Abstract
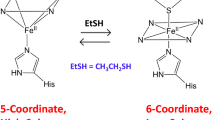
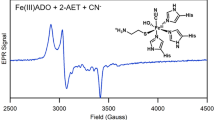
Thiol dioxygenases are a subset of non-heme mononuclear iron oxygenases that catalyze the O2-dependent oxidation of thiol-bearing substrates to yield sulfinic acid products. Cysteine dioxygenase (CDO) and 3-mercaptopropionic acid (3MPA) dioxygenase (MDO) are the most extensively characterized members of this enzyme family. As with many non-heme mononuclear iron oxidase/oxygenases, CDO and MDO exhibit an obligate-ordered addition of organic substrate before dioxygen. As this substrate-gated O2-reactivity extends to the oxygen-surrogate, nitric oxide (NO), EPR spectroscopy has long been used to interrogate the [substrate:NO:enzyme] ternary complex. In principle, these studies can be extrapolated to provide information about transient iron-oxo intermediates produced during catalytic turnover with dioxygen. In this work, we demonstrate that cyanide mimics the native thiol-substrate in ordered-addition experiments with MDO cloned from Azotobacter vinelandii (AvMDO). Following treatment of the catalytically active Fe(II)-AvMDO with excess cyanide, addition of NO yields a low-spin (S = 1/2) (CN/NO)-Fe-complex. Continuous wave and pulsed X-band EPR characterization of this complex produced in wild-type and H157N variant AvMDO reveal multiple nuclear hyperfine features diagnostic of interactions within the first- and outer-coordination sphere of the enzymatic Fe-site. Spectroscopically validated computational models indicate simultaneous coordination of two cyanide ligands replaces the bidentate (thiol and carboxylate) coordination of 3MPA allowing for NO-binding at the catalytically relevant O2-binding site. This promiscuous substrate-gated reactivity of AvMDO with NO provides an instructive counterpoint to the high substrate-specificity exhibited by mammalian CDO for l-cysteine.
Graphical abstract








Similar content being viewed by others
Data availability
The datasets generated during and/or analysed during the current study are available from the corresponding author on reasonable request.
Abbreviations
- CDO:
-
Cysteine dioxygenase
- MDO:
-
3-Mercaptopropionic acid dioxygenase
- 3MPA :
-
3-Mercaptopropionic acid
- CYS :
-
L-cysteine
- CA :
-
Cysteamine (2-aminoethanethiol)
- ET :
-
Ethanethiol
- β-Ala:
-
β-Alanine
- 3BPA :
-
3-Bromopropionic acid
- 3IPA :
-
3-Iodopropionic acid
- CSA :
-
Cysteine sulfinic acid
- HT :
-
Hypotaurine
- 3SPA :
-
3-Sulfinopropionic acid
- Av :
-
Azotobacter vinelandii
- CW:
-
Continuous wave
- FT:
-
Fourier transform (or pulsed): EPR: electron paramagnetic resonance
- HYSCORE:
-
Hyperfine sublevel correlation spectroscopy
- DFT:
-
Density functional theory
References
Dominy JE, Simmons CR, Hirschberger LL, Hwang J, Coloso RM, Stipanuk MH (2007) J Biol Chem 282:25189–25198
Stipanuk M, Simmons C, Andrew Karplus A, Dominy J (2010) Amino Acids, 1–12
Ewetz L, Sorbo B (1966) Biochim Biophys Acta 128:296–305
Sorbo B, Ewetz L (1965) Biochem Biophys Res Commun 18:359–363
Lombardini JB, Singer TP, Boyer PD (1969) J Biol Chem 244:1172–1175
Gordon C, Emery P, Bradley H, Waring H (1992) Lancett 229:25–26
Dominy JE Jr, Simmons CR, Karplus PA, Gehring AM, Stipanuk MH (2006) J Bacteriol 188:5561–5569
Heafield MT, Fearn S, Steventon GB, Waring RH, Williams AC, Sturman SG (1990) Neurosci Lett 110:216–220
Sarkar B, Kulharia M, Mantha AK (2017) Int J Exp Pathol 98:52–66
Brown CD, Neidig ML, Neibergall MB, Lipscomb JD, Solomon EI (2007) J Am Chem Soc 129:7427–7438
Yamaguchi K, Sakakibara S, Koga K, Ueda I (1971) Biochim Biophys Acta Gen Subj 237:502–512
Perry TL, Norman MG, Yong VW, Whiting S, Crichton JU, Hansen S, Kish SJ (1985) Ann Neurol 18:482–489
Bradley H, Gough A, Sokhi RS, Hassell A, Waring R, Emery P (1994) J Rheumatol 21:1192–1196
Deth R, Muratore C, Benzecry J, Power-Charnitsky V-A, Waly M (2008) Neurotoxicology 29:190–201
James SJ, Cutler P, Melnyk S, Jernigan S, Janak L, Gaylor DW, Neubrander JA (2004) Am J Clin Nutr 80:1611–1617
Reddie KG, Carroll KS (2008) Curr Opin Chem Biol 12:746–754
Winyard PG, Moody CJ, Jacob C (2005) Trends Biochem Sci 30:453–461
Trachootham D, Alexandre J, Huang P (2009) Nat Rev Drug Discov 8:579–591
Behave DP, Muse WB, Carroll KS (2007) Infect Disord Drug Targets 7:140–158
Gunawardana DM, Heathcote KC, Flashman E (2021) FEBS J
Weits DA, Giuntoli B, Kosmacz M, Parlanti S, Hubberten H-M, Riegler H, Hoefgen R, Perata P, van Dongen JT, Licausi F (2014) Nat Commun 5:3425
White MD, Klecker M, Hopkinson RJ, Weits DA, Mueller C, Naumann C, O’Neill R, Wickens J, Yang J, Brooks-Bartlett JC, Garman EF, Grossmann TN, Dissmeyer N, Flashman E (2017) Nat Commun 8:14690
Masson N, Keeley TP, Giuntoli B, White MD, Puerta ML, Perata P, Hopkinson RJ, Flashman E, Licausi F, Ratcliffe PJ (2019) Science 365:65–69
Fernandez RL, Elmendorf LD, Smith RW, Bingman CA, Fox BG, Brunold TC (2021) Biochemistry 60:3728–3737
Wang Y, Shin I, Li J, Liu A (2021) J Biol Chem 297:101176
Tchesnokov EP, Fellner M, Siakkou E, Kleffmann T, Martin LW, Aloi S, Lamont IL, Wilbanks SM, Jameson GNL (2015). J Biol Chem. https://doi.org/10.1074/jbc.M114.635672
Tchesnokov EP, Faponle AS, Davies CG, Quesne MG, Turner R, Fellner M, Souness RJ, Wilbanks SM, de Visser SP, Jameson GNL (2016) Chem Commun (Camb) 52:8814–8817
Crowell JK, Sardar S, Hossain MS, Foss FW, Pierce BS (2016) Arch Biochem Biophys 604:86–94
Pierce BS, Subedi BP, Sardar S, Crowell JK (2015) Biochemistry 54:7477–7490
York NJ, Lockart MM, Sardar S, Khadka N, Shi W, Stenkamp RE, Zhang J, Kiser PD, Pierce BS (2021) J Biol Chem 296:100496
York NJ, Lockart MM, Pierce BS (2021) Inorg Chem 60:18639–18651
Sardar S, Weitz A, Hendrich MP, Pierce BS (2019) Biochemistry 58:5135–5150
Li W, Blaesi EJ, Pecore MD, Crowell JK, Pierce BS (2013) Biochemistry 52:9104–9119
Fernandez RL, Juntunen ND, Brunold TC (2022) Acc Chem Res 55:2480–2490
Weitz AC, Giri N, Caranto JD, Kurtz DM, Bominaar EL, Hendrich MP (2017) J Am Chem Soc 139:12009–12019
Tierney DL, Rocklin AM, Lipscomb JD, Que L, Hoffman BM (2005) J Am Chem Soc 127:7005–7013
Casey TM, Grzyska PK, Hausinger RP, McCracken J (2013) J Phys Chem B 117:10384–10394
Li J, Koto T, Davis I, Liu A (2019) Biochemistry 58:2218–2227
McCracken J, Eser BE, Mannikko D, Krzyaniak MD, Fitzpatrick PF (2015) Biochemistry 54:3759–3771
Pierce BS, Gardner JD, Bailey LJ, Brunold TC, Fox BG (2007) Biochemistry 46:8569–8578
McQuilken AC, Ha Y, Sutherlin KD, Siegler MA, Hodgson KO, Hedman B, Solomon EI, Jameson GNL, Goldberg DP (2013) J Am Chem Soc 135:14024–14027
Stoll S, Schweiger A (2006) J Magn Reson 178:42–55
Neese F (2017) Wiley Interdiscip Rev Comput Mol Sci 8:e1327
Tao J, Perdew JP, Staroverov VN, Scuseria GE (2003) Phys Rev Lett 91:146401
Weigend F, Ahlrichs R (2005) Phys Chem Chem Phys 7:3297–3305
Grimme S, Antony J, Ehrlich S, Krieg H (2010) J Chem Phys 132:154104
Vahtras O, Almlöf J, Feyereisen MW (1993) Chem Phys Lett 213:514–518
Kendall RA, Früchtl HA (1997) Theor Chem Acc 97:158–163
Cossi M, Rega N, Scalmani G, Barone V (2003) J Comput Chem 24:669–681
Staroverov VN, Scuseria GE, Tao J, Perdew JP (2003) J Chem Phys 119:12129–12137
Römelt M, Ye S, Neese F (2009) Inorg Chem 48:784–785
Rega N, Cossi M, Barone V (1996) J Chem Phys 105:11060–11067
Timoshin AA, Vanin AF, Orlova TR, Sanina NA, Ruuge EK, Aldoshin SM, Chazov EI (2007) Nitric Oxide 16:286–293
Vanin AF (2018) Cell Biochem Biophys 76:3–17
Vanin AF, Serezhenkov VA, Mikoyan VD, Genkin MV (1998) Nitric Oxide 2:224–234
Ye S, Neese F (2010) J Am Chem Soc 132:3646–3647
Sellmann D, Blum N, Heinemann FW, Hess BA (2001) Chem Eur J 7:1874–1880
Enemark JH, Feltham RD (1974) Coord Chem Rev 13:339–406
Lewandowska H, Kalinowska M, Brzóska K, Wójciuk K, Wójciuk G, Kruszewski M (2011) Dalton Trans 40:8273–8289
Truzzi DR, Augusto O, Iretskii AV, Ford PC (2019) Inorg Chem 58:13446–13456
Reginato N, McCrory CTC, Pervitsky D, Li L (1999) J Am Chem Soc 121:10217–10218
Tsai F-T, Kuo T-S, Liaw W-F (2009) J Am Chem Soc 131:3426–3427
Blaesi EJ, Gardner JD, Fox BG, Brunold TC (2013) Biochemistry 52:6040–6051
Li M, Bonnet D, Bill E, Neese F, Weyhermèuller T, Blum N, Sellmann D, Wieghardt K (2002) Inorg Chem 41:3444–3456
Orville AM, Lipscomb JD (1997) Biochemistry 36:14044–14055
Stevens TH, Chan SI (1981) J Biol Chem 256:1069–1071
Yonetani T, Yamamoto H, Erman JE, Leigh JS, Reed GH (1972) J Biol Chem 247:2447–2455
Fischer AA, Miller JR, Jodts RJ, Ekanayake DM, Lindeman SV, Brunold TC, Fiedler AT (2019) Inorg Chem 58:16487–16499
Abragam A, Bleaney B (1970) Electron paramagnetic resonance of transition ions (International series of monographs on physics)
Dikanov SA, Davydov RM, Gräslund A, Bowman MK (1998) J Am Chem Soc 120:6797–6805
Forlano PM, Olabe JA, Magallanes JF, Blesa MA (1997) Can J Chem 75:9–13
Traore ES, Liu A (2022) ACS Catal 12:6191–6208
Acknowledgements
This work was supported by the National Institutes of Health (2 R15 GM117511-03) to B.S.P.
Author information
Authors and Affiliations
Contributions
All authors contributed to the writing of this text and have given their approval to the final version of the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
York, N.J., Lockart, M.M., Schmittou, A.N. et al. Cyanide replaces substrate in obligate-ordered addition of nitric oxide to the non-heme mononuclear iron AvMDO active site. J Biol Inorg Chem 28, 285–299 (2023). https://doi.org/10.1007/s00775-023-01990-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00775-023-01990-7