Abstract
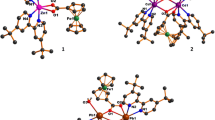
Three mononuclear coordination complexes of Co(II), Ni(II), and Cu(II) have been synthesized from 2,4-dichloro-6-{(E)-[(5-chloro-2-sulfanylphenyl)imino]methyl}phenol ligand (H 2 L) obtained by simple condensation reaction of 3,5-dichloro-2-hydroxybenzaldehyde and 2-amino-4-chlorobenzenethiol and characterized by elemental analysis, spectral (FT-IR, electronic, and 1H-NMR), molar conductance, thermal, SEM, PXRD, and fluorescence studies. The PXRD analysis and SEM-EDX micrographs show the crystalline nature of complexes. The domain size and the lattice strain of synthesized compounds have been determined according to Williamson–Hall plot. TG of the synthesized complexes illustrates the general decomposition pattern of the complexes. The ligand exhibits an interesting fluorescence property which is suppressed after complex formation. The Co(II) complex adopted a distorted octahedral configuration while Ni(II) and Cu(II) complexes showed square planar geometry around metal center. The geometry optimization, HOMO-LUMO, molecular electrostatic potential map (MEP), and spin density of synthesized compounds have been performed by density functional theory (DFT) method using B3LYP/6-31G and B3LYP/LANL2DZ as basis set.

Three new coordination complexes of Co(II), Ni(II) and Cu(II) with 2,4-dichloro-6-{(E)-[(5-chloro-2 sulfanylphenyl)imino]methyl}phenol Schiff base ligand.










Similar content being viewed by others
References
Marusak RA , Doan K, Cummings SD (2007) Integrated approach to coordination chemistry an inorganic laboratory guide. Wiley, New York, pp 22–28
Vigato PA, Tamburini S (2004) Coord Chem Rev 248:1717–2128
Vigato PA, Tamburini S (2008) Coord Chem Rev 252:1871–1995
Vigato PA, Peruzzo V, Tamburini S (2012) Coord Chem Rev 256:953–1114
Nelson W, Wallis, Sue C (1974) Cummings Inorg Chem 13:991–994
Cincic D, Kaitner B (2011) Cryst Eng Comm 13:4351–4357
Mishra AP, Jain R (2013) Proc Natl Acad Sci A Phys Sci 83(3):213–223
Uddin AJ, Räisänen MT, Martin N, Markku L, Timo R (2012) Inorg Chim Acta 384:275–280
Bhardwaj VK, Hundal MS, Corbella M, Gomez V, Hundal G (2012) Polyhedron 38:224–234
Rosette M, Roat-Malone (2007) Bioinorganic chemistry a short course, second edn. Wiley, New York, pp 1–28
Dong X, Li Y, Li Z, Cui Y, Zhu H (2012) J Inorg Biochem 108:22–29
Chen W et al (2010) Euro J Med Chem 45:4473–4478
Pelosi G et al (2010) J Med Chem 53:8765–8769
Vanco J et al (2008) J Inorg Biochem 102(4):595–605
Katherine H (2006) Thompson and Chris Orvig. Dalton Trans 761–764. doi:10.1039/b513476e
Shukla S, Mishra AP (2014) Arabian J Chem doi:10.1016/j.arabjc.2014.08.020
Sarah B, Creaven B (2010) Dalton Trans 39:10854–10865
Pandey R et al (2011) Inorg Chem 50:3189–3197
Cozzi PG et al (2003) New J Chem 27:692–697
Strianese M, Milione S, Bertolasi V, Pellecchia C (2013) Inorg Chem 52:11778–11786
Trujillo A et al (2010) Inorg Chem 49:2750–2764
Liu C-G, Guan X-H, Su Z-M (2011) J Phys Chem C 115:6024–6032
Dehno A, Nikookar KM, Fejfarova K, Dusek M (2014) J Mol Str 1071:6–10
Gutiérrez A, Felisa Perpiñán M, Sánchez AE, Carmen Torralba M, Rosario Torres M (2012) Polyhedron 44:165–173
Banerjee S et al (2011) Dalton Trans 40:1652–1661
Nasr-Esfahani M, Zendehdel M, Nia NY, Babadi BJMK (2014) RSC Adv 4:15961–15967
Zhang J et al (2012) J Mater Chem 22:16448–16457
Biswas S et al (2014) RSC Adv 4:34248–34256
Gupta KC, Kumar Sutar A (2008) Coord Chem Rev 252:1420–1450
Cozzi PG (2004) Chem Soc Rev 33:410–421
Su H, Li Z, Huo Q, Guan J, Kan Q (2014) RSC Adv 4:9990–9996
Adhikary J et al (2013) Inorg Chem 52:13442–13452
Das C et al (2014) Inorg Chem 53:11426–11437
Khalaji AD, Chermahini AN, Fejfarova K, Dusek M (2010) Struct Chem 21:153–157
Takaichi J et al (2014) Inorg Chem 53:6159–6169
Lutz OMD et al (2013) J Phys Chem Lett 4:1502–1506
Antunes JA et al (2012) J Mol Str 1013:126–133
Jafarian M et al (2012) J Phys Chem C 116:18518–18532
Upadhyay KK, Kumar A, Upadhyay S, Mishra PC (2008) J Mol Str 873:5–16
Bonhommeau S et al (2012) J Phys Chem C 116:11251–11255
Di Bella S (2001) Chem Soc Rev 30:355–366
Krishnamoorthy P, Sathyadevi P, Muthiah PT, Dharmaraj N (2012) RSC Adv 2:12190–12203
Barone G et al (2013) Coord Chem Rev 257:2848–2862
Frisch MJ, Trucks GW, Schlegel HB, Scuseria GE, Robb MA, Cheeseman JR, Montgomery JA, Vreven T Jr, Kudin KN, Burant JC, Millam JM, Iyengar SS, Tomasi J, Barone V, Mennucci B, Cossi M, Scalmani G, Rega N, Petersson GA, Nakatsuji H, Hada M, Ehara M, Toyota K, Fukuda R, Hasegawa J, Ishida M, Nakajima T, Honda Y, Kitao O, Nakai H, Klene M, Li X, Knox JE, Hratchian HP, Cross JB, Bakken V, Adamo C, Jaramillo J, Gomperts R, Stratmann RE, Yazyev O, Austin AJ, Cammi R, Pomelli C, Ochterski JW, Ayala PY, Morokuma K, Voth GA, Salvador P, Dannenberg JJ, Zakrzewski VG, Dapprich S, Daniels AD, Strain MC, Farkas O, Malick DK, Rabuck AD, Raghavachari K, Foresman JB, Ortiz JV, Cui Q, Baboul AG, Clifford S, Cioslowski J, Stefanov BB, Liu G, Liashenko A, Piskorz P, Komaromi I, Martin RL, Fox DJ, Keith T, Al-Laham MA, Peng CY, Nanayakkara A, Challacombe M, Gill PMW, Johnson B, Chen W, Wong MW, Gonzalez C, and Pople JA (2004) Gaussian 03, Revision D.01. Gaussian Inc, Wallingford
G A Zhurko; Chemcraft version 1.7(build375) http://www.chemcraftprog.com
Arish D, Sivasankaran M (2010) Nair J Mol Str 983:112–121
Refat MS, El-Sayed MY, Adam AMA (2013) J Mol Str 1038:62–72
Sebastian M et al (2010) Polyhedron 29:3014–3020
Cui Y, Dong X, Li Y, Li Z, Chen W (2013) Euro J Med Chem 58:323–331
Kazuo N (2009) Infrared and Raman spectra of inorganic and coordination compounds part B: applications in coordination, organometallic, and bioinorganic chemistry, sixthth edn. Wiley, Hoboken, pp 210–222
Tyagi P, Chandra S, Saraswat BS (2015) Spectrochim Acta A 134:200–209
Shebl M, Khalil SME, Ahmed SA, Medien HAA (2010) J Mol Str 980:39–50
Singha BK, Prakasha A, Rajourb HK, Bhojakc N, Adhikari D (2010) Spectrochim Acta A 76:376–383
Kavitha P, Saritha M, Laxma Reddy K (2013) Spectrochim Acta A 102:159–168
Soliman AA, Linert W (1999) Thermochim Acta 338:67–75
Ourari A et al (2014) Polyhedron 67:59–64
Sundar A, Prabhu M, Indra Gandhi N, Marappan M, Rajagopal G (2014) Spectrochim Acta A 129:509–518
Klessinger M, Michl J (1995) Excited states and photochemistry of organic molecules, VCH, Weinheim, pp 63–135
Raman N, Mahalakshmi R, Arun T, Packianathan S, Rajkumar R (2014) J Photochem Photobiol B: Biol 138:211–222
Taha A, Farag AAM, Ammarb AH, Ahmed HM (2014) Spectrochim Acta A 130:494–501
Scherrer P (1918) Nachr Ges Wiss Göttinger 2:98–100
Barnes P, Jacques S, Vicker M (2015) Chapter 15: the concept of peak sShape. School of Crystallography, Birkbeck College, University of London. http://pd.chem.ucl.ac.uk/pdnn/relnotes/rel15.htm
Venkateswarlu K, ChandraBose A, Rameshbabu N (2010) Physica B 405:4256–4261. doi:10.1016/j.physb.2010.07.020
Ghosh P, Kar A, Patra A (2010) J Appl Phys 108:113506
Martin JD (2008) X Powder a software package for powder x-ray diffraction analysis user guide ver 2004.04.82. http://www.xpowder.com/download/xpowder.pdf
Balzar D (1993) J Res Natl Inst Stand Technol 98:321–353
Sen Gupta SP, Chatterjee P (2002) PINSA 68(3):267–291
Ghosh P, Patra A (2007) J Phys Chem C 111:7004–7010
Guha A et al (2013) J Mol Str 1042:104–111
Islam SM et al (2011) J Mol Catal A 336:106–114
Abdel-Nasser MA (2014) Alaghaz J Mol Str 1072:103–113
Jain RK, Mishra AP, Gupta P (2012) J Therm Anal Calorim 110(2):529–534
Shukla S, Mishra AP (2012) J Therm Anal Calorim 107:111–117
Alaghaz A-NMA, Ammara YA, Bayoumi HA, Aldhlmani SA (2014) J Mol Str 1074:359–375
Cavalheiro ÉTG et al (2001) Thermochim Acta 370:129–133
Dilek D, Doğan F, Bilici A, Kaya I (2011) Thermochim Acta 518:72–81
Zhou L et al (2012) Anal Chim Acta 735:96–106
Barwiolek M, Szlyk E, Muzioł TM, Lis T (2011) Dalton Trans 40:11012–11022
Singh K, Kumar Y, Puri P, Kumar M, Sharma C (2012) Eur J Med Chem 52:313–321
Patil SA, Naik VH, Kulkarni AD, Badami PS (2010) Spectrochim Acta A 75:347–354
Hameed SA, Alrouby SK, Hilal R (2013) J Mol Model 19:559–569
Tanaka H, Agar AA, Buyukgungor O (2012) Spectrochim Acta A 87:15–24
Gupta SK, Hitchcock PB, Argal GS (2008) Inorg Chim Acta 361:2139–2146
Govindarasu K, Kavitha E (2014) Spectrochim Acta A 133:799–810
Kianfar AH, Ramazani S, Fath RH, Roushani M (2013) Spectrochim Acta A 105:374–382
Ebrahimipour SY, Abaszadeh M, Castro J, Seifi M (2014) Polyhedron 79:138–150
Ghosh M, Weyhermüller T, Wieghardt K (2010) Dalton Trans 39:1996–2007
Yousef TA, El-Gammal OA, Ahmed SF, Abu El-Reash GM (2015) Spectrochim Acta A 135:690–703
Elamurugu Porchelvi E, Muthu S (2015) Spectrochim Acta A 134:453–464
Anbuselvan C, Jayabharathi J, Thanikachalam V, Tamilselvi G (2012) Spectrochim Acta A 97:125–130
Acknowledgments
BSK specially thanks UGC, New Delhi, India, for financial assistance. He is thankful to the Head, Department of Chemistry, Dr. H. S. Gour University, Sagar India, for departmental facilities. He highly acknowledges Mr. Sandeep Tiwari, Department of Physical Science, M.G.C.G. Vishwvidyalaya, Chitrakoot, Satna (M.P.), India and Ms. Anjali Tiwari, Department of Chemistry, Dr. H. S. Gour University, Sagar India for their valuable guidance and motivational support time to time. He is also thankful to Sophisticated Instrumentation Center, Dr. H. S. Gour University, Sagar for making available Powder X-ray Diffraction, thermal analysis and SEM-EDAX facility.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
ESM 1
(DOCX 1922 kb)
Rights and permissions
About this article
Cite this article
Kusmariya, B.S., Mishra, A.P. Theoretical and experimental studies on three new coordination complexes of Co(II), Ni(II), and Cu(II) with 2,4-dichloro-6-{(E)-[(5-chloro-2 sulfanylphenyl)imino]methyl}phenol Schiff base ligand. J Mol Model 21, 278 (2015). https://doi.org/10.1007/s00894-015-2805-z
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00894-015-2805-z