Abstract
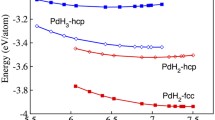
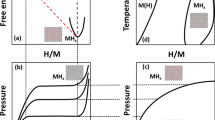
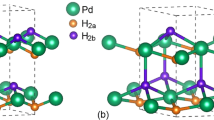
Palladium hydrides (Pd-H) research is an important topic in materials research with many practical industrial applications. The complex behavior of the Pd-H alloy system such as phase miscibility gap, however, presents a huge challenge for developing reliable computational models. The embedded atom method (EAM) offers an advantage of computational efficiency and being suited to the metal-hydride system. We propose a new EAM interatomic potential for the complete mathematical modeling of palladium hydride. The present interatomic potential well predicts the lattice constant, cohesive energy, bulk modulus, other elastic constants, and stable alloy crystal structures during molecular dynamics simulations. The phase miscibility gap is also accurately predicted for the Pd-H system using the present potential. To our knowledge, only two Pd-H EAM potentials were used for predicting the phase miscibility gap for the PdH system. The predicted values from these works, however, considerably deviated from the experimental result, which hinders further application to the palladium hydride system. The present potential is reliably accurate and can be used to study the Pd-H system with its compete description of the mathematical formalism.










Similar content being viewed by others
References
Graham T (1866) On the absorption and dialytic separation of gases by colloid septa. Philos Trans R Soc London 156:399–439
Mueller WM, Blackledge JP, Libowitz GG (1968) Metal hydrides. Academic Press, New York
Povel R, Feucht K, Gelse W, Withalm G (1989) Hydrogen fuel for motorcars. Interdiscip Sci Rev 14:365
Johnson JW as quoted by Troiano AR (1974) Bernstein IM, Thompson AW (eds.) Hydrogen in metals. American Society of Metals, pp 3–5
Anisimkin VI, Kotelyanskii IM, Verardi P, Verona E (1995) Elastic properties of thin film palladium for surface-acoustic-wave (SAW) sensors. Sens Actuators B 23:203–208
Nygren LA, Leisure RG (1988) Elastic constants of α-phase over the temperature range 4–300 K. Phys Rev B 37:6482
Hsu DK, Leisure RG (1979) Elastic constants of palladium and ρ-phase palladium hydride between 4 and 300 K. Phys Rev B 20:1339
Frieske H, Wicke E (1973) Magnetic susceptibility and equilibrium diagram of PdHn Ber. Bunsenges Phys Chem 77:48–52
Wicke E, Nernst GH (1964) Phase diagram and thermodynamic behavior of the palladium-hydrogen and of the palladium-deuterium system at normal temperature; H/D separation effect. Ber Bunsenges Phys Chem 68:224–235
Lässer R, Klatt KH (1983) Solubility of hydrogen isotopes in palladium 1983. Phys Rev 28:748
Wicke E, Blaurock J (1987) New experiments on and interpretations of hysteresis effects of Pd-D2 and Pd-H2. J Less-Common Met 130:351–363
Lässer R, Powell GL (1986) Solubility of H, D, and T in Pd at low concentrations. Phys Rev B 34:578
Chen WC, Heuser BJ (2000) Solubility and kinetic properties of deuterium in single crystal Pd. J Alloys Comp 312:176–180
McNicholl E-A, Lewis FA (1990) The hydride phase miscibility gap in palladium-rare earth alloys, Platin. Met Rev 34:81–84
Zhou XW, Zimmerman JA, Wong BM, Hoyt JJ (2008) An embedded-atom method interatomic potential for Pd-H alloy. J Mater Res 23:704–718
Wolf RJ, Lee MW, Davis RC, Fay PJ, Ray JR (1993) Pressure–composition isotherms for palladium hydride. Phys Rev B 48:12415
Daw MS, Baskes MI (1983) Semiempirical, quantum mechanical calculation of hydrogen embrittlement in metals. Phys Rev Lett 50:1285
Foiles SM, Baskes MI, Daw MS (1986) Embedded-atom-method functions for the fcc metals Cu, Ag, Au, Ni, Pd, Pt, and their alloys. Phys Rev B 33(12):7983
Hijazi IA, Park YH (2009) Consistent analytic embedded atom potential for face-centered cubic metals and alloys. J Matter Sci Technol 25:835–846
Foiles S.M. and Hoyt J.J. 2001. Computer simulation of bubble growth in metals due to He (Sandia National Laboratories)
Rose JH, Smith JR, Guinea F, Ferrante J (1984) Universal features of the equation of state of metals. Phys Rev B 29(6):2963
Morse P (1929) Diatomic molecules according to the wave mechanics. II. Vibrational levels. Phys Rev 34:57–64
Matsubara T, Matsushita E (1984) Further comment on isotope effect in hydrogen bonded crystal. Progr Theor Phys 67:209–211
Zaluzniak V, Zolotov O (2015) Towards a universal embedded atom method interatomic potential for pure metals. Math Phys 8:230–249
Puska J, Nieminen RM, Manninen M (1981) Atoms embedded in an electron gas: immersion energies. Phys Rev B 24:3037
Bambakidis G (1981) Metal hydrides. Plenum, New York
Ilawe NV, Zimmerman JA, Wong BM (2015) Breaking badly: DFT-D2 gives sizeable errors for tensile strengths in palladium-hydride solids. J Chem Theory Comput 11:5426–5435
Schwarz RB, Bach HT, Harms U, Tuggle D (2005) Elastic properties of Pd–hydrogen, Pd–deuterium, and Pd–tritium single crystals. Acta Mater 53:569–580
Schirber JE, Morosin B (1975) Lattice constants of b-PdHx and b-PdDx with x near 1.0. Phys Rev B 12:117
Sakamoto Y, Yuwasa K, Hirayama K (1982) X-ray investigation of the absorption of hydrogen by several palladium and nickel solid solution alloys. J Less-Comm Metals 88:115
Jacobs MHG, Oonk HAJ (2006) The calculation of ternary miscibility gaps using linear contributions method: problems, benchmark systems and an application to (K, Li, Na)Br. Comput Coupling Phase Diag Thermochem 30:185–190
Emelianko M, Liu Z-K, Dy Q (2006) A new algorithm for the automation of phase diagram calculation. Comput Mater Sci 35:61–74
Lewis FA (1967) The palladium-hydrogen system. Academic, New York
Alefeld G, Völkl J (1978) Hydrogen in metals. Springer, Heidelberg
Antonov VE (2008) Phase transformations, crystal and magnetic structures of high-pressure. J All Comp 330–332:110–116
Author information
Authors and Affiliations
Corresponding author
Appendix: EAM potential functions for palladium
Appendix: EAM potential functions for palladium
Note the pair potential function is truncated at r cut = 5.35 Å
The density function f Pd is also truncated at r cut = 5.35 Å. A function of distance g(r) is then modified to
with n = 20.
The embedding function F is fitted as follows:
Rights and permissions
About this article
Cite this article
Park, Y.H., Hijazi, I. Development of physics based analytical interatomic potential for palladium-hydride. J Mol Model 23, 108 (2017). https://doi.org/10.1007/s00894-017-3288-x
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00894-017-3288-x