Abstract
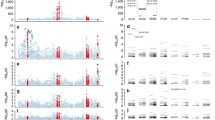
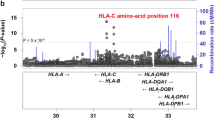
The 40-year-old association of HLA-B27 with ankylosing spondylitis is one of the best examples of disease association with a hereditary marker. Genomewide association and family studies suggest that other important major histocompatibility complex (MHC) influences are operative in ankylosing spondylitis (AS) susceptibility. HLA-B27 positive hepatitis C individuals are immunologically more efficient in combating viral infections such as HIV-1, hepatitis C, and influenza and less efficient in combating against certain bacteria (and perhaps other organisms) capable of surviving intracellularly. A recent representative population survey of the frequency of HLA-B27 in the USA found a lower frequency of HLA-B27 in older US adults, perhaps reflecting this. Other HLA class I and class II alleles have been implicated in AS susceptibility, the most consistent being HLA-B*40/B60 (B*40:01) but also B14, B15, A*0201, DRB1*04:04, and certain DPA1 and DPB1 alleles. Non-HLA MHC alleles have also been implicated, although many such studies have been inconsistent, likely due to power issues related to the low number of HLA-B27-negative AS patients examined. The best evidence is for major histocompatibility complex class I chain-related gene A (MICA) whose recognition by intestinal epithelial T cells expressing different V-delta-1 gamma/delta TCR further implicates the gut in AS pathogenesis. The HLA class I and class II and other non-HLA allelic associations underscore the importance of T cells in AS pathogenesis.



Similar content being viewed by others
References
Schlosstein L, Terasaki PI, Bluestone R, Pearson CM (1973) High association of an HL-A antigen, W27, with ankylosing spondylitis. N Engl J Med 288:704–706
Brewerton DA, Hart FD, Nicholls A et al (1973) Ankylosing spondylitis and HL-A 27. Lancet 301(7809):904–907
Cortes A, Hadler J, Pointon JP et al (2013) Multiple novel loci harbouring common and rare variants implicated in ankylosing spondylitis. International Genetics of Ankylosing Spondylitis Consortium (IGAS). Nat Genet 45:730–738
Brown MA, Kennedy LG, MacGregor AJ et al (1997) Susceptibility to ankylosing spondylitis in twins: the role of genes, HLA, and the environment. Arthritis Rheum 40:1823–1828
Zhang G, Luo J, Bruckel J et al (2004) Genetic studies in familial ankylosing spondylitis susceptibility. Arthritis Rheum 50:2246–2254
Joshi R, Reveille JD, Brown MA et al (2012) Is there a higher genetic load of susceptibility loci in familial ankylosing spondylitis? Arthritis Care Res (Hoboken) 64:780–784
Australo-Anglo-American Spondyloarthritis Consortium (TASC) and the Wellcome Trust Case Control Consortium 2 (WTCCC2) (2011) Genome-wide association study in ankylosing spondylitis identifies further non-MHC associations, and demonstrates that the ERAP1 association is restricted to HLA-B27 positive cases implicating peptide presentation as the likely mechanism underlying the association of HLA-B27 with the disease. Nat Genet 43:761–767
WTCCC, TASC (2007) Association scan of 14,500 nsSNPs in four common diseases identifies variants involved in autoimmunity. Nat Genet 39:1329–1337
Tam LS, Gu J, Yu D (2010) Pathogenesis of ankylosing spondylitis. Nat Rev Rheumatol 6:399–405
Layh-Schmitt G, Colbert RA (2008) The interleukin-23/interleukin-17 axis in spondyloarthritis. Curr Opin Rheum 20:392–397
Hannu H, Inman RD, Granfors K, Leirisalo-Repo M (2006) Reactive arthritis or postinfectious arthritis. Best Pract Res Clin Rheum 20:419–433
Rosenbaum JT, Davey MP (2011) Time for a gut check: evidence for the hypothesis that HLA-B27 predisposes to ankylosing spondylitis by altering the microbiome. Arthritis Rheum 63:3195–3198
Reveille JD, Maganti RM (2009) Subtypes of HLA-B27: history and implications in the pathogenesis of ankylosing spondylitis. Adv Exp Med Biol 649:159–176
Hendel H, Caillat-Zucman S, Lebuanec H et al (1999) New class I and II HLA alleles strongly associated with opposite patterns of progression to AIDS. J Immunol 162:6942–6946
Dorak MT, Tang J, Tang S et al (2003) Influence of human leukocyte antigen-B22 alleles on the course of human immunodeficiency virus type 1 infection in 3 cohorts of white men. J Infect Dis 188:856–863
Roger M (1998) Influence of host genes on HIV-1 disease progression. FASEB J 12:625–632
Goulder PJ, Phillips RE, Colbert RA et al (1997) Late escape from an immunodominant cytotoxic T-lymphocyte response associated with progression to AIDS. Nat Med 3:212–217
Streeck H, Lichterfeld M, Alter G et al (2007) Recognition of a defined region within p24 gag by CD8+ T cells during primary human immunodeficiency virus type 1 infection in individuals expressing protective HLA class I alleles. J Virol 81:7725–7731
Martin MP, Gao X, Lee JH et al (2002) Epistatic interaction between KIR3DS1 and HLA-B delays the progression to AIDS. Nat Genet 31:429–434
Neumann-Haefelin C (2013) HLA-B27-mediated protection in HIV and hepatitis C virus infection and pathogenesis in spondyloarthritis: two sides of the same coin? Curr Opin Rheumatol 25:426–433
Neumann-Haefelin C, Oniangue-Ndza C, Kuntzen T et al (2011) Human leukocyte antigen B27 selects for rare escape mutations that significantly impair hepatitis C virus replication and require compensatory mutations. Hepatology 54:1157–1166
Nitschke K, Barriga A, Schmidt J et al (2014) HLA-B*27 subtype specificity determines targeting and viral evolution of a hepatitis C virus-specific CD8+ T cell epitope. J Hepatol 60:22–29
Mathieu A, Paladini F, Vacca A et al (2009) The interplay between the geographic distribution of HLA-B27 alleles and their role in infectious and autoimmune diseases: a unifying hypothesis. Autoimmun Rev 8(5):420–425
Reveille JD, Hirsch R, Dillon CF, Carroll MD, Weisman MH (2012) The Prevalence of HLA-B27 in the United States: Data from the U.S. National Health and Nutrition Examination Survey, 2009. Arthritis Rheum 64:1407–1411
Breban M (1998) Genetic studies of spondylarthropathies. French Spondylarthropathy Genetic Study Group. Ann Med Interne (Paris) 149:142–144
Kawaguchi G, Kato N, Kashiwase K et al (1993) Structural analysis of HLA-B40 epitopes. Hum Immunol 36:193–198
Robinson WP, van der Linden SM, Khan MA et al (1989) HLA-Bw60 increases susceptibility to ankylosing spondylitis in HLA-B27+ patients. Arthritis Rheum 32:1135–1141
Brown MA, Pile KD, Kennedy LG et al (1996) HLA class I associations of ankylosing spondylitis in the white population in the United Kingdom. Ann Rheum Dis 55:268–270
van Gaalen FA, Verduijn W, Roelen DL et al (2013) Epistasis between two HLA antigens defines a subset of individuals at a very high risk for ankylosing spondylitis. Ann Rheum Dis 72:974–978
Wei JC, Tsai WC, Lin HS, Tsai CY, Chou CT (2004) HLA-B60 and B61 are strongly associated with ankylosing spondylitis in HLA-B27-negative Taiwan Chinese patients. Rheumatology (Oxford) 43:839–842
López-Larrea C, Mijiyawa M, González S et al (2002) Association of ankylosing spondylitis with HLA-B*1403 in a West African population. Arthritis Rheum 46:2968–2971
Díaz-Peña R, Blanco-Gelaz MA, Njobvu P et al (2008) Influence of HLA-B*5703 and HLA-B*1403 on susceptibility to spondyloarthropathies in the Zambian population. J Rheumatol 35:2236–2240
Merino Galocha B, Vázquez MN, López de Castro JA (2008) Disparate folding and stability of the ankylosing spondylitis-associated HLA-B*1403 and B*2705 proteins. Arthritis Rheum 58:3693–3704
Vargas-Alarcón G, Hernández-Pacheco G, Pacheco-Tena C et al (2002) Effect of HLA-B and HLA-DR genes on susceptibility to and severity of spondyloarthropathies in Mexican patients. Ann Rheum Dis 61:714–717
Mielants, Veys EM, Joos R, Noens L,Cuvelier C, De Vos M (1987) HLA antigens in seronegative spondyloarthropathies. Reactive arthritis and arthritis in ankylosing spondylitis. Relation to gut Inflammation. J Rheumatol. 14:466–471
Mielants, Veys EM, De vos M, Cuvelier C et al (1995) The evolution of spondyloarthropathies in relation to gut histology. I. Clinical aspects. J Rheumatol 22:2266–2272.
Siala M, Mahfoudh N, Fourati H et al (2009) MHC class I and class II genes in Tunisian patients with reactive and undifferentiated arthritis. Clin Exp Rheumatol 27:208–213
Said-Nahal R, Miceli-Richard C, Gautreau C et al (2002) The role of HLA genes in familial spondyloarthropathy: a comprehensive study of 70 multiplex families. Ann Rheum Dis 61:201–206
Wang J, Yang Y, Guo S et al (2013) Association between copy number variations of HLA-DQA1 and ankylosing spondylitis in the Chinese Han population. Genes Immun 14:500–503
Ploski R, Flato B, Vinje O et al (1995) Association to HLA-DRB1*08, HLA-DPB1*0301 and homozygosity for an HLA-linked proteasome gene in juvenile ankylosing spondylitis. Hum Immunol 44:88–96
Díaz-Peña R, Aransay AM, Bruges-Armas J et al (2011) Fine mapping of a major histocompatibility complex in ankylosing spondylitis: association of the HLA-DPA1 and HLA-DPB1 regions. Arthritis Rheum 63:3305–3312
Groh V, Steinle A, Bauer S, Spies T (1998) Recognition of stress-induced MHC molecules by intestinal epithelial gamma-delta T cells. Science 279:1737–1740
Goto K, Ota M, Ohno S et al (1997) MICA gene and anky losing spondylitis: linkage analysis via a transmembrane-encoded triplet repeat polymorphism. Tissue Antigens 49:503–507
Yabuki K, Ota M, Goto K et al (1999) Triplet repeat polymorphism in the MICA gene in HLA-B27 positive and negative Caucasian patients with ankylosing spondylitis. Hum Immunol 60:83–86
Ricci-Vitiani L, Vacca A, Potolicchio I et al (2000) MICA gene triplet repeat polymorphism in patients with HLA-B27 positive and negative ankylosing spondylitis from Sardinia. J Rheumatol 27:2193–2197
Zhou X, Wang J, Zou H, et al. (2013) MICA, a gene contributing strong susceptibility to ankylosing spondylitis.Ann Rheum Dis. Jun 1. [Epub ahead of print]
Hohler T, Schaper T, Schneider PM et al (1998) Association of different tumor necrosis factor alpha promoter allele frequencies with ankylosing spondylitis in HLA-B27 positive individuals. Arthritis Rheum 41:1489–1492
Fraile A, Collado MD, Mataran L et al (2000) TAP1 and TAP2 polymorphism in Spanish patients with ankylosing spondylitis. Exp Clin Immunogenet 17:199–204
Barron KS, Reveille JD, Carrington M, Mann DL, Robinson MA (1995) Susceptibility to Reiter's syndrome is associated with alleles of TAP genes. Arthritis Rheum 38:684–689
Maksymowych WP, Wessler A, Schmitt-Egenolf M et al (1997) Polymorphism in an HLA linked proteasome gene influences phenotypic expression of disease in HLA-B27 positive individuals. J Rheumatol 21:665–669
Acknowledgments
This work was supported by the National Institutes of Health-National Institute of Allergy and Infectious Diseases (NIH-NIAIS) grant UO1 AI09090, National Institute of Arthritis, Musculoskeletal and Skin Diseases (NIH-NIAMS) grants R01 AR-46208 and 2P01AR052915-06A1, and by University Clinical Research Grants M01-RR-02558 (The University of Texas Health Science Center at Houston.
Disclosures
None.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Reveille, J.D. An update on the contribution of the MHC to as susceptibility. Clin Rheumatol 33, 749–757 (2014). https://doi.org/10.1007/s10067-014-2662-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10067-014-2662-7