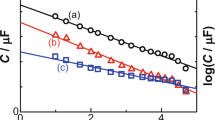
Abstract
Terminology of electrodes and electrode materials used in supercapacitors as well as naming of electrode processes and devices prepared with these electrodes is confusing and rather unregulated. Consequently, misunderstanding in communication about research and development is somehow matched with an incomplete understanding of the reasons of the observed capacitive, pseudocapacitive, or Faradaic behavior. Observed and investigated phenomena relevant for supercapacitor electrodes are briefly reviewed and explained in terms of electrode processes and interfacial phenomena. Further research possibly useful in more fundamental understanding and rational improvement of materials is proposed.








Similar content being viewed by others
Notes
C DL is different from the integral double layer capacitance C D = dQ/(E − E pzc); the latter is invoked only infrequently. Both quantities are related according to: C DL = (E − E pzc) (dC D/dE) + C D
The terms supercapacitor™ (as well as ultracap/ultracapacitor) or abbreviated supercap (SC) seemingly lack a generally accepted, proper definition. At first glance, it appears sufficient to assume that capacitors based on the capacitive properties of the electrochemical double layer instead of a dielectric material like Al2O3 or Ta2O5 showing huge capacities are correctly called supercapacitors. Temporarily, the latter term was trademarked (from August 1978 on) to NEC Corporation; currently, this protection has apparently expired. The acronym SC seems to be too short to enable immediate identification. Acronyms like ES for electrochemical supercapacitor or FS for Faradaic supercapacitor do nothing beyond enlarging the confusion. Recently, this device wherein purely electrostatic charge storage in the double layer is operative has been frequently called electrochemical double layer capacitor (EDLC). Thus, it appears to be reasonable to call devices, wherein charge storage is based both on electrostatic charge separation (like in an EDLC) and on Faradaic redox processes (pseudocapacity) supercapacitors. Because of the combination of these fundamentally different charge storage mechanisms, these devices are also sometimes called hybrids—adding further to the confusion. A device wherein two effects or mechanisms are utilized is not necessarily a hybrid one—when both effects as in most SCs act only in addition to each other. In the present report, supercapacitors are such “hybrid devices”; the term ultracapacitor is not used at all. Its use to designate only those devices employing pseudocapacitances seems to be a loosing proposition [A. Burke, J. Power Sources 91, 37 (2000)]. The statement that Conway coined the term supercapacitor in 1991 is apparently erroneous. The rich collection of terms—some of them presumably protected by trademarks—does not help really: APowerCap, BestCap, BoostCap, CAP-XX, DLCAP, EneCapTen, EVerCAP, DynaCap, Faradcap, GreenCap, Goldcap, HY-CAP, Kapton capacitor, Supercapacitor, SuperCap, PAS Capacitor, PowerStor, PseudoCap, etc.
In this study, Na2SO4 was used as an electrolyte; other alkali ions can be used instead also.
Assignment of a species to any of these classes was based on spectroscopic evidence certainly not applicable here; more recently observations from CV have been utilized at least for a rough classification.
Simulation was done with software Polar 4.1 for Windows, Dr. Huang Pty Ltd., Sydney, Australia.
This state is frequently and erroneously called the reduced one. A reduced one would correctly be reached by reduction of the neutral one (the leucoemeraldine state). This has not been observed so far with PANI.
References
Nicholson RS, Shain I (1964) Theory of stationary electrode polarography. Single scan and cyclic methods applied to reversible, irreversible, and kinetic systems. Anal Chem 36:706–723
Nicholson RS, Shain I (1965) Theory of stationary electrode polarography for a chemical reaction coupled between two charge transfers. Anal Chem 37:178–190
Nicholson RS (1965) Theory and application of cyclic voltammetry for measurement of electrode reaction kinetics. Anal Chem 37:1351–1355
Gosser DK Jr (1993) Cyclic Voltammetry. VCH, New York
Noel M, Vasu KI (1990) Cyclic voltammetry and the frontiers of electrochemistry. Oxford & IBH Publishing Co., New Delhi
Bard AJ, Faulkner LR (2001) Electrochemical methods. Wiley, New York
Conway BE, Gileadi E (1962) Trans Faraday Soc 58:2493–2509
Conway BE, Pell WG (2003) J Solid State Electrochem 7:637–644
Kurzweil P (2009) Electrochemical capacitors, encyclopedia of electrochemical power sources Vol. 3. In: Garche J, Dyer CK, Moseley PT, Ogumi Z, Rand DAJ, Scrosati B (eds) . Elsevier, Amsterdam, pp. 596–606
Dubal DP, Holze R (2014) Pure&Appl Chem 86:611–632
Dubal DP, Wu Y, Holze R (2016) ChemTexts 2:13
Dubal DP, Wu Y, Holze R (2015) Bunsen-Magazin 17:216–227
Kurzweil P (2009) Capacitors: electrochemical double-layer capacitors, encyclopedia of electrochemical power sources Vol. 1. In: Garche J, Dyer CK, Moseley PT, Ogumi Z, DAJ R, Scrosati B (eds) . Elsevier, Amsterdam, pp. 607–633
Kurzweil P (2009) Capacitors: electrochemical double-layer capacitors: carbon materials, encyclopedia of electrochemical power sources Vol. 1. In: Garche J, Dyer CK, Moseley PT, Ogumi Z, DAJ R, Scrosati B (eds) . Elsevier, Amsterdam, pp. 634–648
Borchardt L, Oschatz M, Kaskel S (2014) Mater Hor 1:157–168
Zhang LL, Gu Y, Zhao XS (2013) J Mater Chem A 1:9395–9408
Lee J, Kim J, Hyeon T (2006) Adv Mater 18:2073–2094
Liu S, Sun S, You XZ (2014) Nanoscale 6:2037–2045
Su DS, Schlögl R (2010) ChemSusChem 3:136–168
Beguin F, Presser V, Balducci A, Frackowiak A (2014) Adv Mater 26:2219–2251
Davies A, Yu A (2011) Can J Chem Engin 89:1342–1357
Ghosh A, Lee YH (2012) ChemSusChem 5:480–499
Zhang LL, Zhao XS (2009) Chem Soc Rev 38:2520–2531
Frackowiak E, Abbas Q, Beguin F (2013) J Energy Chem 22:226–240
Jiang H, Lee PS, Li C (2013) Energy Environm Sci 6:41–53
Dutta S, Bhaumik A, Wu KCW (2014) Energy Environm Sci 7:3574–3592
Bose S, Kuila T, Mishra AK, Rajasekar R, Kim NH, Lee JH (2012) J Mater Chem 22:767–784
Conway BE (1999) Electrochemical supercapacitors: scientific fundamentals and technological applications. Springer, New York
Galizzioli D, Tantardini F, Trasatti S (1974) J Appl Electrochem 4:57–67
Toupin M, Brousse T, Belanger D (2004) Chem Mater 16:3184–3190
Hu CC, Tsou TW (2002) Electrochem Commun 4:105–109
Chun SE, Pyun SI, Lee GJ (2006) Electrochim Acta 51:6479–6486
Ghaemi M, Ataherian F, Zolfaghari A, Jafari SM (2008) Electrochim Acta 53:4607–4614
Damaskin BB, Petrii OA, Batrakov VV (1975) Adsorption organischer Verbindungen an Elektroden. Akademie-Verlag, Berlin
Brousse T, Belanger D, Long JW (2015) J Electrochem Soc 162:A5185–A5189
Robin MB, Day P (1967) Adv Inorg Radiochem 10:247–422
Leschke M, Lang H, Holze R (2003) J Solid State Electrochem 7:518–524
Weiß T, Lang H, Holze R (2002) J Electroanal Chem 533:127–133
Stein T, Lang H, Holze R (2002) J Electroanal Chem 520:163–167
Leschke M, Lang H, Holze R (2003) Electrochim Acta 48:919–924
Hubbard AT, Anson F (1970) Electroanalytical chemistry. A series of advances. In: Bard AJ (ed) , vol 4. Marcel Dekker, New York, pp. 129–214
Lane RF, Hubbard AT (1978) J Phys Chem 77:1401–1410
Laviron E (1979) J Electroanal Chem 100:263–270
Angerstein-Kozlowska H, Klinger J, Conway BE (1977) J Electroanal Chem 75:45–60
Angerstein-Kozlowska H, Klinger J, Conway BE (1977) J Electroanal Chem 75:61–75
Schreurs J, Barendrecht E (1984) Rec Trav Chim Pays-Bas 103:205–215
Burke LD, Morrissey JA (1994) J Electrochem Soc 141:2361–2368
Burke LD (1994) Electrochim Acta 39:1841–1848
Burke LD, Casey JK (1992) Electrochim Acta 37:1817–1829
Burke LD, Buckley DT, Morrissey JA (1994) Analyst 119:841–845
Burke LD, Casey JK, Cunnane VJ, Murphy OJ, Twomey TAM (1985) J Electroanal Chem 189:353–362
Young MJ, Holder AM, George SM, Musgrave CB (2015) Chem Mater 27:1172–1180
Holze R (2001) Handbook of advanced electronic and photonic materials and devices, Vol. 8. In: Nalwa HS (ed) . Academic Press, San Diego, pp. 209–301
Holze R (2001) Advanced functional molecules and polymers, Vol. 2. In: Nalwa HS (ed) . Gordon&Breach, Amsterdam, pp. 171–221
Cui CQ, Ong LH, Tan TC, Lee JY (1993) J Electroanal Chem 346:477–482
Brandl V, Holze R (1997) Ber Bunsenges Phys Chem 101:251–256
Heinze J (1990) Topics in current chemistry Vol. 152. Springer, Berlin Heidelberg, pp. 1–45
for an earlier overview see: Heinze J, Mortensen J, Störzbach M (1987) Springer series in solid state Sci. In: Kuzmany H, Roth S (eds) Electronic properties of conjugated polymers, vol 76. Springer-Verlag, Berlin, p, 385–390
Bull RA, Bard AJ, Fan FRF (1982) J Electrochem Soc 129:1009–1015
Diaz AF, Castillo JI, Logan JA, Lee WA (1981) J Electroanal Chem 129:115–132
Feldberg SW (1984) J Am Chem Soc 106:4671–4674
Jakobs RCM, Janssen LJJ, Barendrecht E (1984) Rec Trav Chim Pays-Bas 103:275–281
Bandeira MCE, Holze R (2006) Microchim Acta 156:125–131
Yan J, Yang L, Cui M, Wang X, Chee KJ, Nguyen VC, Kumar V, Sumboja A, Wang M (2014) Adv Energ Mater 4:400781
Heinze J, Störzbach M, Mortensen J (1987) Ber Bunsenges Phys Chem 91:960–967
Acknowledgements
Preparation of this note has been supported by a travel grant from Deutsche Forschungsgemeinschaft. The generous hospitality of Amartya Mukhopadhyay, Department of Metallurgical Engineering and Materials Science, IIT Bombay, India, with its stimulating environment and inspiring discussions is gratefully appreciated.
Author information
Authors and Affiliations
Corresponding author
Additional information
Dedicated to Chandrakant D. Lokhande on the occasion of his 60th birthday in recognition of his major contributions to thin film science: from preparation to applications.
Rights and permissions
About this article
Cite this article
Holze, R. From current peaks to waves and capacitive currents—on the origins of capacitor-like electrode behavior. J Solid State Electrochem 21, 2601–2607 (2017). https://doi.org/10.1007/s10008-016-3483-1
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10008-016-3483-1