Abstract
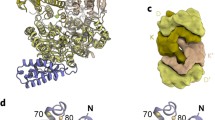
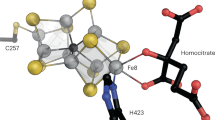
Nitrogenases catalyze the biological fixation of inert N2 into bioavailable ammonium. They are bipartite systems consisting of the catalytic dinitrogenase and a complementary reductase, the Fe protein that is also the site where ATP is hydrolyzed to drive the reaction forward. Three different subclasses of dinitrogenases are known, employing either molybdenum, vanadium or only iron at their active site cofactor. Although in all these classes the mode and mechanism of interaction with Fe protein is conserved, each one encodes its own orthologue of the reductase in the corresponding gene cluster. Here we present the 2.2 Å resolution structure of VnfH from Azotobacter vinelandii, the Fe protein of the alternative, vanadium-dependent nitrogenase system, in its ADP-bound state. VnfH adopts the same conformation that was observed for NifH, the Fe protein of molybdenum nitrogenase, in complex with ADP, representing a state of the functional cycle that is ready for reduction and subsequent nucleotide exchange. The overall similarity of NifH and VnfH confirms the experimentally determined cross-reactivity of both ATP-hydrolyzing reductases.




Similar content being viewed by others
Abbreviations
- EPR:
-
Electron paramagnetic resonance
- MR:
-
Molecular replacement
References
Rees DC (1993) Curr Opin Struct Biol 3:921–928
Leigh GJ (2004) The world’s greatest fix: A history of nitrogen in agriculture. Oxford University Press, Oxford
Smil V (2002) Ambio 31:126–131
Rees DC, Tezcan FA, Haynes CA, Walton MY, Andrade S, Einsle O, Howard JB (2005) Philos Trans R Soc Lond A 363:971–984
Howard JB, Rees DC (1996) Chem Rev 96:2965–2982
Georgiadis MM, Komiya H, Chakrabarti P, Woo D, Kornuc JJ, Rees DC (1992) Science 257:1653–1659
Burgess BK, Lowe DJ (1996) Chem Rev 96:2983–3011
Grossmann JG, Hasnain SS, Yousafzai FK, Smith BE, Eady RR, Schindelin H, Kisker C, Howard JB, Tsuruta H, Muller J, Rees DC (1999) Acta Crystallogr D 55:727–728
Peters JW, Fisher K, Newton WE, Dean DR (1995) J Biol Chem 270:27007–27013
Angove HC, Yoo SJ, Münck E, Burgess BK (1999) J Inorg Biochem 74:65
Danyal K, Dean DR, Hoffman BM, Seefeldt LC (2011) Biochemistry 50:9255–9263
Hoffman BM, Lukoyanov D, Yang ZY, Dean DR, Seefeldt LC (2014) Chem Rev 114:4041–4062
Howard JB, Kechris KJ, Rees DC, Glazer AN (2013) Multiple amino acid sequence alignment nitrogenase component 1: insights into phylogenetics and structure-function relationships. Plos One 8:e72751
Einsle O (2014) J Biol Inorg Chem 19:737–745
Spatzal T, Aksoyoğlu M, Zhang LM, Andrade SLA, Schleicher E, Weber S, Rees DC, Einsle O (2011) Science 334:940
Lancaster KM, Roemelt M, Ettenhuber P, Hu YL, Ribbe MW, Neese F, Bergmann U, DeBeer S (2011) Science 334:974–977
Sippel D, Einsle O (2017) Nat Chem Biol 13:956–960
Lee CC, Hu YL, Ribbe MW (2010) Science 329:642
Sippel D, Rohde M, Netzer J, Trncik C, Gies J, Grunau K, Djurdjevic I, Decamps L, Andrade SLA, Einsle O (2018) Science 359:1484–1489
Zheng Y, Harris DF, Yu Z, Fu Y, Poudel S, Ledbetter RN, Fixen KR, Yang ZY, Boyd ES, Lidstrom ME, Seefedt LC, Harwood CS (2018) Nat Microbiol 3:281–286
Setubal JC, dos Santos P, Goldman BS, Ertesvag H, Espin G, Rubio LM, Valla S, Almeida NF, Balasubramanian D, Cromes L, Curatti L, Du ZJ, Godsy E, Goodner B, Hellner-Burris K, Hernandez JA, Houmiel K, Imperial J, Kennedy C, Larson TJ, Latreille P, Ligon LS, Lu J, Maerk M, Miller NM, Norton S, O’Carroll IP, Paulsen I, Raulfs EC, Roemer R, Rosser J, Segura D, Slater S, Stricklin SL, Studholme DJ, Sun J, Viana CJ, Wallin E, Wang BM, Wheeler C, Zhu HJ, Dean DR, Dixon R, Wood D (2009) J Bacteriol 191:4534–4545
Dos Santos PC, Fang Z, Mason SW, Setubal JC, Dixon R (2012) Distribution of nitrogen fixation and nitrogenase-like sequences amongst microbial genomes. BMC Genom 13:162–174
Owens CP, Katz FEH, Carter CH, Oswald VF, Tezcan FA (2016) J Am Chem Soc 138:10124–10127
Hales BJ, Langosch DJ, Case EE (1986) J Biol Chem 261:5301–5306
Schlesier J, Rohde M, Gerhardt S, Einsle O (2016) J Am Chem Soc 138:239–247
Lipman JG (1903) Rep N J Agric Exp Stn 24:217–285
Burk D, Lineweaver H (1930) J Bacteriol 19:389–414
Sippel D, Schlesier J, Rohde M, Trncik C, Decamps L, Djurdjevic I, Spatzal T, Andrade SLA, Einsle O (2017) J Biol Inorg Chem 22:161–168
Kabsch W (2010) Acta Crystallogr D 66:125–132
Winn MD, Ballard CC, Cowtan KD, Dodson EJ, Emsley P, Evans PR, Keegan RM, Krissinel EB, Leslie AGW, McCoy A, McNicholas SJ, Murshudov GN, Pannu NS, Potterton EA, Powell HR, Read RJ, Vagin A, Wilson KS (2011) Acta Crystallogr D 67:235–242
McCoy AJ, Grosse-Kunstleve RW, Adams PD, Winn MD, Storoni LC, Read RJ (2007) J Appl Crystallogr 40:658–674
Emsley P, Lohkamp B, Scott WG, Cowtan K (2010) Acta Crystallogr D 66:486–501
Blanc E, Roversi P, Vonrhein C, Flensburg C, Lea SM, Bricogne G (2004) Acta Crystallogr D 60:2210–2221
Schrödinger LLC (2010) The PyMOL molecular graphics system
Jurrus E, Engel D, Star K, Monson K, Brandi J, Felberg LE, Brookes DH, Wilson L, Chen JH, Liles K, Chun MJ, Li P, Gohara DW, Dolinsky T, Konecny R, Koes DR, Nielsen JE, Head-Gordon T, Geng WH, Krasny R, Wei GW, Holst MJ, McCammon JA, Baker NA (2018) Protein Sci 27:112–128
Schindelin H, Kisker C, Sehlessman JL, Howard JB, Rees DC (1997) Nature 387:370–376
Tezcan FA, Kaiser JT, Mustafi D, Walton MY, Howard JB, Rees DC (2005) Science 309:1377–1380
Owens CP, Katz FE, Carter CH, Luca MA, Tezcan FA (2015) J Am Chem Soc 137:12704–12712
Danyal K, Shaw S, Page TR, Duval S, Horitani M, Marts AR, Lukoyanov D, Dean DR, Raugei S, Hoffman BM, Seefeldt LC, Antony E (2016) Proc Natl Acad Sci USA 113:E5783–E5791
Kaiser JT, Hu YL, Wiig JA, Rees DC, Ribbe MW (2011) Science 331:91–94
Hu Y, Ribbe MW (2011) Coord Chem Rev 255:1218–1224
Hu YL, Fay AW, Lee CC, Wiig JA, Ribbe MW (2010) Dalton T 39:2964–2971
Weiss M, Hilgenfeld R (1997) J Appl Crystallogr 30:203–205
Karplus PA, Diederichs K (2012) Science 336:1030–1033
Cruickshank DWJ (1999) Acta Crystallogr D 55:583–601
Laskowski RA, MacArthur MW, Moss DS, Thornton JM (1993) J Appl Crystallogr 26:283–291
Acknowledgements
The authors thank the staff at beam line X06DA of the Swiss Light Source, Paul Scherrer Institute, Villigen, CH, for their excellent assistance with diffraction data collection. This work was supported by the Deutsche Forschungsgemeinschaft (RTG 1976 and PP 1927) and the European Research Council (Grant no. 310656).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Rohde, M., Trncik, C., Sippel, D. et al. Crystal structure of VnfH, the iron protein component of vanadium nitrogenase. J Biol Inorg Chem 23, 1049–1056 (2018). https://doi.org/10.1007/s00775-018-1602-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00775-018-1602-4