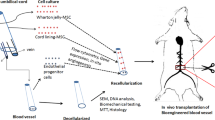
Abstract
In the tissue engineering research field, the presence of circulating endothelial progenitor cells (EPCs) in the peripheral blood of adults represents a promising cell source to grow autologous endothelium on blood-contacting devices. Materials functionalized with EPC-specific molecules are an intriguing strategy to induce the homing and differentiation of the trapped EPCs into endothelial cells to generate a non-thrombogenic surface. Although the EPCs have been identified in adult peripheral blood about 30 years ago, in the subsequent literatures, the term “EPCs” has encompassed different cell populations with a mixed ability to contribute to the formation of blood vessels. This confusion is due to limited functional characterization of “EPCs”, an improper nomenclature, and the poorly standardized protocols. This review will screen the literature about “EPCs” to propose a specific nomenclature, phenotypic characterization, and, eventually, a protocol to perform reliable experiments

Similar content being viewed by others
References
Potente M, Gerhardt H, Carmeliet P. Basic and therapeutic aspects of angiogenesis. Cell. 2011;146:873–87.
Gerhardt H, Golding M, Fruttiger M, Ruhrberg C, Lundkvist A, Abramsson A, et al. VEGF guides angiogenic sprouting utilizing endothelial tip cell filopodia. J Cell Biol. 2003;161:1163–77.
Valaitis J, McGrew EA, McGrath RGRS. Capillary cell clusters inperipheral blood of postoperative cancerand noncancer patients. Acta Cytol. 1968;12:439.
Stump MM, Jordan JL Jr, DeBakey MEHB. Endothelium grownfrom circulating blood on isolated intra-vascular Dacron hub. Am J Pathol. 1963;43:361–7.
Mackenzie JR, Hackett M, Topuzlu CTD. Origin of arterial pros-thesis lining from circulating blood cells. Arch Surg. 1968;97:879–85.
Asahara T, Murohara T, Sullivan A, Silver M, van der Zee R, Li T, et al. Isolation of putative progenitor endothelial cells for angiogenesis. Science. 1997;275:964–7.
Testa U, Saulle E, Castelli G, Pelosi E. Endothelial progenitor cells in hematologic malignancies. Stem Cell Investig. 2016;3:26–26.
Tomanek RJ, Schatteman GC. Angiogenesis: new insights and therapeutic potential. Anat Rec. 2000;261:126–35.
Hur J, Yoon CH, Kim HS, Choi JH, Kang HJ, Hwang KK, et al. Characterization of two types of endothelial progenitor cells and their different contributions to neovasculogenesis. Arterioscler Thromb Vasc Biol. 2004;24:288–93.
Hirschi KK, Ingram DA, Yoder MC. Assessing identity, phenotype, and fate of endothelial progenitor cells. Arterioscler Thromb Vasc Biol. 2008;28:1584–95.
Medina RJ, O’Neill CL, Humphreys MW, Gardiner TA, Stitt AW. Outgrowth endothelial cells: characterization and their potential for reversing ischemic retinopathy. Investig Ophthalmol Vis Sci. 2010;51:5906–13.
Siemerink MJ, Klaassen I, Vogels IMC, Griffioen AW, Van Noorden CJF, Schlingemann RO. CD34 marks angiogenic tip cells in human vascular endothelial cell cultures. Angiogenesis. 2012;15:151–63.
Cheng C-C, Chang S-J, Chueh Y-N, Huang T-S, Huang P-H, Cheng S-M, et al. Distinct angiogenesis roles and surface markers of early and late endothelial progenitor cells revealed by functional group analyses. BMC Genom. 2013;14:182.
Bou Khzam L, Bouchereau O, Boulahya R, Hachem A, Zaid Y, Abou-Saleh H, et al. Early outgrowth cells versus endothelial colony forming cells functions in platelet aggregation. J Transl Med BioMed Central. 2015;13:353.
Joo HJ, Song S, Seo HR, Shin JH, Choi SC, Park JH, et al. Human endothelial colony forming cells from adult peripheral blood have enhanced sprouting angiogenic potential through up-regulating VEGFR2 signaling. Int J Cardiol. 2015;197:33–43. https://doi.org/10.1016/j.ijcard.2015.06.013.
Hebbel RP. Blood endothelial cells: utility from ambiguity. J Clin Investig. 2017;127:1613–5.
Medina RJ, Barber CL, Sabatier F, Dignat-George F, Melero-Martin JM, Khosrotehrani K, et al. Endothelial progenitors: a consensus statement on nomenclature. Stem Cells Transl Med. 2017;6(5):1316–20.
Shaik MV, Shaik M, Gangapatnam S. Analysis of endothelial progenitor subpopulation cells, oxidative DNA damage, and their role in coronary artery disease. Biomed Biotechnol Res J. 2018;2:136–41.
Friedrich EB, Walenta K, Scharlau J, Nickenig G, Werner N, Cd CD, et al. CD34−/CD133+/VEGFR-2+ endothelial progenitor cell subpopulation with potent vasoregenerative capacities. Circ Res. 2006;98:20–5.
Peichev M, Naiyer A, Pereira D, Zhu Z, Lane W, Williams M, et al. Expression of VEGFR-2 and AC133 bycirculating human CD34(+) cells identifies a population of functional endothelial precursors. Blood. 2000;95:952–8.
Avci-Adali M, Perle N, Ziemer G, Wendel HP. Current concepts and new developments for autologous in vivo endothelialisation of biomaterials for intravascular applications. Eur Cell Mater. 2011;21:157–76.
Formiga F, Pelacho B, Garbayo E, Abizanda G, Gavira J, Simon-Yarza T, et al. Sustained release of VEGF through PLGA microparticles improves vasculogenesis and tissue remodeling in an acute myocardial ischemia-reperfusion model. J Control Release. 2010;47:30–7.
Hauser S, Jung F, Pietzsch J. Human endothelial cell models in biomaterial research. Trends Biotechnol. 2017;35:1–13.
Salven P, Mustjoki S, Alitalo R, Alitalo K, Rafii S. VEGFR-3 and CD133 identify a population of CD34+ lymphatic/vascular endothelial precursor cells. Blood. 2003;101:168–72.
Castelli G, Parolini I, Cerio AM, D’Angiò A, Pasquini L, Carollo M, et al. Conditioned medium from human umbilical vein endothelial cells markedly improves the proliferation and differentiation of circulating endothelial progenitors. Blood Cells Mol Dis. 2016;61:58–655.
Parham KA, Pitson SM, Bonder CS. Regulation of EPCs: the gateway to blood vessel formation. New J Sci. 2014;2014:972043. https://doi.org/10.1155/2014/972043.
Acknowledgements
We gratefully acknowledge the financial support of the S-innovation Research Program for the “Development of the biofunctional materials for realization of innovative medicine”, Japan Agency for Medical Research and Development (AMED).
Author information
Authors and Affiliations
Contributions
MCM: interpretation and manuscript writing. TY: interpretation and manuscript writing. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors have no conflict of interest to declare.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Munisso, M.C., Yamaoka, T. Circulating endothelial progenitor cells in small-diameter artificial blood vessel. J Artif Organs 23, 6–13 (2020). https://doi.org/10.1007/s10047-019-01114-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10047-019-01114-6