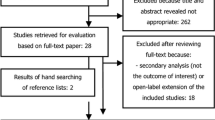
Abstract
Significant portion of patients with psoriatic arthritis (PsA) could not tolerate or do not have a satisfactory response to either non-steroidal anti-inflammatory drugs (NSAIDs), non-biologic disease-modifying anti-rheumatic drugs (DMARDs), or even TNF inhibitors. Non-TNF inhibitor biologic agents have emerged as second-line therapy in such situation. However, the comparative efficacy of these agents remains unknown as head-to-head randomized controlled trials (RCTs) are not available. RCTs examining the efficacy of non-TNF inhibitor biologic agents in patients with PsA who experienced inadequate response or intolerance of TNF inhibitors were identified. If more than one RCT was available for a given biologic agent, the pooled odds ratio (OR) and 95 % confidence interval (CI) of achieving 20 % improvement according to American College of Rheumatology criteria (ACR20) response across trials were calculated. The pooled OR for each biologic agent was then compared using the indirect comparison technique. Five RCTs of four non-TNF inhibitor biologic agents, including abatacept, secukinumab, ustekinumab, and apremilast, with 675 participants were identified and included in the data analyses. We found no significant difference in any comparisons, with the p values ranging from 0.14 to 0.98. Our study demonstrates that the likelihood of achieving the ACR20 response in patients with TNF inhibitor experience is not significantly different between the four non-TNF biologic agents. However, the interpretation of this analysis is limited by the small sample sizes. Head-to-head comparisons are still required to confirm the comparative efficacy.


Similar content being viewed by others
References
Boehncke WH, Schon MP (2015) Psoriasis. Lancet 386:983–994
Huynh D, Kavanaugh A (2015) Psoriatic arthritis: current therapy and future approaches. Rheumatology (Oxford) 54:20–28
Anandarajah AP, Ritchlin CT (2009) The diagnosis and treatment of early psoriatic arthritis. Nat Rev Rheumatol 5:634–641
Gladman DD, Antoni C, Mease P et al (2005) Psoriatic arthritis: epidemiology, clinical features, course, and outcome. Ann Rheum Dis 64(Suppl 2):ii14–ii17
Walsh JA, McFadden ML, Morgan MD et al (2014) Work productivity loss and fatigue in psoriatic arthritis. J Rheumatol 41:1670–1674
Ungprasert P, Sanguankeo A, Upala S, Suksaranjit P (2014) Psoriasis and risk of venous thromboembolism: a systematic review and meta-analysis. QJM 107:793–797
Benson MM, Frishman WH (2015) The heartbreak of psoriasis: a review of cardiovascular risk in patients with psoriasis. Cardiol Rev 23:312–316
Kingsley GH, Kowalczyk A, Taylor H et al (2012) A randomized placebo-controlled trial of methotrexate in psoriatic arthritis. Rheumatology (Oxford) 51:1368–1377
Willkens RF, Williams HJ, Ward JR et al (1984) Randomized, double-blind, placebo controlled trial of low-dose pulse methotrexate in psoriatic arthritis. Arthritis Rheum 27:376–381
Soriano ER, McHugh NJ (2006) Therapies for peripheral joint disease in psoriatic arthritis. A systematic review. J Rheumatol 33:1422–1430
Antoni C, Krueger GG, de Vlam K et al (2005) Infliximab improves signs and symptoms of psoriatic arthritis: results of the IMPACT 2 trial. Ann Rheum Dis 64:1150–1157
Mease PJ, Gladman DD, Ritchlin CT et al (2005) Adalimumab for the treatment of patients with moderately to severely active psoriatic arthritis: results of a double-blind, randomized, placebo-controlled trial. Arthritis Rheum 52:3279–3289
Mease PJ, Goffe BS, Metz J, Vanderstoep A, Finck B, Burge DJ (2000) Etanercept in the treatment of psoriatic arthritis and psoriasis: a randomised trial. Lancet 356:385–390
Kavanaugh A, McInnes I, Mease P et al (2009) Golimumab, a new human tumor necrosis factor alpha antibody, administered every four weeks as a subcutaneous injection in psoriatic arthritis: twenty-four-week efficacy and safety results of a randomized, placebo-controlled study. Arthritis Rheum 60:976–986
Mease PJ, Fleischmann R, Deodhar AA et al (2014) Effect of certolizumab pegol on signs and symptoms in patients with psoriatic arthritis: 24-week results of a Phase 3 double-blind randomised placebo-controlled study (RAPID-PsA). Ann Rheum Dis 73:48–55
Jadad AR, Moore RA, Carroll D et al (1996) Assessing the quality of reports of randomized clinical trials: is blinding necessary? Control Clin Trials 17:1–12
Felson DT, Anderson JJ, Boers M et al (1993) The American College of Rheumatology preliminary core set of disease activity measures for rheumatoid arthritis clinical trials. The Committee on Outcome Measures in Rheumatoid Arthritis Clinical Trials. Arthritis Rheum 36:729–740
Hedges LV, Vevea JL (1998) Fixed- and random-effects models in meta-analysis. Psychol Methods 3:486–504
Higgins JP, Thompson SG, Deeks JJ, Altman DG (2003) Measuring inconsistency in meta-analyses. BMJ 327:557–560
Bucher HC, Guyatt GH, Griffith LE, Walter SD (1997) The results of direct and indirect treatment comparisons in meta-analysis of randomized controlled trials. J Clin Epidemiol 50:683–691
Song F, Altman DG, Glenny AM, Deeks JJ (2003) Validity of indirect comparison for estimating efficacy of competing interventions: empirical evidence from published meta-analyses. BMJ 326:472
Gottlieb A, Menter A, Mendelsohn A et al (2009) Ustekinumab, a human interleukin 12/23 monoclonal antibody, for psoriatic arthritis: randomised, double-blind, placebo-controlled, crossover trial. Lancet 373:633–640
Schett G, Wollenhaupt J, Papp K et al (2012) Oral apremilast in the treatment of active psoriatic arthritis: results of a multicenter, randomized, double-blind, placebo-controlled study. Arthritis Rheum 64:3156–3167
Mease P, Genovese MC, Gladstein G et al (2011) Abatacept in the treatment of patients with psoriatic arthritis. Results of six-month, multicenter, randomized, double-blind, placebo–controlled, phase II trial. Arthritis Rheum 63:939–948
Ritchlin C, Rahman P, Kavanaugh A et al (2014) Efficacy and safety of the anti-IL-12/23 p40 monoclonal antibody, ustekinumab, in patients with active psoriatic arthritis despite conventional non-biological and biological anti-tumour necrosis factor therapy: 6-month and 1-year results of the phase 3, multicentre, double-blind, placebo-controlled, randomised PSUMMIT 2 trial. Ann Rheum Dis 73:990–999
Kavanaugh A, Mease PJ, Gomez-Reino JJ et al (2014) Treatment of psoriatic arthritis in a phase 3 randomised, placebo-controlled trial with apremilast, an oral phosphodiesterase 4 inhibitor. Ann Rheum Dis 73:1020–1026
Mease PJ, McInnes IB, Kirkham B et al (2015) Secukinumab inhibition of interleukin-17A in patients with psoriatic arthritis. N Engl J Med 373:1329–1339
McInnes IB, Mease PJ, Kirkham B et al (2015) Secukinumab, a human anti-interleukin-17A monoclonal antibody, in patients with psoriatic arthritis (FUTURE 2): a randomized, double-blind, placebo-controlled, phase 3 trial. Lancet 386:1137–1146
Glintborg B, Østergaard M, Dreyer L et al (2011) Treatment response, drug survival, and predictors thereof in 764 patients with psoriatic arthritis treated with anti-tumor necrosis factor α therapy: results from the nationwide Danish DANBIO registry. Arthritis Rheum 63:382–390
Antoni CE, Kavanaugh A, Kirkham B et al (2005) Sustained benefits of infliximab therapy for dermatologic and articular manifestations of psoriatic arthritis: results from the infliximab multinational psoriatic arthritis controlled trial (IMPACT). Arthritis Rheum 52:1227–1236
Genovese MC, Mease PJ, Thomson GT et al (2007) Safety and efficacy of adalimumab in treatment of patients with psoriatic arthritis who had failed disease modifying antirheumatic drug therapy. J Rheumatol 34:1040–1050
Mease PJ, Kivitz AJ, Burch FX et al (2004) Etanercept treatment of psoriatic arthritis: safety, efficacy, and effect on disease progression. Arthritis Rheum 50:2264–2272
Lemos LL, de Oliveira CJ, Almeida AM et al (2014) Treatment of psoriatic arthritis with anti-TNF agents: a systematic review and meta-analysis of efficacy, effectiveness and safety. Rheumatol Int 34:1345–1360
Yildiz A, Vieta E, Correll CU, Nikodem M, Baldessarini RJ (2014) Critical issues on the use of network meta-analysis in psychiatry. Harv Rev Psychiatry 22:367–372
Salanti G, Del Giovane C, Chaimani A, Caldwell DM, Higgins JP (2014) Evaluating the quality of evidence from a network meta-analysis. PLoS One 9:e99682
Acknowledgments
Author’s contribution
Patompong Ungprasert did the study design, literature search, statistical analysis, and writing of the manuscript. Charat Thongprayoon did the literature search and manuscript revision. John Davis did the study design and manuscript revision.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Funding
None.
Conflict of interest statement
John M. Davis is a site investigator for trials sponsored by Pfizer and Roche/Genentech. Division of Rheumatology, Mayo Clinic, receives research funding from Pfizer for John M. Davis’s role on a grant.
Electronic supplementary material
Below is the link to the electronic supplementary material.
ESM 1
(DOC 23 kb)
Rights and permissions
About this article
Cite this article
Ungprasert, P., Thongprayoon, C. & Davis, J.M. Indirect comparisons of the efficacy of subsequent biological agents in patients with psoriatic arthritis with an inadequate response to tumor necrosis factor inhibitors: a meta-analysis. Clin Rheumatol 35, 1795–1803 (2016). https://doi.org/10.1007/s10067-016-3204-2
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10067-016-3204-2