Abstract
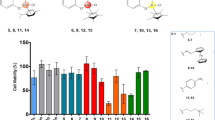
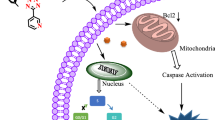
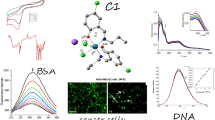
Six novel ruthenium(III) complexes of general formula [RuCl3(L)3] (1,3,5) and [RuCl3(H2O)(L)2] (2,4,6), where L stands for three different triazolopyrimidine-derived ligands, are reported. The compounds have been structurally characterized (IR, EPR, SCXRD), and their magnetic moments have been determined. The single-crystal X-ray diffraction study revealed a slightly distorted octahedral geometry of the Ru(III) complexes with mer configuration in 1 and 5, and fac configuration in 3. In 2 and 4, three chloride ions are in mer configuration and the two triazolopyrimidines are oriented trans mutually with the water molecule playing the role of the sixth ligand. All complexes have been thoroughly screened for their in vitro cytotoxicity against human breast cancer cell line MCF-7, human cervical cancer cell line HeLa, and L929 murine fibroblast cells, uncovering among others that the most lipophilic complexes 5 and 6, containing the bulky ligand dptp (5,7-diphenyl-1,2,4-triazolo[1,5-a]pyrimidine), display high cytotoxic activity against MCF-7, and HeLa cells. Moreover, it was also revealed that during the interaction of the complexes 1–6 with the cancer MCF-7 cell line, reactive oxygen species are released intracellularly, which could indicate that they are involved in cell apoptosis. Furthermore, extensive studies have been carried out to reveal the mechanism by which complexes 1–6 interact with DNA, albumin, and apotransferrin. The biological studies were complemented by detailed kinetic studies of the hydrolysis of the complexes in the pH range 5–8, to determine the stability of the complexes in solution.
Graphic abstract
Six novel ruthenium(III) complexes with triazolopyrimidine derivatives demonstrated the potential for use as anticancer agents by maintaining the toxic effect on MCF-7 and HeLa cells.











Similar content being viewed by others
References
Zamble DB, Lippard SJ (1995) Trends Biochem Sci 20:435–439
Sasada T, Iwata S, Sato N, Kitaoka Y, Hirota K, Nakamura K, Nishiyama A, Taniguchi Y, Takabayashi A, Yodoi J (1996) J Clin Invest 97:2268–2276
Shen DW, Pouliot LM, Hall MD, Gottesman MM (2012) Pharmacol Rev 64(3):706–721
Antonarakis ES, Emadi A (2010) Cancer Chemother Pharmacol 66:1–9
Sava G, Zorzet S, Giraldi T, Mestroni G, Zassinovich G (1984) Eur J Cancer Clin Oncol 20(6):841–847
Sava G, Bergamo A (2000) Int J Oncol 17(2):353–365
Schluga P, Hartinger CG, Egger A, Reisner E, Galanski M, Jakupec MA, Keppler BK (2006) Dalton Trans 14:1796–1802
Reedijk J (2003) Proc Natl Acad Sci USA 100(7):3611–3616
Clarke MJ, Zhu F, Frasca DR (1999) Chem Rev 99(9):2511–2534
Gagliardi R, Sava G, Pacor S, Mestroni G, Alessio E (1994) Clin Exp Metastasis 12(2):93–100
Bergamo A, Masi A, Dyson PJ, Sava G (2008) Int J Oncol 33(6):1281–1289
Schäfer S, Ott I, Gust R, Sheldrick WS (2007) Eur J Inorg Chem 19:3034–3046
Pluim D, van Waardenburg RC, Beijnen JH, Schellens JH (2004) Cancer Chemother Pharmacol 54(1):71–78
Kapitza S, Jakupec MA, Uhl M, Keppler BK, Marian B (2005) Cancer Lett 226(2):115–121
Chen H, Parkinson JA, Morris RE, Sadler PJ (2003) J Am Chem Soc 125(1):173–186
Kostova I (2006) Curr Med Chem 13(9):1085–1107
Sava G, Alessio E, Bergamo A, Mestroni G (1999) Top Biol Inorg Chem 1:143–169
Pieper T, Borsky K, Keppler BK (1999) Top Biol Inorg Chem 1:171–199
Sava G, Pacor S, Mestroni G, Alessio E (1992) Clin Exp Metastasis 10(4):273–280
Sava G, Pacor S, Bergamo A, Cocchietto M, Mestroni G, Alessio E (1995) Chem Biol Interact 95(1–2):109–126
Jakupec MA, Arion VB, Kapitza S, Reisner E, Eichinger A, Pongratz M, Marian B, von Keyserlingk Graf N, Keppler BK (2005) Int J Clin Pharmacol Ther 43(12):595–596
Hartinger CG, Jakupec MA, Zorbas-Seifried S, Groessl M, Egger A, Berger W, Zorbas H, Dyson PJ, Keppler BK (2008) Chem Biodivers 5(10):2140–2155
Łakomska I, Fandzloch M (2016) Coord Chem Rev 327–328:221–241
Salas JM, Romero MA, Purificación Sánchez M, Quirós M (1999) Coord Chem Rev 193–195:1119–1142
Pyatakov DA, Sokolov AN, Astakhov AV, Chernenko AY, Fakhrutdinov AN, Rybakov VB, Chernyshev VV, Chernyshev VM (2015) J Org Chem 80(21):10694–10709 (references therein)
Lauria A, Abbate I, Patella C, Martorana A, Dattolo G, Almerico AM (2013) Eur J Med Chem 62:416–424
Said SA, El-Galil A, Amr E, Sabry NM, Abdalla MM (2009) Eur J Med Chem 44:4787–4792
Ganesana SM, Morriseya JM, Kea H, Paintera HJ, Laroiyaa K, Phillips MA, Rathod PK, Mathera MW, Vaidya AB (2011) Mol Biochem Parasitol 177:29–34
Trianaa MAH, Huynhb M-H, Garavitoa MF, Foxc BA, Bzikc DJ, Carruthersb VB, Löfflerd M, Zimmermanna BH (2012) Mol Biochem Parasitol 184:71–81
Ojha PK, Roy K (2010) Eur J Med Chem 45:4645–4656
Da Silva ER, Boechat N, Pinheiro LCS, Bastos MM, Costa CCP, Bartholomeu JC, da Costa TH (2015) Chem Biol Drug Des 5:969–978
Caballero AB, Salas JM, Sánchez-Moreno M (2014) In: Claborn DM (ed) Leishmaniasis—trends in epidemiology, diagnosis and treatment. IntechOpen, London
El-Gendy MM, Shaaban M, Shaaban KA, El-Bondkly AM, Laatsch H (2008) J Antibiot 61:149–157
Chen Q, Zhu X-L, Jiang L-L, Liu Z-M, Yang GF (2008) Eur J Med Chem 43:595–603
Massari S, Nannetti G, Desantis J, Muratore G, Sabatini S, Manfroni G, Mercorelli B, Cecchetti V, Palù G, Cruciani G, Loregian A, Goracci L, Tabarrini O (2015) J Med Chem 58:3830–3842
Huang B, Li C, Chen W, Liu T, Yu M, Fu L, Sun Y, Liu H, De Clercq E, Pannecouque C, Balzarini J, Zhan P, Liu X (2015) Eur J Med Chem 92:754–765
Brigance RP, Meng W, Fura A, Harrity T, Wang A, Zahler R, Kirby MS, Hamann LG (2010) Bioorg Med Chem Lett 20:4395–4398
Cornec AS, James MJ, Kovalevich J, Trojanowski JQ, Lee VMY, Smith AB III, Ballatore C, Brunden KR (2015) Bioorg Med Chem Lett 25:4980–4982
Faizi M, Dabirian S, Tajali H, Ahmadi F, Zavareh ER, Shahhosseini S, Tabatabai SA (2015) Bioorg Med Chem 23:480–487
Saito T, Obitsu T, Minamoto C, Sugiura T, Matsumura N, Ueno S, Kishi A, Katsumata S, Nakai H, Toda M (2011) Bioorg Med Chem 19:5955–5966
Ohnishi H, Yamaguchi K, Shimada S, Suzuki Y, Kumagai A (1981) Life Sci 28:1641–1646
Zhou L, Schandené L, Mordvinov VA, Chatelain P, Pradier O, Goldman M, Stordeur P (2004) Int Immunopharmacol 4:863–871
Rodríguez-Torres M, Yoshida EM, Marcellin P, Srinivasan S, Purohit VS, Wang C, Hammond JL (2014) Ann Hepatol 13:364–375
Łakomska I, Fandzloch M, Muzioł T, Lis T, Jezierska J (2013) Dalton Trans 42:6219–6226
Velders AH, Pazderski L, Ugozzoli F, Biagini-Cingi M, Manotti-Lanfredi AM, Haasnoot JG, Reedijk J (1998) Inorg Chim Acta 273:259–265
Bülow C, Haas K (1909) Berichte 42:4638–4644
Grodzicki A, Szłyk E, Pazderski L, Goliński A (1996) Magn Res Chem 34:725–727
Günay E, Mutikainen I, Turpeinen U, van Albada GA, Haasnoot JG, Reedijk J (2010) J Chem Crystallogr 40:1006–1010
Balkaran JM, van Bezouw SCP, van Rruchem J, Verasdonck J, Verkerk PC, Volbeda AG, Mutikainen I, Turpeinen U, van Albada GA, Gamez P, Balkaran JM, van Bezouw SCP, van Rruchem J, Verasdonck J, Verkerk PC, Volbeda AG, Mutikainen I, Turpeinen U, van Albada GA, Gamez P, Haasnoot JG, Reedijk J (2009) Inorg Chim Acta 362:861–868
Rigaku Oxford Diffraction (2015) CrysAlisPro Software System version 1.171. 38.43d. Rigaku Corporation, Oxford
SMART (Version 5.628) (2002) Data Collection Software. Bruker Analytical X-ray Instruments Inc, Madison
SAINT+ (Version 6.45) (2003) Data Reduction Software. Bruker Analytical X-ray Instruments Inc, Madison
Sheldrick GM SADABS (version 2.03) (2002) Program for empirical absorption correction of area detector data. University of Göttingen, Göttingen
Sheldrick GM (2008) A short history of SHELX. Acta Crystallogr A64:112–122. https://doi.org/10.1107/S0108767307043930
Macrae CF, Bruno IJ, Chisholm JA, Edgington PR, McCabe P, Pidcock E, Rodriguez-Monge L, Taylor R, Van De Streek J, Wood PA (2008) Mercury CSD 2.0—new features for the visualization and investigation of crystal structures. J Appl Crystallogr 41:466–470. https://doi.org/10.1107/s0021889807067908
Persistence of Vision Pty. Ltd. (2004) Persistence of Vision (TM) Raytracer. Persistence of Vision Pty. Ltd., Williamstown, Victoria, Australia. http://www.povray.org/
Albert A (1979) Selective toxicity the physico-chemical basis of therapy. Chapman and Hall, London, pp 662–664
Mestroni G, Alessio E, Sava G (1998) New salts of anionic complexes of Ru(III) as antimetastatic and antineoplastic agents. International Patent WO 98/00431
Muzioł T, Wiśniewska J (2015) CSD Communication
Velders AH, Ugozzoli F, Biagini-Cingi M, Manotti-Lanfredi AM, Haasnoot JG, Reedijk J (1999) Eur J Inorg Chem 213–215
Łakomska I, Stefańczak K, Fandzloch M, Sitkowski J, Filip-Psurska B, Wojtczak A (2016) There is one crystal structure of Ru(II) with ibmtp, namely: dichloro-(7-isobutyl-5-methyl[1,2,4]triazolo[1,5-a]pyrimidine)-(η6-1-isopropyl-4-methylbenzene)-ruthenium (TUZROX). Polyhedron 109:33–39
Medhi K, Agarwala U (1980) Inorg Chem 19:1381–1384
LaChance-Galang KJ, Doan PE, Clarke MJ, Rae U, Yamano A, Hoffman BM (1995) J Am Chem Soc 117:3529–3538
Bleaney B, O’Brien MCM (1956) Proc Phys Soc Lond 69B:1216–1230
McGarvey BR (1998) Quim Nova 21:206–213
McGarvey BR (1998) Coord Chem Rev 170:5–92
Fandzloch M, Méndez Arriaga JM, Sánchez-Moreno M, Wojtczak A, Sitkowski J, Wiśniewska J, Salas JM, Łakomska I (2017) J Inorg Biochem 176:144–155
Moreno V, Lorenzo J, Aviles FX, Garcia MH, Ribeiro JP, Morais TS, Florindo P, Robalo MP (2010) Bioinorg Chem Appl 1–11
Fandzloch M (2015) PhD thesis/unpublished
Simon HU, Haj-Yehia A, Levi-Schaffer F (2000) Apoptosis 5:415–418
Arai M, Shibata Y, Pugdee K, Abiko Y, Ogata Y (2007) IUBMB Life 59:27–33
Cao W, Zheng W, Chen T (2015) Sci Rep 5:1–11
Luo Z, Yu L, Yang F, Zhao Z, Yu B, Lai H, Wong KH, Ngai SM, Zheng W, Chen T (2014) Metallomics 6:1480–1490
Vijayalakshmi R, Kanthimathi M, Parthsarathi R, Nair BU (2006) Bioorg Med Chem 14:3300–3306
Messori L, Orioli P, Vullo D, Alessio E, Iengo E (2000) Eur J Biochem 267:1206–1213
Sun H, Li H, Sadler PJ (1999) Chem Rev 99:2817–2842
Young SP, Bomford A, Williams R (1984) Biochem J 219:505–510
Lincoln P, Tuite E, Norden B (1997) J Am Chem Soc 119:1454–1455
Shahabadi N, Kashanian S, Darabi F (2010) J Med Chem 45:4239–4245
Martínez A, Suárez J, Shand T, Magliozzo RS, Sánchez-Delgado RA (2011) J Inorg Biochem 105:39–45
Acknowledgements
Financial support from the National Science Center (NCN) Poland through Grant Etiuda no. 2013/08/T/ST5/00391 (to M.F.) is gratefully acknowledged. M. F. is thankful to J. Masternak, DSc PhD from the Jan Kochanowski University in Kielce for magnetic measurements and Prof. Juan M. Salas from the University of Granada for support and advice during the internship. His extraordinary personality and great mentoring will forever remain in my memory.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
There are no conflicts of interest to declare.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
775_2019_1743_MOESM1_ESM.doc
Supplementary material 1 (DOC 1487 kb) [Table S1 presented selected bond lengths and angles for 1–4, representation (Fig. S1) and crystal data for 5, Fig. S2 and Fig. S3 with concentration-dependent effect of the ruthenium complexes 1–6 on the viability of L929 fibroblasts and MCF-7 cells or HeLa in comparison to both cell lines stimulated with cisplatin, and Fig. S4 presented a summary of the concentration-dependent impact of ruthenium complexes 1–6, cisplatin and NAMI-A on cell viability and intracellular ROS production]. See https://doi.org/10.1039/x0xx00000x
Rights and permissions
About this article
Cite this article
Fandzloch, M., Dobrzańska, L., Jędrzejewski, T. et al. Synthesis, structure and biological evaluation of ruthenium(III) complexes of triazolopyrimidines with anticancer properties. J Biol Inorg Chem 25, 109–124 (2020). https://doi.org/10.1007/s00775-019-01743-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00775-019-01743-5