Abstract
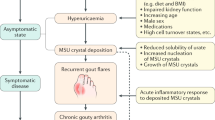
Gout is a disease with acute and/or chronic inflammation and tissue damage induced by the precipitation of monosodium urate crystal (MSU) crystals in bone joints, kidneys, and subcutaneous sites. In recent years, with the continuous research on gout animal models and patient clinical investigations, the mechanism of inflammation activation of gout has been further discovered. Studies have shown that pro-inflammatory factors such as interleukin (IL)-1β, IL-8 and IL-17, NLRP3 inflammasome, and tumor necrosis factor alpha (TNF-α), anti-inflammatory factors such as IL-10, IL-37 are all involved in the MSU-induced gout inflammatory process. And the immune cells in gout, including neutrophils, monocytes/macrophages, and lymphocytes, all play important roles in the pathogenesis of gout. In this review, we mainly emphasize the understanding of various cytokines, inflammasome, and immune cells involved in the onset of gout, in order to provide a systematic and theoretical basis for the novel exploration of inflammatory therapeutic targets for gout.




Similar content being viewed by others
Abbreviations
- ASC:
-
apoptosis-associated speck-like protein
- AGA:
-
acute gouty arthritis
- ATP:
-
adenosine triphosphate;
- CAM-1:
-
cellular adhesion molecule 1
- CIA:
-
collagen arthritis
- CLIC:
-
intracellular chloride channel protein
- DAMP:
-
dangerous-associated molecular patterns
- GM-CSF:
-
granulocyte-macrophage stimulating factor
- HC:
-
healthy control
- IL:
-
interleukin
- LRR:
-
leucine-rich repeat domain
- LPS :
-
bacterial lipopolysaccharide
- MAVS:
-
mitochondrial antiviral signaling protein
- MSU:
-
monosodium urate monohydrate
- MIP-2:
-
macrophage inflammatory protein 2
- Mfn2:
-
Mitochondrial fusion protein 2
- NF-κB:
-
activate nuclear transcription factor
- NLR:
-
NOD-like receptors
- NAGA:
-
non-acute gouty arthritis
- PRR:
-
pattern recognition receptor
- PAMP:
-
pathogen-associated molecular patterns
- PYD:
-
pyrin domain
- P2X7:
-
P2X purine receptor 7
- RANKL :
-
NF-κΒ ligands
- ROS:
-
active oxygen
- SOCS3:
-
signal transduction inhibitor 3
- TGF-β1 :
-
transforming growth factor-β1
- TNF-α:
-
tumor necrosis factor alpha
- TLR:
-
Toll-like receptor
References
Dalbeth N, Phipps-Green A, Frampton C, Neogi T, Taylor WJ, Merriman TR (2018) Relationship between serum urate concentration and clinically evident incident gout: an individual participant data analysis. Ann Rheum Dis 77(7):1048–1052
Dalbeth N, House ME, Aati O et al (2015) Urate crystal deposition in asymptomatic hyperuricaemia and symptomatic gout: a dual energy CT study. Ann Rheum Dis 74:908–911
So AK, Martinon F (2017) Inflammation in gout: mechanisms and therapeutic targets. Nat Rev Rheumatol 13(11):639–647
Scott P, Ma H, Viriyakosol S, Terkeltaub R, Liu-Bryan R (2006) Engagement of CD14 mediates the inflammatory potential of monosodium urate crystals. J Immunol 177(9):6370–6378
Barbero F, Russo L, Vitali M, Piella J, Salvo I, Borrajo ML, Busquets-Fité M, Grandori R, Bastús NG, Casals E, Puntes V (2017) Formation of the protein corona: the Interface between nanoparticles and the immune system. Semin Immunol 34:52–60
Pascual E (1994) Hyperuricemia and gout. Curr Opin Rheumatol 6(4):454–458
Renaudin F, Sarda S, Campillo-Gimenez L, Séverac C, Léger T, Charvillat C, Rey C, Lioté F, Camadro JM, Ea HK, Combes C (2019) Adsorption of proteins on m-CPPD and urate crystals inhibits crystal-induced cell responses: study on albumin-crystal interaction. J Funct Biomater 10(2):18
Piccini A, Carta S, Tassi S, Lasiglie D, Fossati G, Rubartelli A (2008) ATP is released by monocytes stimulated with pathogen-sensing receptor ligands and induces IL-1β and IL-18 secretion in an autocrine way. Proc Natl Acad Sci U S A 105:8067–8072
Scanu A, Luisetto R, Oliviero F, Gruaz L, Sfriso P, Burger D, Punzi L (2015) High-density lipoproteins inhibit urate crystal-induced inflammation in mice. Ann Rheum Dis 74:587–594
Rock KL, Kataoka H, Lai JJ (2013) Uric acid as a danger signal in gout and its comorbidities. Nat Rev Rheumatol 9(1):13–23
Joosten LA, Abdollahi-Roodsaz S, Dinarello CA et al (2016) Toll-like receptors and chronic inflammation in rheumatic diseases: new developments. Nat Rev Rheumatol 12(6):344–357
Narayanan KB, Park HH (2015) Toll/interleukin-1 receptor (TIR) domain-mediated cellular signaling pathways. Apoptosis. 20(2):196–209
Liu-Bryan R, Scott P, Sydlaske A, Rose DM, Terkeltaub R (2005) Innate immunity conferred by toll-like receptors 2 and 4 and myeloid differentiation factor 88 expression is pivotal to monosodium urate monohydrate crystal-induced inflammation. Arthritis Rheum 52(9):2936–2946
Chen CJ, Shi Y, Hearn A, Fitzgerald K, Golenbock D, Reed G, Akira S, Rock KL (2006) MyD88-dependent IL-1 receptor signaling is essential for gouty inflammation stimulated by monosodium urate crystals. J Clin Invest 116(8):2262–2271
Qi W, Cheng X-S (2015) Research advances in NLRP3 inflammasome. Basic Clin Med 15(1):117–121
Elliott EI, Sutterwala FS (2015) Initiation and perpetuation of NLRP3 inflammasome activation and assembly. Immunol Rev 265(1):35–52
Samways DS, Li Z, Egan TM (2014) Principles and properties of ion flow in P2X receptors. Front Cell Neurosci 8:6
Di A, Xiong S, Ye Z et al (2018) The TWIK2 potassium efflux channel in macrophages mediates NLRP3 inflammasome-induced inflammation. Immunity 49(1):56–65.e4
Triantafilou K, Hughes TR, Triantafilou M, Morgan BP (2013) The complement membrane attack complex triggers intracellular Ca2+ fluxes leading to NLRP3 inflammasome activation. J Cell Sci 126(Pt 13):2903–2913
Laudisi F, Spreafico R, Evrard M, Hughes TR, Mandriani B, Kandasamy M, Morgan BP, Sivasankar B, Mortellaro A (2013) Cutting edge: the NLRP3 inflammasome links complement-mediated inflammation and IL-1β release. J Immunol 191(3):1006–1010
Asgari E, Le Friec G, Yamamoto H et al (2013) C3a modulates IL-1β secretion in human monocytes by regulating ATP efflux and subsequent NLRP3 inflammasome activation. Blood. 122(20):3473–3481
Muñoz-Planillo R, Kuffa P, Martínez-Colón G, Smith BL, Rajendiran TM, Núñez G (2013) K+ efflux is the common trigger of NLRP3 inflammasome activation by bacterial toxins and particulate matter. Immunity. 38(6):1142–1153
Gaidt MM, Ebert TS, Chauhan D, Schmidt T, Schmid-Burgk JL, Rapino F, Robertson AAB, Cooper MA, Graf T, Hornung V (2016) Human monocytes engage an alternative inflammasome pathway. Immunity. 44(4):833–846
Groß CJ, Mishra R, Schneider KS, Médard G, Wettmarshausen J, Dittlein DC, Shi H, Gorka O, Koenig PA, Fromm S, Magnani G, Ćiković T, Hartjes L, Smollich J, Robertson AAB, Cooper MA, Schmidt-Supprian M, Schuster M, Schroder K, Broz P, Traidl-Hoffmann C, Beutler B, Kuster B, Ruland J, Schneider S, Perocchi F, Groß O (2016) K+ efflux-independent NLRP3 inflammasome activation by small molecules targeting mitochondria. Immunity. 45(4):761–773
Courbet A, Bec N, Constant C, Larroque C, Pugniere M, el Messaoudi S, Zghaib Z, Khier S, Deleuze-Masquefa C, Gattacceca F (2017) Imidazoquinoxaline anticancer derivatives and imiquimod interact with tubulin: characterization of molecular microtubule inhibiting mechanisms in correlation with cytotoxicity. PLoS One 12(8):e0182022
Zghaib Z, Guichou JF, Vappiani J, Bec N, Hadj-Kaddour K, Vincent LA, Paniagua-Gayraud S, Larroque C, Moarbess G, Cuq P, Kassab I, Deleuze-Masquéfa C, Diab-Assaf M, Bonnet PA (2016) New imidazoquinoxaline derivatives: synthesis, biological evaluation on melanoma, effect on tubulin polymerization and structure-activity relationships. Bioorg Med Chem 24(11):2433–2440
Haneklaus M, O’Neill LA, Coll RC (2013) Modulatory mechanismscontrolling the NLRP3 inflammasome in inflammation: recent developments. Curr Opin Immunol 25(1):40–45
Zhou R, Tardivel A, Thorens B, Choi I, Tschopp J (2010) Thioredoxin-interacting protein links oxidative stress to inflammasome activation. Nat Immunol 11(2):136–140
Chen J, Chen ZJ (2018) PtdIns4P on dispersed trans-Golgi network mediates NLRP3 inflammasome activation. Nature. 564(7734):71–76
Martin WJ, Walton M, Harper J (2009) Resident macrophages initiating and driving inflammation in a monosodium Urate monohydrate crystal-induced murine peritoneal model of acute gout. Arthritis Rheum 60(1):281–289
Hoffman HM, Scott P, Mueller JL, Misaghi A, Stevens S, Yancopoulos GD, Murphy A, Valenzuela DM, Liu-Bryan R (2010) Role of the leucine-richrepeat domain of cryopyrin/NLRP3 in monosodium urate crystai-induced inflammation in mice. Arthritis Rheum 62(7):2170–2179
Martinon F, Pétrilli V, Mayor A, Tardivel A, Tschopp J (2006) Gout-associated uric acid crystals activate the NALP3 inflammasome. Nature. 440(7081):237–241
Pisetsky DS, Ward MM (2012) Advances in the treatment of inflammatory arthritis. Best Pract Res Clin Rheumatol 26(2):251–261
Bauernfeind F, Niepmann S, Knolle PA, Hornung V (2016) Aging-associated TNF production primes inflammasome activation and NLRP3-related metabolic disturbances. J Immunol 197(7):2900–2908
Yokose K, Sato S, Asano T, Yashiro M, Kobayashi H, Watanabe H, Suzuki E, Sato C, Kozuru H, Yatsuhashi H, Migita K (2018) TNF-α potentiates uric acid-induced interleukin-1β (IL-1β) secretion in human neutrophils. Mod Rheumatol 28(3):513–517
Amaral FA, Bastos LF, Oliveira TH et al (2016) Transmembrane TNF-α is sufficient for articular inflammation and hypernociception in a mouse model of gout. Eur J Immunol 46(1):204–211
Cabrera SM, Wang X, Chen YG, Jia S, Kaldunski ML, Greenbaum CJ, the Type 1 Diabetes TrialNet Canakinumab Study Group, Mandrup-Poulsen T, the AIDA Study Group, Hessner MJ (2016) Interleukin-1 antagonism moderates the inflammatory state associated with type 1 diabetes during clinical trials conducted at disease onset. Eur J Immunol 46(4):1030–1046
Busso N, So A (2010) Mechanisms of inflammation in gout. Arthritis Res Ther 12(2):206
Sutton C, Brereton C, Keogh B, Mills KHG, Lavelle EC (2006) A crucial role for interleukin (IL)-1 in the induction of IL-17-producing T cells that mediate autoimmune encephalomyelitis. J Exp Med 203(7):1685–1691
Janssen CA, Oude Voshaar MAH, Vonkeman HE, Jansen TLTA, Janssen M, Kok MR, Radovits B, van Durme C, Baan H, van de Laar MAFJ (2019) Anakinra for the treatment of acute gout flares: a randomized, double-blind, placebo-controlled, active-comparator, non-inferiority trial. Rheumatology (Oxford) 58:1344–1352
Kim KW, Kim BM, Lee KA et al (2019) Reciprocal interaction between macrophage migration inhibitory factor and interleukin-8 in gout. Clin Exp Rheumatol 37(2):270–278
Liu R, Aupperle K, Terkeltaub R (2001) Src family protein tyrosine kinase signaling mediates monosodium urate crystal-induced IL-8 expression by monocytic THP-1 cells. J Leukoc Biol 70(6):961–968
Liu R, O’Connell M, Johnson K et al (2000) Extracellular signal-regulated kinase 1/extracellular signal-regulated kinase 2 mitogen-activated protein kinase signaling and activation of activator protein 1 and nuclear factor kappaB transcription factors play central roles in interleukin-8 expression stimulated by monosodium urate monohydrate and calcium pyrophosphate crystals in monocytic cells. Arthritis Rheum 43(5):1145–1155
Conforti-Andreoni C, Spreafico R, Qian HL, Riteau N, Ryffel B, Ricciardi-Castagnoli P, Mortellaro A (2011) Uric acid-driven Th17 differentiation requires inflammasome-derived IL-1 and IL-18. J Immunol 187(11):5842–5850
Mills KH, Dungan LS, Jones SA et al (2013) The role of inflammasome-derived IL-1 in driving IL-17 responses. J Leukoc Biol 93(4):489–497
Liu Y, Zhao Q, Yin Y, McNutt MA, Zhang T, Cao Y (2018) Serum levels of IL-17 are elevated in patients with acute gouty arthritis. Biochem Biophys Res Commun 497(3):897–902
Johnson JL, Jones MB, Cobb BA (2018) Polysaccharide-experienced effector T cells induce IL-10 in FoxP3+ regulatory T cells to prevent pulmonary inflammation. Glycobiology. 28(1):50–58
Chen S, Chen B, Wen Z, Huang Z, Ye L (2017) IL-33/ST2-mediated inflammation in macrophages is directly abrogated by IL-10 during rheumatoid arthritis. Oncotarget. 8(20):32407–32418
Chen YH, Hsieh SC, Chen WY, Li KJ, Wu CH, Wu PC, Tsai CY, Yu CL (2011) Spontaneous resolution of acute gouty arthritis is associated with rapid induction of the anti-inflammatory factors TGF-β1, IL-10 and soluble TNF receptors and the intracellular cytoldne negative regulators CIS and SOCS3. Ann Rheum Dis 70(9):1655–1663
Schreiber S, Heinig T, Thiele HG, Raedler A (1995) Immuno-regulatory role of interleukin-10 in patients with inflammatory bowel disease. Gastroenterology. 108(5):1434–1444
Zhuang X, Wu B, Li J, Shi H, Jin B, Luo X (2017) The emerging role of interleukin-37 in cardiovascular diseases. Immun Inflamm Dis 5(3):373–379
Eisenmesser EZ, Gottschlich A, Redzic JS, Paukovich N, Nix JC, Azam T, Zhang L, Zhao R, Kieft JS, The E, Meng X, Dinarello CA (2019) Interleukin-37 monomer is the active form for reducing innate immunity. Proc Natl Acad Sci U S A 116(12):5514–5522
Nold MF, Nold-Petty CA, Zepp JA et al (2010) IL-37 is a fundamental inhibitor of innate immunity. Nat Immunol 11(11):1014–1022
Liu L, Xue Y, Zhu Y, Xuan D, Yang X, Liang M, Wang J, Zhu X, Zhang J, Zou H (2016) Interleukin 37 limits monosodium urate crystal-induced innate immune responses in human and murine models of gout. Arthritis Res Ther 18(1):268
Zeng M, Dang W, Chen B, Qing Y, Xie W, Zhao M, Zhou J (2016) IL-37 inhibits the production of proinflammatory cytokines in MSU crystal-induced inflammatory response. Clin Rheumatol 35:2251–2258
Alvarez-Soria MA, Herrero-Beaumont G, Sánchez-Pernaute O et al (2008) Diacerein has a weak effect on the catabolic pathway of human osteoarthritis synovial fibroblast—comparison to its effects on osteoarthritic chondrocytes. Rheumatology(Oxford). 47(5):627–633
Martin WJ, Shaw O, Liu X, Steiger S, Harper JL (2011) Monosodium urate monohydrate crystal-recruited noninflammatory monocytes differentiate into M1-like proinflammatory macrophages in a peritoneal murine model of gout. Arthritis Rheum 63:1322–1332
Luo C (2015) Distinct impact of uric acid crystals and high uric acid on human monocytes/macrophages THP-1. Shantou University R589.7: 1–77
Yang H, Yang X, Yufeng Q et al (2018) Study on the expression and significance of Th1/Th2 cells in the blood of patients with primary gouty arthritis. Chin J Rheumatol 22(11):731–736
Yang H, Yang X, Xiaowu Z et al (2016) Role of Thl7/Treg cell balance in the pathogenesis of primary gout arthritis. Chin J Rheumatol 20(8):520–525
Lee SJ, Nam KI, Jin HM, Cho YN, Lee SE, Kim TJ, Lee SS, Kee SJ, Lee KB, Kim N, Park YW (2011) Bone destruction by receptor activator of nuclear factor kappaΒ ligand-expressing T cells in chronic gouty arthritis. Arthritis Res Ther 13(5):R164
Luo CY, Wang L, Sun C, Li DJ (2011) Estrogen enhances the functions of CD4(+)CD25(+) Foxp3(+) regulatory T cells that suppress osteoclast differentiation and bone resorption in vitro. Cell Mol Immunol 8(1):50–58
Scanu A, Oliviero F, Ramonda R, Frallonardo P, Dayer JM, Punzi L (2012) Cytokine levels in human synovial fluid during the different stages of acute gout: role of transforming growth factor β1 in the resolution phase. Ann Rheum Dis 71(4):621–624
Satoh T, Otsuka A, Contassot E, French LE (2015) The inflammasome and IL-1β: implications for the treatment of inflammatory diseases. Immunotherapy. 7(3):243–254
Mokuda S, Kanno M, Takasugi K et al (2014) Tocilizumab improved clinical symptoms of a patient with systemic tophaceous gout who had symmetric polyarthritis and fever: an alternative treatment by blockade of interleukin-6 signaling. SAGE Open Med Case Rep 2:2050313X13519774
Yang CS, Kim JJ, Kim TS, Lee PY, Kim SY, Lee HM, Shin DM, Nguyen LT, Lee MS, Jin HS, Kim KK, Lee CH, Kim MH, Park SG, Kim JM, Choi HS, Jo EK (2015) Small heterodimer partner interacts with NLRP3 and negatively regulates activation of the NLRP3 inflammasome. Nat Commun 6:6115
Funding
This work was supported by grants from the National Natural Science Foundation of China (No. 81270577), the Science and Technology Innovation Foundation of Shenzhen, China (No. JCYJ20160427191026117), the Science and Technology Innovation Foundation of Baoan, Shenzhen, China (No. 2018JD237, No. 2016CX191), and the Construction Units of Key Specialties in Clinical Medicine, Baoan District, Shenzhen, China (No. 8, 214-2018, Health Commission of Baoan, Shenzhen City).
Author information
Authors and Affiliations
Contributions
Literature review: Meimei Wu and Chengshan Guo; Drafting of manuscript: Meimei Wu, Ye Tian, and Qianqian Wang. Revision of manuscript: Chengshan Guo and Meimei Wu. All authors have read and approved the final submitted manuscript.
Corresponding author
Ethics declarations
Disclosures
None.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Wu, M., Tian, Y., Wang, Q. et al. Gout: a disease involved with complicated immunoinflammatory responses: a narrative review. Clin Rheumatol 39, 2849–2859 (2020). https://doi.org/10.1007/s10067-020-05090-8
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10067-020-05090-8