Abstract
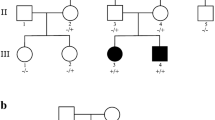
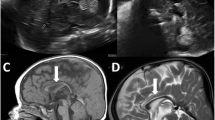
Hindbrain malformations with predominant cerebellar involvement have many causes including chromosomal disorders, specific genetic syndromes, and prenatal disruptions. The combination of a hindbrain malformation and myoclonic epilepsy is rare. Using exome sequencing in a consanguineous family, we identified a homozygous genomic deletion of 1770 bp within the INPP4A gene in a patient with myoclonic epilepsy, microcephaly, and atrophy of the inferior vermis and cerebellum. INPP4A participates in the excitatory glutamate signaling pathway and is essential for the degradation of phosphatidylinositol (3,4)-bisphosphate. Glutamatergic signaling is important for hindbrain development and is implicated in the pathogenesis of epilepsy, as well as excitotoxic cell death. Indeed, excessive glutamatergic stimulation was previously reported in INPP4A knockout mice. Our data adds a new etiology to the spectrum of hindbrain malformations in human, and when presented with myoclonic epilepsy may lead to the clinical suspicion of INPP4A defect. The present report further underscores the importance of phosphoinositides for the development of the inferior cerebellum and vermis.



Similar content being viewed by others
References
Doherty D, Millen KJ, Barkovich AJ (2013) Midbrain and hindbrain malformations: advances in clinical diagnosis, imaging, and genetics. Lancet Neurol 12:381–393
Edvardson S, Cinnamon Y, Jalas C, Shaag A, Maayan C, Axelrod FB, Elpeleg O (2012) Hereditary sensory autonomic neuropathy caused by a mutation in dystonin. Ann Neurol 71:569–572
Schwarz JM, Rödelsperger C, Schuelke M, Seelow D (2010) MutationTaster evaluates disease-causing potential of sequence alterations. Nat Methods 7:575–576
Nystuen A, Legare ME, Shultz LD, Frankel WN (2001) A null mutation in inositol polyphosphate 4-phosphatase type I causes selective neuronal loss in weeble mutant mice. Neuron 32:203–212
Dzubay JA, Otis TS (2002) Climbing fiber activation of metabotropic glutamate receptors on cerebellar Purkinje neurons. Neuron 36:1159–1167
Berridge MJ, Dawson RM, Downes CP, Heslop JP, Irvine RF (1983) Changes in the levels of inositol phosphates after agonist-dependent hydrolysis of membrane phosphoinositides. Biochem J 212:473–482
Berridge MJ, Bootman MD, Roderick HL (2003) Calcium signalling: dynamics, homeostasis and remodelling. Nat Rev Mol Cell Biol 4:517–529
Norris FA, Auethavekiat V, Majerus PW (1995) The isolation and characterization of cDNA encoding human and rat brain inositol polyphosphate 4-phosphatase. J Biol Chem 270:16128–16133
Sasaki J, Kofuji S, Itoh R et al (2010) The PtdIns(3,4)P(2) phosphatase INPP4A is a suppressor of excitotoxic neuronal death. Nature 465:497–501
Barkovich AJ, Millen KJ, Dobyns WB (2009) A developmental and genetic classification for midbrain-hindbrain malformations. Brain 132:3199–3230
Khazipov R, Esclapez M, Caillard O et al (2001) Early development of neuronal activity in the primate hippocampus in utero. J Neurosci 21:9770–9781
Acknowledgments
Authors thank the patient’s family for participating in this work. This study was supported in part by Karl Kahane Foundation.
Ethical standards statement
The experiments comply with the current laws of Israel.
Conflict of interest
All the coauthors declare that they have no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Supplementary Table 1
(DOCX 14 kb)
Rights and permissions
About this article
Cite this article
Sheffer, R., Bennett-Back, O., Yaacov, B. et al. Hindbrain malformation and myoclonic seizures associated with a deleterious mutation in the INPP4A gene. Neurogenetics 16, 23–26 (2015). https://doi.org/10.1007/s10048-014-0428-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10048-014-0428-7