were assessed by several established diagnostic criteria. The pairwise Student’s t test, Chi-square test, and GEE were performed (accepted at p ≤ 0.05).
Results
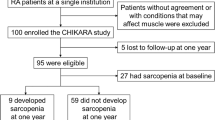
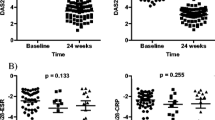
Of 90 patients recruited, 81 completed the study. Most patients were women (88.9%), and the mean age was 56.5 ± 7.3 years. At baseline, the median DAS28-CRP was 3.0 (IQR, 1.0–3.0), 13.3–30.0% of the included patients had RC, while none met criteria for classic cachexia. The prevalence of cachexia did not change after 12 months. Disease activity status and treatment with biologic disease-modifying antirheumatic drugs were significantly associated with changes on body composition and physical function (p < 0.05).
Conclusions
In this cohort, RC was common, while classic cachexia was absent. Disease activity and use of biologic therapies were associated with changes on body composition and physical function, underscoring the importance of aiming for remission when treating RA.
Key Points • Rheumatoid cachexia is common in RA patients. • Classical cachexia is not often in RA patients. • Disease activity and use of biologic therapies are associated with changes on body composition and physical function. |




Similar content being viewed by others
References
Smolen JS, Aletaha D, Barton A, Burmester GR, Emery P, Firestein GS et al (2018) Rheumatoid arthritis. Nat Rev Dis Prim 8(4):18001. https://doi.org/10.1038/nrdp.2018.1
Lee DM, Weinblatt ME (2001) Rheumatoid arthritis. Lancet. 358:903–911
Myasoedova E, Crowson CS, Kremers HM, Therneau TM, Gabriel SE (2010) Is the incidence of rheumatoid arthritis rising? Results from Olmsted County, Minnesota, 1955-2007. Arthritis Rheum 62:1576–1582. https://doi.org/10.1002/art.27425
Tobón GJ, Youinou P, Saraux A (2010) The environment, geo-epidemiology, and autoimmune disease: rheumatoid arthritis. J Autoimmun 35(1):10–14. https://doi.org/10.1016/j.jaut.2009.12.009
Cross M, Smith E, Hoy D, Carmona L, Wolfe F, Vos T, Williams B, Gabriel S, Lassere M, Johns N, Buchbinder R, Woolf A, March L (2014) The global burden of rheumatoid arthritis: estimates from the Global Burden of Disease 2010 study. Ann Rheum Dis 73:1316–1322. https://doi.org/10.1136/annrheumdis-2013-204627
Aslam F, Khan NA (2018) Tools for the assessment of comorbidity burden in rheumatoid arthritis. Front Med (Lausanne). 16;5:39. doi: https://doi.org/10.3389/fmed.2018.00039
Norton S, Koduri G, Nikiphorou E, Dixey J, Williams P, Young A (2013) A study of baseline prevalence and cumulative incidence of comorbidity and extra-articular manifestations in RA and their impact on outcome. Rheumatology (Oxford) 52:99–110. https://doi.org/10.1093/rheumatology/kes262
Roubenoff R, Roubenoff RA, Cannon JG, Kehayias JJ, Zhuang H, Dawson-Hughes B, Dinarello CA, Rosenberg IH (1994) Rheumatoid cachexia: cytokine-driven hypermetabolism accompanying reduced body cell mass in chronic inflammation. J Clin Invest 93:2379–2386. https://doi.org/10.1172/JCI117244
Rall LC, Roubenoff R (2004) Rheumatoid cachexia: metabolic abnormalities, mechanisms and interventions. Rheumatology. 43:1219–1223
Walsmith J, Abad L, Kehayias J, Roubenoff R (2004) Tumor necrosis factor-alpha production is associated with less body cell mass in women with rheumatoid arthritis. J Rheumatol 31:23–29
Roubenoff R (2009) Rheumatoid cachexia: a complication of rheumatoid arthritis moves into the 21st century. Arthritis Res Ther 11:108. https://doi.org/10.1186/ar2658
Walsmith J, Roubenoff R (2002) Cachexia in rheumatoid arthritis. Int J Cardiol 85:89–99
Evans WJ, Morley JE, Argilés J, Bales C, Baracos V, Guttridge D, Jatoi A, Kalantar-Zadeh K, Lochs H, Mantovani G, Marks D, Mitch WE, Muscaritoli M, Najand A, Ponikowski P, Rossi Fanelli F, Schambelan M, Schols A, Schuster M, Thomas D, Wolfe R, Anker SD (2008) Cachexia: a new definition. Clin Nutr 27:793–799. https://doi.org/10.1016/j.clnu.2008.06.013
Muscaritoli M, Anker SD, Argilés J, Aversa Z, Bauer JM, Biolo G, Boirie Y, Bosaeus I, Cederholm T, Costelli P, Fearon KC, Laviano A, Maggio M, Fanelli FR, Schneider SM, Schols A, Sieber CC (2010) Consensus definition of sarcopenia, cachexia and pre-cachexia: Joint document elaborated by Special Interest Groups (SIG) “cachexia-anorexia in chronic wasting diseases” and “ nutrition in geriatrics”. Clin Nutr 29:154–159. https://doi.org/10.1016/j.clnu.2009.12.004
Engvall IL, Elkan AC, Tengstrand B, Cederholm T, Brismar K, Hafstrom I (2008) Cachexia in rheumatoid arthritis is associated with inflammatory activity, physical disability, and low bioavailable insulin-like growth factor. Scand J Rheumatol 37:321–328. https://doi.org/10.1080/03009740802055984
Elkan A-C, Hakansson N, Frostegard J, Cederholm T, Hafstrom I (2009) Rheumatoid cachexia is associated with dyslipidemia and low levels of atheroprotective natural antibodies against phosphorylcholine but not with dietary fat in patients with rheumatoid arthritis: a cross-sectional study. Arthritis Res Ther 11:R37. https://doi.org/10.1186/ar2643
Santo RCE, Fernandes KZ, Lora PS, Filippin LI, Xavier RM (2018) Prevalence of rheumatoid cachexia in rheumatoid arthritis: a systematic review and meta-analysis. J Cachexia Sarcopenia Muscle 9:816–825. https://doi.org/10.1002/jcsm.12320
Metsios GS, Stavropoulos-Kalinoglou A, Panoulas VF, Sandoo A, Toms TE, Nevill AM, Koutedakis Y, Kitas GD (2009) Rheumatoid cachexia and cardiovascular disease. Clin Exp Rheumatol 27:985–988
Hugo M, Cetre NM, Pierreisnard A, Schaeverbeke T, Gin H, Rigalleau V (2016) Energy expenditure and nutritional complications of metabolic syndrome and rheumatoid cachexia in rheumatoid arthritis: an observational study using calorimetry and actimetry. Rheumatol. 55:1202–1209. https://doi.org/10.1093/rheumatology/kew038
Aletaha D, Neogi T, Silman AJ, Funovits J, Felson DT, Bingham CO et al (2010) 2010 rheumatoid arthritis classification criteria: an American College of Rheumatology/European League Against Rheumatism collaborative initiative. Arthritis Rheum 62:2569–2581. https://doi.org/10.1002/art.27584
Matsudo S, Araujo T, Matsudo V, Andrade D, Andrade E, Oliveira LC et al (2001) Questionario Internacional de Atividade Fisica (Ipaq): Estudo De Validade E Reprodutibilidade No Brasil. Ativ Fís & Sau 6:2. https://doi.org/10.12820/rbafs.v.6n2p5-18
Strand V, Rentz AM, Cifaldi MA, Chen N, Roy S, Revicki D (2012) Health-related quality of life outcomes of adalimumab for patients with early rheumatoid arthritis: results from a randomized multicenter study. J Rheumatol 39:63–72. https://doi.org/10.3899/jrheum.101161
Fujiwara M, Kita Y (2013) Reexamination of the assessment criteria for rheumatoid arthritis disease activity based on comparison of the disease activity score 28 with other simpler assessment methods. Mod Rheumatol 23:260–268. https://doi.org/10.1007/s10165-012-0652-7
Bruce B, Fries JF (2005) The health assessment questionnaire (HAQ). Clin Exp Rheumatol 23:S14–S18
Baker JF, Von Feldt JM, Mostoufi-Moab S, Kim W, Taratuta E, Leonard MB (2015) Insulin-like growth factor 1 and adiponectin and associations with muscle deficits, disease characteristics, and treatments in rheumatoid arthritis. J Rheumatol 42:2038–2045. https://doi.org/10.3899/jrheum.150280
Schutz Y, Kyle UUG, Pichard C (2002) Fat-free mass index and fat mass index percentiles in Caucasians aged 18-98 y. Int J Obes Relat Metab Disord 26:953–960
Wolfe F, Michaud K (2012) Effect of body mass index on mortality and clinical status in rheumatoid arthritis. Arthritis Care Res 64:1471–1479. https://doi.org/10.1002/acr.21627
Cruz-Jentoft AJ, Baeyens JP, Bauer JM, Boirie Y, Cederholm T, Landi F, Martin FC, Michel JP, Rolland Y, Schneider SM, Topinková E, Vandewoude M, Zamboni M, European Working Group on Sarcopenia in Older People (2010) Sarcopenia: European consensus on definition and diagnosis. Age Ageing 39:412–423. https://doi.org/10.1093/ageing/afq034
Dias JA, Ovando AC, Külkamp W, Junior NGB (2010) Hand grip strength: evaluation methods and factors influencing this measure. Rev Bras Cineantropom Desempenho Hum 12:209–216. https://doi.org/10.5007/1980-0037.2010v12n3p209
Cella D, Yount S, Sorensen M, Chartash E, Sengupta N, Grober J (2005) Validation of the functional assessment of chronic illness therapy fatigue scale relative to other instrumentation in patients with rheumatoid arthritis. J Rheumatol 32:811–819
Chang VT, Xia Q, Kasimis B (2005) The functional assessment of anorexia/cachexia therapy (FAACT) appetite scale in veteran cancer patients. J Support Oncol 3:377–382
Tournadre A, Pereira B, Dutheil F, Giraud C, Courteix D, Sapin V, Frayssac T, Mathieu S, Malochet-Guinamand S, Soubrier M (2017) Changes in body composition and metabolic profile during interleukin 6 inhibition in rheumatoid arthritis. J Cachexia Sarcopenia Muscle 8:639–646. https://doi.org/10.1002/jcsm.12189
Lemmey AB, Wilkinson TJ, Clayton RJ, Sheikh F, Whale J, Jones HSJ, Ahmad YA, Chitale S, Jones JG, Maddison PJ, O’Brien TD (2016) Tight control of disease activity fails to improve body composition or physical function in rheumatoid arthritis patients. Rheumatology. 55:1736–1745. https://doi.org/10.1093/rheumatology/kew243
Binymin K, Herrick A, Carlson G, Hopkins S (2011) The effect of disease activity on body composition and resting energy expenditure in patients with rheumatoid arthritis. J Inflamm Res 4:61–66. https://doi.org/10.2147/JIR.S16508
Marcora SM, Chester KR, Mittal G, Lemmey AB, Maddison PJ (2006) Randomized phase 2 trial of anti-tumor necrosis factor therapy for cachexia in patients with early rheumatoid arthritis. Am J Clin Nutr 84:1463–1472
van Bokhorst-de van der Schueren MAE, Konijn NPC, Bultink IEM, Lems WF, Earthman CP, van Tuyl LHD (2012) Relevance of the new pre-cachexia and cachexia definitions for patients with rheumatoid arthritis. Clin Nutr 31:1008–1010. https://doi.org/10.1016/j.clnu.2012.05.012
Stavropoulos-Kalinoglou A, Metsios GS, Smith JP, Panoulas VF, Douglas KMJ, Jamurtas AZ, Koutedakis Y, Kitas GD (2010) What predicts obesity in patients with rheumatoid arthritis an investigation of the interactions between lifestyle and inflammation. Int J Obes 34:295–301. https://doi.org/10.1038/ijo.2009.220
Baker JF, Sauer BC, Cannon GW, Teng C-C, Michaud K, Ibrahim S, Jorgenson E, Davis L, Caplan L, Cannella A, Mikuls TR (2016) Changes in body mass related to the initiation of disease-modifying therapies in rheumatoid arthritis. Arthritis Rheum 68:1818–1827. https://doi.org/10.1002/art.39647
Jurgens MS, Jacobs JWG, Geenen R, Bossema ER, Bakker MF, Bijlsma JWJ, van Albada-Kuipers IA, Ehrlich JC, Lafeber FPJG, Welsing PMJ, on behalf of The Utrecht Arthritis Cohort Study Group (2013) Increase of body mass index in a tight controlled methotrexate-based strategy with prednisone in early rheumatoid arthritis: side effect of the prednisone or better control of disease activity? Arthritis Care Res 65:88–93. https://doi.org/10.1002/acr.21797
Popescu C, Bojincă V, Opriş D, Ionescu R (2015) Dual X-ray absorptiometry whole body composition of adipose tissue in rheumatoid arthritis. Rom J Intern Med 53:1–11
Kim JK, Park MG, Shin SJ (2014) What is the minimum clinically important difference in grip strength? Clin Orthop Relat Res 472:2536–2541. https://doi.org/10.1007/s11999-014-3666-y
Palamar D, Er G, Terlemez R, Ustun I, Can G, Saridogan M (2017) Disease activity, handgrip strengths, and hand dexterity in patients with rheumatoid arthritis. Clin Rheumatol 36:2201–2208. https://doi.org/10.1007/s10067-017-3756-9
Sferra da Silva G, de Almeida Lourenço M, de Assis MR (2018) Hand strength in patients with RA correlates strongly with function but not with activity of disease. Adv Rheumatol. 3;58:20. doi: https://doi.org/10.1186/s42358-018-0020-1
Cruz-Jentoft AJ, Bahat G, Bauer J, Boirie Y, Bruyère O, Cederholm T, et al (2019) Sarcopenia: revised European consensus on definition and diagnosis. Age Ageing. 1;48:601. doi: https://doi.org/10.1093/ageing/afy169
Acknowledgments
We thank the Coordination for the Improvement of Higher Level Personnel (Coordenação de Aperfeiçoamento de Pessoal de Nível Superior, CAPES) and the Rio Grande do Sul State Research Foundation (Fundação de Amparo à Pesquisa do Estado do Rio Grande do Sul, FAPERGS) for granting scholarships to the students that contributed to this study. We thank the HCPA Department of Rheumatology and Biostatistics Service for scientific support. Financial support was provided by the HCPA Research and Events Incentive Fund (Fundo de Incentivo à Pesquisa e Eventos, FIPE), the Research Support Fund of the Rio Grande do Sul Society of Rheumatology (Fundo de Apoio à Pesquisa da Sociedade de Reumatologia do Rio Grande do Sul), and National Council for Scientific and Technological Development (Conselho Nacional de Desenvolvimento Científico e Tecnológico, CNPq).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
None declared.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Santo, R.C., Silva, J.M., Lora, P.S. et al. Cachexia in patients with rheumatoid arthritis: a cohort study. Clin Rheumatol 39, 3603–3613 (2020). https://doi.org/10.1007/s10067-020-05119-y
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10067-020-05119-y