Abstract
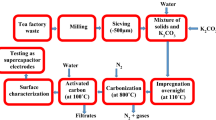
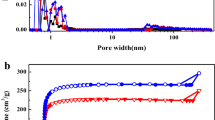
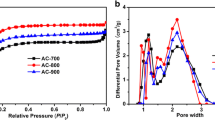
In this paper, activated carbon materials were synthesized from pomegranate rind through carbonization and alkaline activation processes. The effects of pyrolytic temperature on the textual properties and electrochemical performance were investigated. The surface area of the activated carbon can reach at least 2200 m2 g−1 at different pyrolytic temperatures. It was found that, at the range of 600–900 °C, decreasing the carbonization temperature leads to the increase of t-plot micropore area, t-plot micropore volume, and capacitance. Further decreasing the carbonization temperature to 500 °C also leads to the increase of t-plot micropore area and t-plot micropore volume, but the capacitance is slightly poorer. The activated carbon carbonized at 600 °C and activated at 800 °C possesses very high specific area (2931 m2 g−1) and exhibits very high capacitance (∼268 F g−1 at 0.1 A g−1 and ∼242 F g−1 at 1 A g−1). There is no capacitance fading after 2000th cycle.








Similar content being viewed by others
References
Hao P, Zhao Z, Tian J, Li H, Sang Y, Yu G, Cai H, Liu H, Wong CP, Umar A (2014) Nanoscale 6:12120–12129
Biswas S, Drzal LT (2010) ACS Appl Mater Interfaces 2:2293–2300
Tamailarasan P, Ramaprabhu S (2012) J Phys Chem C 116:14179–14187
Zhang JT, Zhao XS (2012) J Phys Chem C 116:5420–5426
Han J, Zhang LL, Lee S, Oh J, Lee KS, Potts JR, Ji J, Zhao X, Ruoff RS, Park S (2013) ACS Nano 7:19–26
Kim T, Jung G, Yoo S, Suh KS, Ruoff RS (2013) ACS Nano 7:6899–6905
Xiong Z, Liao C, Wang X (2014) J Mater Chem A 2:19141–19144
Kalugin ON, Chaban VV, Loskutov VV, Prezhdo OV (2008) Nano Lett 8:2126–2130
Chen X, Wang HW, Yi H, Wang XF, Yan XR, Guo ZH (2014) J Phys Chem C 118:8262–8270
Subramanian V, Luo C, Stephan AM, Nahm KS, Thomas S, Wei B (2007) J Phys Chem C 111:7527–7531
Liu X, Zheng M, Xiao Y, Yang Y, Yang L, Liu Y, Lei B, Dong H, Zhang H, Fu H (2013) ACS Appl Mater Interfaces 5:4667–4677
Liu X, Zhou L, Zhao Y, Bian L, Feng X, Pu Q (2013) ACS Appl Mater Interfaces 5:10280–10287
Qian H, Kucernak AR, Greenhalgh ES, Bismarck A, Shaffer MS (2013) ACS Appl Mater Interfaces 5:6113–6122
Sun H, He W, Zong C, Lu L (2013) ACS Appl Mater Interfaces 5:2261–2268
Wang H, Chen Z, Liu HK, Guo Z (2014) RSC Adv 4:65074–65080
Wang C, Wang Y (2013) ACS Nano 7:11156–11165
Dai YH, Jiang H, Hu YJ, Fu Y, Li CZ (2014) Ind Eng Chem Res 53:3125–3130
Kim C, Choi Y-O, Lee W-J, Yang K-S (2004) Electrochim Acta 50:883–887
Lin J-H, Ko T-H, Lin Y-H, Pan C-K (2009) Energy Fuel 23:4668–4677
Barranco V, Lillo-Rodenas MA, Linares-Solano A, Oya A, Pico F, Ibanez J, Agullo-Rueda F, Amarilla JM, Rojo JM (2010) J Phys Chem C 114:10302–10307
Feng S, Li W, Wang J, Song Y, Elzatahry AA, Xia Y, Zhao D (2014) Nanoscale 6:14657–14661
Balathanigaimani MS, Shim WG, Lee MJ, Kim C, Lee JW, Moon H (2008) Electrochem Commun 10:868–871
Liu MC, Kong LB, Lu C, Li XM, Luo YC, Kang L (2012) RSC Adv 2:1890–1896
Bhattacharjya D, Yu JS (2014) J Power Sources 262:224–231
Wang Y, Zhang L, Wang H, Wang J, Yu W, Peng B, Yang Z, Chai L (2014) J Solid State Electrochem 18:3209–3214
Zhang L, Wang Y, Peng B, Yu W, Wang H, Wang T, Deng B, Chai L, Zhang K, Wang J (2014) Green Chem 16:3926–3934
Wang H, Li X, Chai L, Zhang L (2015) Chem Commun 51:8524–8527
Wang S, Ren Z, Li J, Ren Y, Zhao L, Yu J (2014) RSC Adv 4:31300
Rufford TE, Hulicova-Jurcakova D, Khosla K, Zhu Z, Lu GQ (2010) J Power Sources 195:912–918
Ismanto AE, Wang S, Soetaredjo FE, Ismadji S (2010) Bioresour Technol 101:3534–3540
He X, Ling P, Yu M, Wang X, Zhang X, Zheng M (2013) Electrochim Acta 105:635–641
Biswal M, Banerjee A, Deo M, Ogale S (2013) Energy Environ Sci 6:1249–1259
Raymundo-Piñero E, Cadek M, Béguin F (2009) Adv Funct Mater 19:1032–1039
Groen JC, LAA P, Perez-Ramirez J (2003) Microporous Mesoporous Mater 60:1–17
Elazari R, Salitra G, Garsuch A, Panchenko A, Aurbach D (2011) Adv Mater 23:5641–5644
Puthusseri D, Aravindan V, Anothumakkool B, Kurungot S, Madhavi S, Ogale S (2014) Small 10:4395–4402
Chmiola J, Yushin G, Gogotsi Y, Portet C, Simon P, Taberna PL (2006) Science 313:1760–1763
Kalpana D, Cho SH, Lee SB, Lee YS, Misra R, Renganathan NG (2009) J Power Sources 190:587–591
Acknowledgments
The authors acknowledge the financial supports from Excellent Youth Foundation (13JJ1003) of Hunan Provincial Science and Technology Department, National Natural Science Foundation of China (Grant no. 51404304), and Natural Science Foundation of Hunan Province (14JJ2001) and other supports which are from the Engineering Research Centre of Advanced Battery Materials, the Ministry of Education, China.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
ESM 1
(DOCX 115 kb)
Rights and permissions
About this article
Cite this article
Qin, F., Zhang, K., Li, J. et al. Pomegranate rind-derived activated carbon as electrode material for high-performance supercapacitors. J Solid State Electrochem 20, 469–477 (2016). https://doi.org/10.1007/s10008-015-3064-8
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10008-015-3064-8