Abstract
Objective
The aim of this study was to assess the presence of herpesviruses and periodontopathic bacteria and to establish their potential association with pericoronitis.
Materials and methods
Fifty samples obtained with paper points (30 from pericoronitis and 20 controls) were subjected to polymerase chain reaction (PCR) analysis. A single-stage and nested PCR assays were used to detect herpesviruses: human cytomegalovirus (HCMV) and Epstein-Barr virus (EBV) and six periodontopathic anaerobic bacteria: Aggregatibacter actinomycetemcomitans, Porphyromonas gingivalis, Prevotella intermedia, Parvimonas micra, Treponema denticola, and Tannarella forsythia.
Results
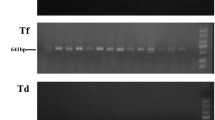
Pericoronitis samples harbored HCMV and EBV at significantly higher rates than the control group (70 vs. 40 % and 46.7 vs. 15 %, P = 0.035, P = 0.021, respectively). P. micra and T. forsythia (66.7 vs. 0 %, and 40 vs. 10 %, P = 0.001, P = 0.021, respectively) were significantly more common in pericoronitis compared to the control group. Multivariate logistic regression analysis showed that the presence of T. forsythia was associated with pericoronitis development (OR 7.3, 95 % CI, 1.2–43.2, P = 0.028).
Conclusion
The occurrence of HCVM and EBV extends our previous knowledge on microbiota in pericoronitis. These PCR-based findings demonstrated that bacterial and viral DNA occurred concomitantly in pericoronitis samples. T. forsythia appeared to be significantly associated with pericoronitis development in the examined sample.
Clinical relevance
Herpesviral-bacterial co-infections might exacerbate the progression of pericoronitis.
Similar content being viewed by others
References
Nitzan DW, Tal O, Sela MN, Shteyer A (1985) Pericoronitis: a reappraisal of its clinical and microbiologic aspects. J Oral Maxillofac Surg 43:510–516
Laine M, Ventä I, Hyrkäs T, Ma J, Konttinen YT (2003) Chronic inflammation around painless partially erupted third molars. Oral Surg Oral Med Oral Pathol Oral Radiol Endod 95:277–282
Leung WK, Theilade E, Comfort MB, Lim PL (1993) Microbiology of the pericoronal pouch in mandibular third molar pericoronitis. Oral Microbiol Immunol 8:306–312
Wade WG, Gray AR, Absi EG, Barker GR (1991) Predominant cultivable flora in pericoronitis. Oral Microbiol Immunol 6:310–312
Rajasuo A, Jousimies-Somer H, Savolainen S, Leppänen J, Murtomaa H, Meurman JH (1996) Bacteriologic findings in tonsillitis and pericoronitis. Clin Infect Dis 23:51–60
Sixou JL, Magaud C, Jolivet-Gougeon A, Cormier M, Bonnaure-Mallet M (2003) Evaluation of the mandibular third molar pericoronitis flora and its susceptibility to different antibiotics prescribed in France. J Clin Microbiol 41:5794–5797
Mombelli A, Buser D, Lang NP, Berthold H (1990) Suspected periodontopathogens in erupting third molar sites of periodontally healthy individuals. J Clin Periodontol 17:48–54
Rajasuo A, Sihvonen OJ, Peltola M, Meurman JH (2007) Periodontal pathogens in erupting third molars of periodontally healthy subjects. Int J Oral Maxillofac Surg 36:818–821
Sixou JL, Magaud C, Jolivet-Gougeon A, Cormier M, Bonnaure-Mallet M (2003) Microbiology of mandibular third molar pericoronitis: incidence of beta-lactamase-producing bacteria. Oral Surg Oral Med Oral Pathol Oral Radiol Endod 95:655–659
Peltroche-Llacsahuanga H, Reichhart E, Schmitt W, Lütticken R, Haase G (2000) Investigation of infectious organisms causing pericoronitis of the mandibular third molar. J Oral Maxillofac Surg 58:611–616
Mansfield JM, Campbell JH, Bhandari AR, Jesionowski AM, Vickerman MM (2012) Molecular analysis of 16S rRNA genes identifies potentially periodontal pathogenic bacteria and archaea in the plaque of partially erupted third molars. J Oral Maxillofac Surg 70:1507–1514.e1-6
Sencimen M, Saygun I, Gulses A, Bal V, Acikel CH, Kubar A (2014) Evaluation of periodontal pathogens of the mandibular third molar pericoronitis by using real time PCR. Int Dent J 64:200–205
Parra B, Slots J (1996) Detection of human viruses in periodontal pockets using polymerase chain reaction. Oral Microbiol Immunol 11:289–293
Yapar M, Saygun I, Ozdemir A, Kubar A, Sahin S (2003) Prevalence of human herpesviruses in patients with aggressive periodontitis. J Periodontol 74:1634–1640
Sabeti M, Valles Y, Nowzari H, Simon JH, Kermani-Arab V, Slots J (2003) Cytomegalovirus and Epstein-Barr virus DNA transcription in endodontic symptomatic lesions. Oral Microbiol Immunol 18:104–108
Sabeti M, Simon JH, Slots J (2003) Cytomegalovirus and Epstein-Barr virus are associated with symptomatic periapical pathosis. Oral Microbiol Immunol 18:327–328
Chen V, Chen Y, Li H, Kent K, Baumgartner JC, Machida CA (2009) Herpesviruses in abscesses and cellulitis of endodontic origin. J Endod 35:182–188
Slots J, Sabeti M, Simon JH (2003) Herpesviruses in periapical pathosis: an etiopathogenic relationship? Oral Surg Oral Med Oral Pathol Oral Radiol Endod 96:327–331
Slots J (2007) Herpesviral-bacterial synergy in the pathogenesis of human periodontitis. Curr Opin Infect Dis 20:278–283
Kamma JJ, Contreras A, Slots J (2001) Herpes viruses and periodontopathic bacteria in early-onset periodontitis. J Clin Periodontol 28:879–885
Saygun I, Kubar A, Ozdemir A, Yapar M, Slots J (2004) Herpesviral-bacterial interrelationships in aggressive periodontitis. J Periodontal Res 39:207–212
Botero JE, Parra B, Jaramillo A, Contreras A (2007) Subgingival human cytomegalovirus correlates with increased clinical periodontal parameters and bacterial coinfection in periodontitis. J Periodontol 78:2303–2310
Imbronito AV, Okuda OS, Maria de Freitas N, Moreira Lotufo RF, Nunes FD (2008) Detection of herpesviruses and periodontal pathogens in subgingival plaque of patients with chronic periodontitis, generalized aggressive periodontitis, or gingivitis. J Periodontol 79:2313–2321
Chalabi M, Rezaie F, Moghim S, Mogharehabed A, Rezaei M, Mehraban B (2010) Periodontopathic bacteria and herpesviruses in chronic periodontitis. Mol Oral Microbiol 25:236–240
Kato A, Imai K, Ochiai K, Ogata Y (2015) Prevalence and quantitative analysis of Epstein-Barr virus DNA and Porphyromonas gingivalis associated with Japanese chronic periodontitis patients. Clin Oral Investig 19:1605–1610
Sabeti M, Slots J (2004) Herpesviral-bacterial coinfection in periapical pathosis. J Endod 30:69–72
Ferreira DC, Rôças IN, Paiva SS, Carmo FL, Cavalcante FS, Rosado AS, Santos KR, Siqueira JF Jr (2011) Viral-bacterial associations in acute apical abscesses. Oral Surg Oral Med Oral Pathol Oral Radiol Endod 112:264–271
Verdugo F, Castillo A, Castillo F, Uribarri A (2015) Epstein-Barr virus associated peri-implantitis: a split-mouth study. Clin Oral Investig 19:535–543
Jakovljevic A, Andric M, Knezevic A, Soldatovic I, Nikolic N, Karalic D, Milasin J (2015) Human cytomegalovirus and Epstein-Barr virus genotypes in apical periodontitis lesions. J Endod 41:1847–1851
Vickerman MM, Brossard KA, Funk DB, Jesionowski AM, Gill SR (2007) Phylogenetic analysis of bacterial and archaeal species in symptomatic and asymptomatic endodontic infection. J Med Microbiol 56:110–118
Ashimoto A, Chen C, Bakker I, Slots J (1996) Polymerase chain reaction detection of 8 putative periodontal pathogens in subgingival plaque of gingivitis and advanced periodontitis lesions. Oral Microbiol Immunol 11:266–273
Slots J, Ashimoto A, Flynn MJ, Li G, Chen C (1995) Detection of putative periodontal pathogens in subgingival specimens by 16S ribosomal DNA amplification with the polymerase chain reaction. Clin Infect Dis 20:S304–S307
Conrads G, Soffner J, Pelz K, Mutters R (1997) Taxonomic update and clinical significance of species within the genus Peptostreptococcus. Clin Infect Dis 25:S94–S97
Baumgartner JC, Watkins BJ, Bae KS, Xia T (1999) Association of black-pigmented bacteria with endodontic infections. J Endod 25:413–415
Andric M, Milasin J, Jovanovic T, Todorovic L (2007) Human cytomegalovirus is present in odontogenic cysts. Oral Microbiol Immunol 22:347–351
Pucar A, Milasin J, Lekovic V, Vukadinovic M, Ristic M, Putnik S, Kenney EB (2007) Correlation between atherosclerosis and periodontal putative pathogenic bacterial infections in coronary and internal mammary arteries. J Periodontol 78:677–682
Aalto SM, Linnavuori K, Peltola H, Vuori E, Weissbrich B, Schubert J, Hedman L, Hedman K (1998) Immunoreactivation of Epstein-Barr virus due to cytomegalovirus primary infection. J Med Virol 56:186–191
Yoneda M, Hirofuji T, Anan H, Matsumoto A, Hamachi T, Nakayama K, Maeda K (2001) Mixed infection of Porphyromonas gingivalis and Bacteroides forsythus in a murine abscess model: involvement of gingipains in a synergistic effect. J Periodontal Res 36:237–243
Jakovljevic A, Andric M, Miletic M, Beljic-Ivanovic K, Knezevic A, Mojsilovic S, Milasin J (2016) Epstein-Barr virus infection induces bone resorption in apical periodontitis via increased production of reactive oxygen species. Med Hypotheses 94:40–42
Acknowledgments
This study was supported by the Ministry of Education, Science and Technological Development of Republic of Serbia, Grant No. 175075.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Funding
This study was supported by the Ministry of Education, Science and Technological Development of the Republic of Serbia, Grant No. 175075.
Ethical approval
This study has been approved by the Ethical Committee of the School of Dental Medicine, University of Belgrade (approval number: 36/27-2013), and has therefore been performed in accordance with the ethical standards laid down in 1964 Declaration of Helsinki and its later amendments.
Informed consent
All persons gave their written consent prior to their inclusion in the study.
Rights and permissions
About this article
Cite this article
Jakovljevic, A., Andric, M., Knezevic, A. et al. Herpesviral-bacterial co-infection in mandibular third molar pericoronitis. Clin Oral Invest 21, 1639–1646 (2017). https://doi.org/10.1007/s00784-016-1955-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00784-016-1955-4