Abstract
Objectives
This study investigated the cytotoxicity, the residual monomer release, degree of conversion (DC), calcium ion (Ca2+) release, and crystal structure of TheraCal PT (ThPT) by comparison with TheraCal LC (ThLC) and mineral trioxide aggregate (MTA).
Materials and methods
The cytotoxicity of the cured materials was evaluated on human dental pulp stem cells (hDPSCs) isolated from third molars by the water-soluble tetrazolium salt (WST-1) method. The monomer release and DC of the resin-containing materials were analyzed by high-performance liquid chromatography (HPLC) and Fourier transform infrared (FTIR), respectively. The chemical composition and Ca2+ release of the materials were determined by scanning electronic microscopy–energy-dispersive spectroscopy (SEM–EDS), X-ray diffractometry (XRD), and inductively coupled plasma-optical emission spectroscopy (ICP-OES), respectively. Statistical differences were evaluated with one-way ANOVA, repeated measure ANOVA, and the Tukey test (p < 0.05).
Results
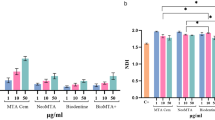
MTA showed significantly lower cytotoxicity than either ThLC or ThPT after 1, 3, and 7 days (p < 0.05). TEGDMA release of ThPT is significantly higher than ThLC (p < 0.05). All materials showed calcium Ca2+ release, with MTA significantly higher than the others (p < 0.05).
Conclusions
MTA showed low cytotoxicity and high Ca2+ release compared to ThLC and ThPT.
Clinical relevance.
The cytotoxicity and residual monomer release of ThLC and ThPT may raise concerns about the viability of hDPSCs. Further investigations with the use of in vivo research models are required to validate in vitro bioactivity properties and the potential adverse biological effects of ThLC and ThPT on hDPSCs.








Similar content being viewed by others
References
Chan C, Lan W, Chang M, Chen Y, Lan W, Chang H, Jeng J (2005) Effects of TGF-βs on the growth, collagen synthesis and collagen lattice contraction of human dental pulp fibroblasts in vitro. Arch Oral Biol 50:469–479. https://doi.org/10.1016/j.archoralbio.2004.10.005
Huang G-J, Gronthos S, Shi S (2009) Mesenchymal stem cells derived from dental tissues vs. those from other sources: their biology and role in regenerative medicine. J Dent Res 88:792–806. https://doi.org/10.1177/0022034509340867
Rosa V, Zhang Z, Grande R, Nör J (2013) Dental pulp tissue engineering in full-length human root canals. J Dent Res 92:970–975. https://doi.org/10.1177/0022034513505772
Tatullo M, Codispoti B, Sied J, Makeeva I, Paduano F, Marrelli M, Spagnuolo G (2019) Stem cells-based and molecular-based approaches in regenerative dentistry: a topical review. Curr Stem Cell Res Ther 14:607–616. https://doi.org/10.2174/1574888X14666190626111154
Kim J-H, Kim S-Y, Woo S-M, Jeong H-N, Jung J-Y, Kim S-M, Lim H-S (2019) Combination of mineral trioxide aggregate and propolis promotes odontoblastic differentiation of human dental pulp stem cells through ERK signaling pathway. Food Science and Biotechnology 28:1801–1809. https://doi.org/10.1007/s10068-019-00609-5
Maria de Lourdes RA, Holland R, Reis A, Bortoluzzi MC, Murata SS, Dezan E Jr, Souza V, Alessandro LD (2008) Evaluation of mineral trioxide aggregate and calcium hydroxide cement as pulp-capping agents in human teeth. J Endod 34:1–6. https://doi.org/10.1016/j.joen.2007.09.012
Camargo S, Camargo C, Hiller KA, Rode S, Schweikl H, Schmalz G (2009) Cytotoxicity and genotoxicity of pulp capping materials in two cell lines. Int Endod J 42:227–237. https://doi.org/10.1111/j.1365-2591.2008.01506.x
Takita T, Hayashi M, Takeichi O, Ogiso B, Suzuki N, Otsuka K, Ito K (2006) Effect of mineral trioxide aggregate on proliferation of cultured human dental pulp cells. Int Endod J 39:415–422. https://doi.org/10.1111/j.1365-2591.2008.01506.x
Collado-González M, García-Bernal D, Oñate-Sánchez R, Ortolani-Seltenerich P, Álvarez-Muro T, Lozano A, Forner L, Llena C, Moraleda J, Rodríguez-Lozano F (2017) Cytotoxicity and bioactivity of various pulpotomy materials on stem cells from human exfoliated primary teeth. Int Endod J 50:19–30. https://doi.org/10.1111/iej.12751
Da Rosa W, Piva E, Da Silva A (2018) Disclosing the physiology of pulp tissue for vital pulp therapy. Int Endod J 51:829–846. https://doi.org/10.1111/iej.12906
Poggio C, Arciola CR, Beltrami R, Monaco A, Dagna A, Lombardini M, Visai L (2014) Cytocompatibility and antibacterial properties of capping materials. Scientific World Journal 2014:1–10. https://doi.org/10.1155/2014/181945
Sawicki L, Pameijer CH, Emerich K, Adamowicz-Klepalska B (2008) Histological evaluation of mineral trioxide aggregate and calcium hydroxide in direct pulp capping of human immature permanent teeth. Am J Dent 21:262–266
Moghaddame-Jafari S, Mantellini MG, Botero TM, McDonald NJ, Nör JE (2005) Effect of ProRoot MTA on pulp cell apoptosis and proliferation in vitro. J Endod 31:387–391. https://doi.org/10.1097/01.don.0000145423.89539.d7
Roberts HW, Toth JM, Berzins DW, Charlton DG (2008) Mineral trioxide aggregate material use in endodontic treatment: a review of the literature. Dent Mater 24:149–164. https://doi.org/10.1016/j.dental.2007.04.007
Yoldaş SE, Bani M, Atabek D, Bodur H (2016) Comparison of the potential discoloration effect of bioaggregate, biodentine, and white mineral trioxide aggregate on bovine teeth: in vitro research. J Endod 42:1815–1818. https://doi.org/10.1016/j.joen.2016.08.020
Ajami AA, Navimipour EJ, Oskoe SS, Kahnamoui MA, Lotfi M, Daneshpooy M (2013) Comparison of shear bond strength of resin-modified glass ionomer and composite resin to three pulp capping agents. J Dent Res Dent Clin Dent Prospects 7(3):164. https://doi.org/10.5681/joddd.2013.026
Hashem DF, Foxton R, Manoharan A, Watson TF, Banerjee A (2014) The physical characteristics of resin composite–calcium silicate interface as part of a layered/laminate adhesive restoration. Dent Mater 30(3):343–349. https://doi.org/10.1016/j.dental.2013.12.010
Nielsen MJ, Casey JA, VanderWeele RA, Vandewalle KS (2016) Mechanical properties of new dental pulp-capping materials. Gen Dent 64(1):44–48
Kim Y, Lee D, Kim HM, Kye M, Kim SY (2021) Biological characteristics and odontogenic differentiation effects of calcium silicate-based pulp capping materials. Materials 14(16):4661. https://doi.org/10.3390/ma14164661
Rodríguez-Lozano FJ, López-García S, Garcia-Bernal D, Sanz J, Lozano A, Pecci-Lloret M, Melo M, López-Ginés C, Forner L (2021) Cytocompatibility and bioactive properties of the new dual-curing resin-modified calcium silicate-based material for vital pulp therapy. Clin Oral Investig 25:5009–5024. https://doi.org/10.1007/s00784-021-03811-0
Sanz JL, Soler-Doria A, López-García S, García-Bernal D, Rodríguez-Lozano FJ, Lozano A, Llena C, Forner L, Guerrero-Gironés J, Melo M (2021) Comparative biological properties and mineralization potential of 3 endodontic materials for vital pulp therapy: Theracal PT, Theracal LC, and Biodentine on human dental pulp stem cells. J Endod 47:1896–1906. https://doi.org/10.1016/j.joen.2021.08.001
Jeanneau C, Laurent P, Rombouts C, Giraud T, About I (2017) Light-cured tricalcium silicate toxicity to the dental pulp. J Endod 43(12):2074–2080. https://doi.org/10.1016/j.joen.2017.07.010
Arias-Moliz FC, Lung CYK, Wismayer PS, Camilleri J (2017) Antimicrobial and biological activity of leachate from light curable pulp capping materials. J Dent 64:45–51. https://doi.org/10.1016/j.jdent.2017.06.006
Chen L, Suh BI (2017) Cytotoxicity and biocompatibility of resin-free and resin-modified direct pulp capping materials: a state-of-the-art review. Dent Mater J 36(1):1–7. https://doi.org/10.4012/dmj.2016-107
Klein-Júnior CA, Zimmer R, Hentschke GS, Machado DC, Santos RBD, Reston EG (2018) Effect of heat treatment on cytotoxicity of self-adhesive resin cements: cell viability analysis. Eur J Dent 12(02):281–286. https://doi.org/10.4103/ejd.ejd_34_18
Klein-Junior CA, Zimmer R, Borghetti DLB, Portell FF, Abich FC, Marinowic DR, Hosaka K, Reston EG (2020) Hot air stream reduces cytotoxicity of light-cured calcium hydroxide based cements. J Clin Exp Dent 12(3):e215. https://doi.org/10.4317/jced.56590
Leite MLdA, Costa CAdS, Duarte RM, Andrade AKMd, Soares DG (2018) Bond strength and cytotoxicity of a universal adhesive according to the hybridization strategies to dentin. Braz Dent J 29:68–75. https://doi.org/10.1590/0103-6440201801698
Salehi S, Gwinner F, Mitchell JC, Pfeifer C (2015) Ferracane JL (2015) Cytotoxicity of resin composites containing bioactive glass fillers. Dent Mater 31(2):195–203. https://doi.org/10.1016/j.dental.2014.12.004
Karaöz E, Doğan BN, Aksoy A, Gacar G, Akyüz S, Ayhan S, Genç ZS, Yürüker S, Duruksu G, Demircan PÇ (2010) Isolation and in vitro characterisation of dental pulp stem cells from natal teeth. Histochem Cell Biol 133:95–112. https://doi.org/10.1007/s00418-009-0646-5
Dominici M, Le Blanc K, Mueller I, Slaper-Cortenbach I, Marini F, Krause D, Deans R, Keating A, Prockop D, Horwitz E (2006) Minimal criteria for defining multipotent mesenchymal stromal cells. The International Society for Cellular Therapy position statement. Cytotherapy 8:315–317. https://doi.org/10.1080/14653240600855905
Rodríguez-Lozano F, Collado-González M, Tomás-Catalá C, García-Bernal D, López S, Onate-Sanchez R, Moraleda J, Murcia L (2019) GuttaFlow Bioseal promotes spontaneous differentiation of human periodontal ligament stem cells into cementoblast-like cells. Dent Mater 35:114–124. https://doi.org/10.1016/j.dental.2018.11.003
Rueggeberg F, Hashinger D, Fairhurst C (1990) Calibration of FTIR conversion analysis of contemporary dental resin composites. Dent Mater 6:241–249. https://doi.org/10.1016/S0109-5641(05)80005-3
Lee B-N, Lee K-N, Koh J-T, Min K-S, Chang H-S, Hwang I-N, Hwang Y-C, Oh W-M (2014) Effects of 3 endodontic bioactive cements on osteogenic differentiation in mesenchymal stem cells. J Endod 40:1217–1222. https://doi.org/10.1016/j.joen.2014.01.036
Buttke TM, McCubrey JA, Owen TC (1993) Use of an aqueous soluble tetrazolium/formazan assay to measure viability and proliferation of lymphokine-dependent cell lines. J Immunol Methods 157:233–240. https://doi.org/10.1016/0022-1759(93)90092-L
Ishiyama M, Tominaga H, Shiga M, Sasamoto K, Ohkura Y, Ueno K (1996) A combined assay of cell vability and in vitro cytotoxicity with a highly water-soluble tetrazolium salt, neutral red and crystal violet. Biol Pharm Bull 19:1518–1520. https://doi.org/10.1248/bpb.19.1518
Ishiyama M, Shiga M, Sasamoto K, Mizoguchi M, P-g HE (1993) A new sulfonated tetrazolium salt that produces a highly water-soluble formazan dye. Chem Pharm Bull 41:1118–1122. https://doi.org/10.1248/cpb.41.1118
Nam OH, Kim J-H, Choi SC, Kim Y (2020) Time-dependent response of human deciduous tooth-derived dental pulp cells treated with TheraCal LC: functional analysis of gene interactions compared to MTA. J Clin Med 9:531. https://doi.org/10.3390/jcm9020531
Bortoluzzi EA, Niu L-n, Palani CD, El-Awady AR, Hammond BD, Pei D-d, Tian F-c, Cutler CW, Pashley DH, Tay FR (2015) Cytotoxicity and osteogenic potential of silicate calcium cements as potential protective materials for pulpal revascularization. Dent Mater 31:1510–1522. https://doi.org/10.1016/j.dental.2015.09.020
Łagocka R, Jakubowska K, Chlubek D, Buczkowska-Radlińska J (2015) Elution study of unreacted TEGDMA from bulk-fill composite (SDR™ Dentsply) using HPLC. Adv Med Sci 60(2):191–198. https://doi.org/10.1016/j.advms.2015.02.003
Schneider TR, Hakami-Tafreshi R, Tomasino-Perez A, Tayebi L, Lobner D (2019) Effects of dental composite resin monomers on dental pulp cells. Dent Mater J 2018-163. https://doi.org/10.4012/dmj.2018-163
Nilsen BW, Jensen E, Örtengren U, Michelsen VB (2017) Analysis of organic components in resin-modified pulp capping materials: critical considerations. Eur J Oral Sci 125:183–194. https://doi.org/10.1111/eos.12347
Di Foggia M, Prati C, Gandolfi MG, Taddei P (2019) Spectroscopic and morphological data assessing the apatite forming ability of calcium hydroxide-releasing materials for pulp capping. Data Brief 23:103719. https://doi.org/10.1016/j.dib.2019.103719
Suh B, Yin R, Cannon M, Martin DE, Polymerizable dental pulp healing, capping, and lining material and method for use. 2008, Google Patents.
Geurtsen W (2000) Biocompatibility of resin-modified filling materials. Crit Rev Oral Biol Med 11:333–355. https://doi.org/10.1177/10454411000110030401
Issa Y, Watts D, Brunton P, Waters C, Duxbury A (2004) Resin composite monomers alter MTT and LDH activity of human gingival fibroblasts in vitro. Dent Mater 20:12–20. https://doi.org/10.1016/S0109-5641(03)00053-8
Janani K, Teja KV, Sandhya R, Alam MK, Al-Qaisi RK, Shrivastava D, Alnusayri MO, Alkhalaf ZA, Sghaireen MG, Srivastava KC (2021) Monomer elution from three resin composites at two different time interval using high performance liquid chromatography—an in-vitro study. Polymers 13(24):4395. https://doi.org/10.3390/polym13244395
Gul P, Miloglu FD, Akgul N (2014) HPLC analysis of eluted monomers from dental composite using different immersion media. J Liq Chromatogr Relat Technol 37(2):155–170. https://doi.org/10.1080/10826076.2012.738619
Väkiparta M, Puska M, Vallittu PK (2006) Residual monomers and degree of conversion of partially bioresorbable fiber-reinforced composite. Acta Biomater 2(1):29–37. https://doi.org/10.1016/j.actbio.2005.08.009
Moharamzadeh K, Noort RV, Brook IM, Scutt AM (2007) HPLC analysis of components released from dental composites with different resin compositions using different extraction media. J Mater Sci Mater Med 18(1):133–137. https://doi.org/10.1007/s10856-006-0671-z
Goldberg M (2008) In vitro and in vivo studies on the toxicity of dental resin components: a review. Clin Oral Investig 12(1):1–8. https://doi.org/10.1007/s00784-007-0162-8
Ferracane J (1994) (1994) Elution of leachable components from composites. J Oral Rehabil 21(4):441–452. https://doi.org/10.1111/j.1365-2842.1994.tb01158.x
Munksgaard EC, Peutzfeldt A, Asmussen E (2000) (2000) Elution of TEGDMA and BisGMA from a resin and a resin composite cured with halogen or plasma light. Eur J Oral Sci 108(4):341–345. https://doi.org/10.1034/j.1600-0722.2000.108004341.x
Imazato S, McCabe JF, Tarumi H, Ehara A, Ebisu S (2001) Degree of conversion of composites measured by DTA and FTIR. Dent Mater 17(2):178–183. https://doi.org/10.1016/s0109-5641(00)00066-x
Stansbury J, Dickens SH (2001) Determination of double bond conversion in dental resins by near infrared spectroscopy. Dent Mater 17(1):71–79. https://doi.org/10.1016/s0109-5641(00)00062-2
Lempel E, Czibulya Z, Kunsági-Máté S, Szalma J, Sümegi B, Böddi K (2014) Quantification of conversion degree and monomer elution from dental composite using HPLC and micro-Raman spectroscopy. Chromatographia 77(17):1137–1144. https://doi.org/10.1007/s10337-014-2647-3
Nocca G, Iori A, Rossini C, Martorana GE, Ciasca G, Arcovito A, Cordaro M, Lupi A, Marigo L (2015) Effects of barriers on chemical and biological properties of two dual resin cements. Eur J Oral Sci 123:208–214. https://doi.org/10.1111/eos.12178
Miletic V, Santini A, Trkulja I (2009) Quantification of monomer elution and carbon–carbon double bonds in dental adhesive systems using HPLC and micro-Raman spectroscopy. J Dent 37:177–184. https://doi.org/10.1016/j.jdent.2008.11.006
Pongprueksa P, De Munck J, Duca RC, Poels K, Covaci A, Hoet P, Godderis L, Van Meerbeek B, Van Landuyt KL (2015) Monomer elution in relation to degree of conversion for different types of composite. J Dent 43:1448–1455. https://doi.org/10.1016/j.jdent.2015.10.013
Manaspon C, Jongwannasiri C, Chumprasert S, Sa-Ard-Iam N, Mahanonda R, Pavasant P, Porntaveetus T, Osathanon T (2021) Human dental pulp stem cell responses to different dental pulp capping materials. BMC Oral Health 21:1–13. https://doi.org/10.1186/s12903-021-01544-w
Lopez-Cazaux S, Bluteau G, Magne D, Lieubeau-Teillet B, Guicheux J, Alliot-Licht B (2006) Culture medium modulates the behaviour of human dental pulp-derived cells. eClls and Mater J 11:35–42
Gandolfi MG, Siboni F, Botero T, Bossù M, Riccitiello F, Prati C (2015) Calcium silicate and calcium hydroxide materials for pulp capping: biointeractivity, porosity, solubility and bioactivity of current formulations. J Appl Biomater Funct Mater 13:43–60. https://doi.org/10.5301/jabfm.5000201
Elbanna A, Atta D, Sherief DI (2022) In vitro bioactivity of newly introduced dual-cured resin-modified calcium silicate cement. Dent Res J 19. https://doi.org/10.4103/1735-3327.33668
Camilleri J (2014) Hydration characteristics of Biodentine and Theracal used as pulp capping materials. Dent Mater 30:709–715. https://doi.org/10.1016/j.dental.2014.03.012
Acknowledgements
The authors thank Ahmet Öztürk and Kocaeli University, Stem Cell and Gene Therapies Research, and Application Center (KÖGEM) for the support of this study.
Funding
This research was supported by the Scientific Research Projects of Cukurova University (project number TSA-2022–14441).
Author information
Authors and Affiliations
Contributions
Conceptualization: C.K, S.N.K., and S.G.B.; methodology: C.K, S.N.K., and S.G.B.; formal analysis and investigation: S.N.K. and S.G.B.; writing—original draft preparation: C.K, S.N.K., and S.G.B.; writing—review and editing: S.N.K. and C.K.; funding acquisition: S.N.K.; resources: C.K, S.N.K., and S.G.B; supervision: S.N.K. and C.K.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethics approval
All methods performed in the present study involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki Declaration and its later modifications or comparable ethical standards. The present study and the protocol were found medically appropriate with the ethics committee report numbered 2021/117.65 of Cukurova University Faculty of Medicine Clinical Research Ethics Committee.
Consent to participate
The written informed consent was obtained from all individual participants included in the study.
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Küden, C., Karakaş, S.N. & Batmaz, S.G. Comparative chemical properties, bioactivity, and cytotoxicity of resin-modified calcium silicate–based pulp capping materials on human dental pulp stem cells. Clin Oral Invest 26, 6839–6853 (2022). https://doi.org/10.1007/s00784-022-04713-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00784-022-04713-5