Abstract
Objectives
The aim of this study is to investigate the influence of prognostic biomarkers on radiosensitivity and survival of advanced head and neck squamous cell carcinomas treated by primary (chemo)radiation.
Material and methods
The clinicopathological data and immunohistochemical staining of p16, c-Met, survivin, PD-1, and PD-L1 of 82 primarily (chemo)irradiated patients with head and neck squamous cell carcinoma were analyzed. Associations with local and locoregional radiation response, overall survival (OS), disease-free (DFS), and disease-specific survival (DSS) were assessed.
Results
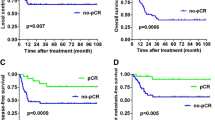
Complete tumor response was associated with increased patient age (p = 0.007), N0-status (p = 0.022), M0-status (p = 0.007), and p16-positivity (p = 0.022). High PD-L1 was associated with M0-status (p = 0.026) and indicated tumor response to irradiation (p = 0.057); survivin expression showed higher rates of response failure (p = 0.073). Low PD-1 was associated with increased T-stage (p = 0.029) and local recurrence (p = 0.014). High PD-1 was strongly correlated with PD-L1-positive tumor infiltrating lymphocytes (p < 0.001). Low PD-L1 showed a significant correlation with high c-Met expression (p = 0.01). Significant predictors for unfavorable univariate survival were incomplete tumor response (DSS, p < 0.001), single radiotherapy (DSS, p = 0.002), M1-status (DSS, p < 0.001), decreased radiation dose (DSS, p = 0.014), high survivin (DSS, p = 0.045), and high c-Met (OS, p < 0.05). Survivin and c-Met also showed prognostic significance in multivariate survival analysis.
Conclusions
P16 and PD-L1 indicate radiosensitivity, whereas survivin and c-Met implicate radioresistance in primarily (chemo)irradiated head and neck squamous cell carcinomas. The role of the PD-1/PD-L1 immune checkpoints in radiation response and survival merits further investigation.
Clinical relevance
The findings may improve patient-specific therapy according to individual tumor characteristics.



Similar content being viewed by others
References
Haddad RI, Shin DM (2008) Recent advances in head and neck cancer. N Engl J Med 359:1143–1154. doi:10.1056/NEJMra0707975
D’Souza G, Kreimer AR, Viscidi R, Pawlita M, Fakhry C, Koch WM, Westra WH, Gillison ML (2007) Case-control study of human papillomavirus and oropharyngeal cancer. N Engl J Med 356:1944–1956
Akervall J, Nandalur S, Zhang J, Qian CN, Goldstein N, Gyllerup P, Gardinger Y, Alm J, Lorenc K, Nilsson K, Resau J, Wilson G, Teh B (2014) A novel panel of biomarkers predicts radioresistance in patients with squamous cell carcinoma of the head and neck. Eur J Cancer 50:570–581. doi:10.1016/j.ejca.2013.11.007
Argiris A, Karamouzis MV, Raben D, Ferris RL (2008) Head and neck cancer. Lancet 371:1695–1709. doi:10.1016/s0140-6736(08)60728-x
Fakhry C, Westra WH, Li S, Cmelak A, Ridge JA, Pinto H, Forastiere A, Gillison ML (2008) Improved survival of patients with human papillomavirus-positive head and neck squamous cell carcinoma in a prospective clinical trial. J Natl Cancer Inst 100:261–269. doi:10.1093/jnci/djn011
Ang KK, Harris J, Wheeler R, Weber R, Rosenthal DI, Nguyen-Tân PF, Westra WH, Chung CH, Jordan RC, Lu C, Kim H, Axelrod R, Silverman C, Redmond KP, Gillison ML (2010) Human papillomavirus and survival of patients with oropharyngeal cancer. N Engl J Med 363:24–35. doi:10.1056/NEJMoa0912217
Bhardwaj V, Cascone T, Cortez MA, Amini A, Evans J, Komaki RU, Heymach JV, Welsh JW (2013) Modulation of c-Met signaling and cellular sensitivity to radiation: potential implications for therapy. Cancer 119:1768–1775. doi:10.1002/cncr.27965
Peruzzi B, Bottaro DP (2006) Targeting the c-Met signaling pathway in cancer. Clin Cancer Res 12:3657–3660. doi:10.1158/1078-0432.CCR-06-0818
Blumenschein GR Jr, Mills GB, Gonzalez-Angulo AM (2012) Targeting the hepatocyte growth factor-cMET axis in cancer therapy. J Clin Oncol 30:3287–3296. doi:10.1200/JCO.2011.40.3774
Seiwert TY, Jagadeeswaran R, Faoro L, Janamanchi V, Nallasura V, El Dinali M, Yala S, Kanteti R, Cohen EE, Lingen MW, Martin L, Krishnaswamy S, Klein-Szanto A, Christensen JG, Vokes EE, Salgia R (2009) The MET receptor tyrosine kinase is a potential novel therapeutic target for head and neck squamous cell carcinoma. Cancer Res 69:3021–3031. doi:10.1158/0008-5472.CAN-08-2881
Baschnagel AM, Williams L, Hanna A, Chen PY, Krauss DJ, Pruetz BL, Akervall J, Wilson GD (2014) c-Met expression is a marker of poor prognosis in patients with locally advanced head and neck squamous cell carcinoma treated with chemoradiation. Int J Radiat Oncol Biol Phys 88:701–707. doi:10.1016/j.ijrobp.2013.11.013
Ettl T, Viale-Bouroncle S, Hautmann M, Gosau M, Kölbl O, Reichert TE, Morsczeck C (2015) AKT and MET signalling mediates antiapoptotic radioresistance in head neck cancer cell lines. Oral Oncol 51:158–163. doi:10.1016/j.oraloncology.2014.11.005
De Bacco F, Luraghi P, Medico E, Reato G, Girolami F, Perera T, Gabriele P, Comoglio PM, Boccaccio C (2011) Induction of MET by ionizing radiation and its role in radioresistance and invasive growth of cancer. J Natl Cancer Inst 103:645–661. doi:10.1093/jnci/djr093
Lippert BM, Knauer SK, Fetz V, Mann W, Stauber RH (2007) Dynamic survivin in head and neck cancer: molecular mechanism and therapeutic potential. Int J Cancer 121:1169–1174. doi:10.1002/ijc.22941
Kim BM, Hong Y, Lee S, Liu P, Lim JH, Lee YH, Lee TH, Chang KT, Hong Y (2015) Therapeutic implications for overcoming radiation resistance in cancer therapy. Int J Mol Sci 16:26880–26913. doi:10.3390/ijms161125991
Cheung CH, Huang CC, Tsai FY, Lee JY, Cheng SM, Chang YC, Huang YC, Chen SH, Chang JY (2013) Survivin—biology and potential as a therapeutic target in oncology. Onco Targets Ther 6:1453–1462. doi:10.2147/OTT.S33374
Ambrosini G, Adida C, Altieri DC (1997) A novel anti-apoptosis gene, survivin, expressed in cancer and lymphoma. Nat Med 3:917–921
Freier K, Pungs S, Sticht C, Flechtenmacher C, Lichter P, Joos S, Hofele C (2007) High survivin expression is associated with favorable outcome in advanced primary oral squamous cell carcinoma after radiation therapy. Int J Cancer 120:942–946. doi:10.1002/ijc.22380
Farnebo L, Tiefenböck K, Ansell A, Thunell LK, Garvin S, Roberg K (2013) Strong expression of survivin is associated with positive response to radiotherapy and improved overall survival in head and neck squamous cell carcinoma patients. Int J Cancer 133:1994–2003. doi:10.1002/ijc.28200
Schoenfeld JD (2015) Immunity in head and neck cancer. Cancer Immunol Res 3:12–17. doi:10.1158/2326-6066.CIR-14-0205
Chen L (2004) Co-inhibitory molecules of the B7-CD28 family in the control of T-cell immunity. Nat Rev Immunol 4:336–347. doi:10.1038/nri1349
D’Incecco A, Andreozzi M, Ludovini V, Rossi E, Capodanno A, Landi L, Tibaldi C, Minuti G, Salvini J, Coppi E, Chella A, Fontanini G, Filice ME, Tornillo L, Incensati RM, Sani S, Crino L, Terracciano L, Cappuzzo F (2015) PD-1 and PD-L1 expression in molecularly selected non-small-cell lung cancer patients. Br J Cancer 112:95–102. doi:10.1038/bjc.2014.555
Dong H, Strome SE, Salomao DR, Tamura H, Hirano F, Flies DB, Roche PC, Lu J, Zhu G, Tamada K, Lennon VA, Celis E, Chen L (2002) Tumor-associated B7-H1 promotes T-cell apoptosis: a potential mechanism of immune evasion. Nat Med 8:793–800. doi:10.1038/nm730
Zou W, Chen L (2008) Inhibitory B7-family molecules in the tumour microenvironment. Nat Rev Immunol 8:467–477. doi:10.1038/nri2326
Taube JM, Anders RA, Young GD, Xu H, Sharma R, McMiller TL, Chen S, Klein AP, Pardoll DM, Topalian SL, Chen L (2012) Colocalization of inflammatory response with B7-h1 expression in human melanocytic lesions supports an adaptive resistance mechanism of immune escape. Sci Transl Med 4:127ra137. doi:10.1126/scitranslmed.3003689
Eppihimer MJ, Gunn J, Freeman GJ, Greenfield EA, Chernova T, Erickson J, Leonard JP (2002) Expression and regulation of the PD-L1 immunoinhibitory molecule on microvascular endothelial cells. Microcirculation 9:133–145. doi:10.1038/sj/mn/7800123
Freeman GJ, Long AJ, Iwai Y, Bourque K, Chernova T, Nishimura H, Fitz LJ, Malenkovich N, Okazaki T, Byrne MC, Horton HF, Fouser L, Carter L, Ling V, Bowman MR, Carreno BM, Collins M, Wood CR, Honjo T (2000) Engagment of PD-1 the immunoinhibitory receptor by a novel B7 family member leads to negative regulation of lymphocyte activation. J Exp Med 192:1027–1034
Iwai Y, Ishida M, Tanaka Y, Okazaki T, Honjo T, Minato N (2002) Involvement of PD-L1 on tumor cells in the escape from host immune system and tumor immunotherapy by PD-L1 blockade. Proc Natl Acad Sci U S A 99:12293–12297. doi:10.1073/pnas.192461099
Dovedi SJ, Adlard AL, Lipowska-Bhalla G, McKenna C, Jones S, Cheadle EJ, Stratford IJ, Poon E, Morrow M, Stewart R, Jones H, Wilkinson RW, Honeychurch J, Illidge TM (2014) Acquired resistance to fractionated radiotherapy can be overcome by concurrent PD-L1 blockade. Cancer Res 74:5458–5468. doi:10.1158/0008-5472.CAN-14-1258
Zeng J, See AP, Phallen J, Jackson CM, Belcaid Z, Ruzevick J, Durham N, Meyer C, Harris TJ, Albesiano E, Pradilla G, Ford E, Wong J, Hammers HJ, Mathios D, Tyler B, Brem H, Tran PT, Pardoll D, Drake CG, Lim M (2013) Anti-PD-1 blockade and stereotactic radiation produce long-term survival in mice with intracranial gliomas. Int J Radiat Oncol Biol Phys 86:343–349. doi:10.1016/j.ijrobp.2012.12.025
Verbrugge I, Hagekyriakou J, Sharp LL, Galli M, West A, McLaughlin NM, Duret H, Yagita H, Johnstone RW, Smyth MJ, Haynes NM (2012) Radiotherapy increases the permissiveness of established mammary tumors to rejection by immunomodulatory antibodies. Cancer Res 72:3163–3174. doi:10.1158/0008-5472.CAN-12-0210
Vokes EE, Liebowitz DN, Weichselbaum RR (1997) Nasopharyngeal carcinoma. Lancet 350:1087–1091. doi:10.1016/s0140-6736(97)07269-3
Pignon JP, le Maitre A, Bourhis J, Group M-NC (2007) Meta-analyses of chemotherapy in head and neck cancer (MACH-NC): an update. Int J Radiat Oncol Biol Phys 69:S112–S114. doi:10.1016/j.ijrobp.2007.04.088
Milanes-Yearsley M, Hammond ME, Pajak TF, Cooper JS, Chang C, Griffin T, Nelson D, Laramore G, Pilepich M (2002) Tissue micro-array: a cost and time-effective method for correlative studies by regional and national cancer study groups. Mod Pathol 15:1366–1373. doi:10.1097/01.MP.0000036345.18944.22
Heiduschka G, Grah A, Oberndorfer F, Kadletz L, Altorjai G, Kornek G, Wrba F, Thurnher D, Selzer E (2015) Improved survival in HPV/p16-positive oropharyngeal cancer patients treated with postoperative radiotherapy. Strahlenther Onkol 191:209–216. doi:10.1007/s00066-014-0753-7
Laurent-Puig P, Cayre A, Manceau G, Buc E, Bachet JB, Lecomte T, Rougier P, Lievre A, Landi B, Boige V, Ducreux M, Ychou M, Bibeau F, Bouche O, Reid J, Stone S, Penault-Llorca F (2009) Analysis of PTEN, BRAF, and EGFR status in determining benefit from cetuximab therapy in wild-type KRAS metastatic colon cancer. J Clin Oncol 27:5924–5930. doi:10.1200/JCO.2008.21.6796
Tsuta K, Kozu Y, Mimae T, Yoshida A, Kohno T, Sekine I, Tamura T, Asamura H, Furuta K, Tsuda H (2012) c-MET/phospho-MET protein expression and MET gene copy number in non-small cell lung carcinomas. J Thorac Oncol 7:331–339
Böger C, Behrens H-M, Mathiak M, Krüger S, Kalthoff H, Röcken C (2016) PD-L1 is an independent prognostic predictor in gastric cancer of western patients. Oncotarget 7:24269–24283
Lassen P, Eriksen JG, Hamilton-Dutoit S, Tramm T, Alsner J, Overgaard J (2009) Effect of HPV-associated p16INK4A expression on response to radiotherapy and survival in squamous cell carcinoma of the head and neck. J Clin Oncol 27:1992–1998. doi:10.1200/JCO.2008.20.2853
Nakao Y, Yang X, Yokoyama M, Ferenczy A, Tang S-C, Pater MM, Pater A (1997) Induction of p16 during immortalization by HPV 16 and 18 and not during malignant transformation. Br J Cancer 75:1410–1416
Gillison ML, Koch WM, Capone RB, Spafford M, Westra WH, Wu L, Zahurak ML, Daniel RW, Viglione M, Symer DE, Shah KV, Sidransky D (2000) Evidence for a causal association between human Papillomvirus and a subset of head and neck cancer. J Natl Cancer Inst 92:709–720
Chen X, Duan N, Zhang C, Zhang W (2016) Survivin and tumorigenesis: molecular mechanisms and therapeutic strategies. J Cancer 7:314–323. doi:10.7150/jca.13332
Capalbo G, Rödel C, Stauber RH, Knauer SK, Bache M, Kappler M, Rödel F (2007) The role of survivin for radiation therapy. Prognostic and predictive factor and therapeutic target. Strahlenther Onkol 183:593–599. doi:10.1007/s00066-007-1800-4
Erpolat OP, Gocun PU, Akmansu M, Karakus E, Akyol G (2012) High expression of nuclear survivin and aurora B predicts poor overall survival in patients with head and neck squamous cell cancer. Strahlenther Onkol 188:248–254. doi:10.1007/s00066-011-0042-7
Lo Muzio L, Pannone G, Staibano S, Mignogna MD, Rubini C, Mariggio MA, Procaccini M, Ferrari F, De Rosa G, Altieri DC (2003) Survivin expression in oral squamous cell carcinoma. Br J Cancer 89:2244–2248. doi:10.1038/sj.bjc.6601402
Li F, Yang J, Ramnath N, Javle MM, Tan D (2005) Nuclear or cytoplasmic expression of survivin: what is the significance? Int J Cancer 114:509–512. doi:10.1002/ijc.20768
Fan S, Ma XY, Wang JA, Yuan RQ, Meng Q, Cao Y, Laterra JJ, Goldberg ID, Rosen EM (2000) The cytokine hepatocyte growth factor/scatter factor inhibits apoptosis and enhances DNA repair by a common mechanism involving signaling through phosphatidyl inositol 3′ kinase. Oncogene 19:2212–2223
Zhang YW, Vande Woude GF (2003) HGF/SF-met signaling in the control of branching morphogenesis and invasion. J Cell Biochem 88:408–417. doi:10.1002/jcb.10358
Su L, Wang Y, Xiao M, Lin Y, Yu L (2010) Up-regulation of survivin in oral squamous cell carcinoma correlates with poor prognosis and chemoresistance. Oral Surg Oral Med Oral Pathol Oral Radiol Endod 110:484–491. doi:10.1016/j.tripleo.2010.04.009
Tang MKS, Zhou HY, Yam JWP, Wong AST (2010) c-Met overexpression contributes to the acquired apoptotic resistance of Nonadherent ovarian cancer cells through a cross talk mediated by phosphatidylinositol 3-kinase and extracellular signal-regulated kinase 1/2. Neoplasia 12:128–IN125. doi:10.1593/neo.91438
Zandberg DP, Strome SE (2014) The role of the PD-L1:PD-1 pathway in squamous cell carcinoma of the head and neck. Oral Oncol 50:627–632. doi:10.1016/j.oraloncology.2014.04.003
Vassilakopoulou M, Avgeris M, Velcheti V, Kotoula V, Rampias T, Chatzopoulos K, Perisanidis C, Kontos CK, Giotakis AI, Scorilas A, Rimm D, Sasaki C, Fountzilas G, Psyrri A (2016) Evaluation of PD-L1 expression and associated tumor-infiltrating lymphocytes in laryngeal squamous cell carcinoma. Clin Cancer Res 22:704–713. doi:10.1158/1078-0432.CCR-15-1543
Pardoll DM (2012) The blockade of immune checkpoints in cancer immunotherapy. Nat Rev Cancer 12:252–264. doi:10.1038/nrc3239
Wu CT, Chen WC, Chang YH, Lin WY, Chen MF (2016) The role of PD-L1 in the radiation response and clinical outcome for bladder cancer. Sci Rep 6:19740–19749. doi:10.1038/srep19740
Lin YM, Sung WW, Hsieh MJ, Tsai SC, Lai HW, Yang SM, Shen KH, Chen MK, Lee H, Yeh KT, Chen CJ (2015) High PD-L1 expression correlates with metastasis and poor prognosis in oral squamous cell carcinoma. PLoS One 10:e0142656. doi:10.1371/journal.pone.0142656
Balan M, Miery Teran E, Waaga-Gasser AM, Gasser M, Choueiri TK, Freeman G, Pal S (2015) Novel roles of c-Met in the survival of renal cancer cells through the regulation of HO-1 and PD-L1 expression. J Biol Chem 290:8110–8120. doi:10.1074/jbc.M114.612689
Taube JM, Klein A, Brahmer JR, Xu H, Pan X, Kim JH, Chen L, Pardoll DM, Topalian SL, Anders RA (2014) Association of PD-1, PD-1 ligands, and other features of the tumor immune microenvironment with response to anti-PD-1 therapy. Clin Cancer Res 20:5064–5074. doi:10.1158/1078-0432.CCR-13-3271
Salama AK, Postow MA, Salama JK (2016) Irradiation and immunotherapy: from concept to the clinic. Cancer 122:1659–1671. doi:10.1002/cncr.29889
Seiwert TY, Burtness B, Mehra R, Weiss J, Berger R, Eder JP, Heath K, McClanahan T, Lunceford J, Gause C, Cheng JD, Chow LQ (2016) Safety and clinical activity of pembrolizumab for treatment of recurrent or metastatic squamous cell carcinoma of the head and neck (KEYNOTE-012): an open-label, multicentre, phase 1b trial. Lancet Oncol 17:956–965. doi:10.1016/s1470-2045(16)30066-3
Topalian S, Hodi F, Brahmer J, Gettinger S, Smith D, McDermott D, Powderly J, Carvajal R, Sosman J, Atkins M, Leming P, Spigel D, Antonia S, Horn L, Drake C, Pardoll D, Chen L, Sharfman W, Anders R, Taube J, McMiller T, Xu H, Korman A, Jure-Kunkel M, Agrawal S, McDonald D, Kollia G, Gupta A, Wigginton J, Sznol M (2012) Safety, activity, and immune correlates of anti–PD-1 antibody in cancer. N Engl J Med 366:2443–2454. doi:10.1056/NEJMoa1200690
Sharabi AB, Lim M, DeWeese TL, Drake CG (2015) Radiation and checkpoint blockade immunotherapy: radiosensitisation and potential mechanisms of synergy. Lancet Oncol 16:e498–e509. doi:10.1016/s1470-2045(15)00007-8
Twyman-Saint Victor C, Rech AJ, Maity A, Rengan R, Pauken KE, Stelekati E, Benci JL, Xu B, Dada H, Odorizzi PM, Herati RS, Mansfield KD, Patsch D, Amaravadi RK, Schuchter LM, Ishwaran H, Mick R, Pryma DA, Xu X, Feldman MD, Gangadhar TC, Hahn SM, Wherry EJ, Vonderheide RH, Minn AJ (2015) Radiation and dual checkpoint blockade activate non-redundant immune mechanisms in cancer. Nature 520:373–377. doi:10.1038/nature14292
Weiner LM, Surana R, Wang S (2010) Monoclonal antibodies: versatile platforms for cancer immunotherapy. Nat Rev Immunol 10:317–327. doi:10.1038/nri2744
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Funding
The work was supported by the Department of Oral and Maxillofacial Surgery and the Institute of Pathology of the University Hospital Regensburg, Germany.
Ethical approval
The study was approved by the Ethics Committee of the University of Regensburg (Nr. 15-101-0336) and have been performed in accordance with the ethical standards as laid down in the 1964 Declaration of Helsinki and its later amendments or comparable ethical standards.
Informed consent
For this type of study, formal consent is not required.
Rights and permissions
About this article
Cite this article
Fiedler, M., Weber, F., Hautmann, M.G. et al. Biological predictors of radiosensitivity in head and neck squamous cell carcinoma. Clin Oral Invest 22, 189–200 (2018). https://doi.org/10.1007/s00784-017-2099-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00784-017-2099-x