Abstract
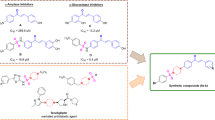
Adenosine deaminase (ADA) is a Zn2+-containing enzyme that catalyzes the irreversible deamination of adenosine to inosine or deoxyadenosine to deoxyinosine. In addition to this enzymatic function, ADA mediates cell-to-cell interactions involved in lymphocyte co-stimulation or endothelial activation. ADA is implicated in cardiovascular pathologies such as atherosclerosis and certain types of cancers, including lymphoma and leukemia. To date, only two drugs (pentostatin and cladribine) have been approved for the treatment of hairy cell leukemia. In search of natural ADA inhibitors, we demonstrated the binding of selected phenolic compounds to the active site of ADA using molecular docking and molecular dynamics simulation. Our results show that phenolic compounds (chlorogenic acid, quercetin, and hyperoside) stabilized the ADA complex by forming persistent interactions with the catalytically essential Zn2+ ion. Furthermore, MM-GBSA ligand binding affinity calculations revealed that hyperoside had a comparable binding energy score (ΔG = − 46.56 ± 8.26 kcal/mol) to that of the cocrystal ligand in the ADA crystal structure (PDB ID: 1O5R) (ΔG = − 51.97 ± 4.70 kcal/mol). Similarly, chlorogenic acid exhibited a binding energy score (ΔG = − 18.76 ± 4.60 kcal/mol) comparable to those of the two approved ADA inhibitor drugs pentostatin (ΔG = − 14.54 ± 2.25 kcal/mol) and cladribine (ΔG = − 25.52 ± 4.10 kcal/mol) while quercetin was found to have modest binding affinity (ΔG = − 8.85 ± 7.32 kcal/mol). This study provides insights into the possible inhibitory potential of these phenolic compounds against ADA.








Similar content being viewed by others
Data availability
All data has been incorporated into the main texts.
References
Adamek RN, Ludford P, Duggan SM, Tor Y, Cohen SM (2020) Identification of adenosine deaminase inhibitors by metal-binding pharmacophore screening. ChemMedChem 15(22):2151–2156. https://doi.org/10.1002/cmdc.202000271
Aghaei M, Karami-Tehrani F, Salami S, Atri M (2005) Adenosine deaminase activity in the serum and malignant tumors of breast cancer: the assessment of isoenzyme ADA1 and ADA2 activities. Clin Biochem 38(10):887–891. https://doi.org/10.1016/j.clinbiochem.2005.05.015
Aier I, Varadwaj PK, Raj U (2016) Structural insights into conformational stability of both wild-type and mutant EZH2 receptor. Scient Rep. https://doi.org/10.1038/srep34984
Alunni S, Orrù M, Ottavi L (2008) A study on the inhibition of adenosine deaminase. J Enzyme Inhib Med Chem 23(2):182–189. https://doi.org/10.1080/14756360701475233
Arun KG, Sharanya CS, Sadasivan C (2018) Computational and experimental validation of morin as adenosine deaminase inhibitor. J Recept Signal Transduct 38(3):240–245. https://doi.org/10.1080/10799893.2018.1476543
Ataie G, Safarian S, Divsalar A, Saboury AA, Moosavi-Movahedi AA, Ranjbar B, Cristalli G, Mardanian S (2008) Kinetic and structural analysis of the inhibition of adenosine deaminase by acetaminophen. J Enzyme Inhib Med Chem 19(1):71–78. https://doi.org/10.1080/14756360310001632741
Baell J, Walters MA (2014) Chemistry: Chemical con artists foil drug discovery. Nature 513(7519):481–483. https://doi.org/10.1038/513481a
Bailey AG, Lowe CP (2009) MILCH SHAKE: an efficient method for constraint dynamics applied to alkanes. J Comput Chem 30(15):2485–2493. https://doi.org/10.1002/jcc.21237
Bhaumik D, Medin J, Gathy K, Coleman MS (1993) Mutational analysis of active site residues of human adenosine deaminase. J Biol Chem 268(8):5464–5470. https://doi.org/10.1016/s0021-9258(18)53344-5
Cannon T, Mobarek D, Wegge J, Tabbara IA (2009) Hairy cell leukemia: current concepts. Cancer Invest 26(8):860–865. https://doi.org/10.1080/07357900801965034
Castello F, Costabile G, Bresciani L, Tassotti M, Naviglio D, Luongo D, Ciciola P, Vitale M, Vetrani C, Galaverna G, Brighenti F, Giacco R, Del Rio D, Mena P (2018) Bioavailability and pharmacokinetic profile of grape pomace phenolic compounds in humans. Arch Biochem Biophys 646:1–9. https://doi.org/10.1016/j.abb.2018.03.021
Cristalli G, Costanzi S, Lambertucci C, Lupidi G, Vittori S, Volpini R, Camaioni E (2001) Adenosine deaminase: functional implications and different classes of inhibitors. Med Res Rev 21(2):105–128. https://doi.org/10.1002/1098-1128(200103)21:2%3c105::Aid-med1002%3e3.0.Co;2-u
Cubukcu HC (2018) Different effects of olive leaf on purine metabolizing enzymes of human gastric tissues in vitro. Cancer Therapy Int J. https://doi.org/10.19080/ctoij.2018.12.555826
Cunningham KG, Manson W, Spring FS, Hutchinson SA (1950) Cordycepin, a metabolic product isolated from cultures of cordyceps militaris (linn) link. Nature 166(4231):949–949. https://doi.org/10.1038/166949a0
Gakis C (1996) Adenosine deaminase (ADA) isoenzymes ADA1 and ADA2: diagnostic and biological role. Eur Respir J 9(4):632–633. https://doi.org/10.1183/09031936.96.09040632
Ghosh A, Rapp CS, Friesner RA (1998) Generalized born model based on a surface integral formulation. J Phys Chem B 102(52):10983–10990. https://doi.org/10.1021/jp982533o
Giovannoni G (2017) Cladribine to treat relapsing forms of multiple sclerosis. Neurotherapeutics 14(4):874–887. https://doi.org/10.1007/s13311-017-0573-4
Gleeson MP, Burton NA, Hillier IH (2003) The mechanism of adenosine deaminase catalysis studied by QM/MM calculations: The role of histidine 238 and the activity of the alanine 238 mutant. Phys Chem Chem Phys. https://doi.org/10.1039/b307416a
Harder E, Damm W, Maple J, Wu C, Reboul M, Xiang JY, Wang L, Lupyan D, Dahlgren MK, Knight JL, Kaus JW, Cerutti DS, Krilov G, Jorgensen WL, Abel R, Friesner RA (2016) OPLS3: a force field providing broad coverage of drug-like small molecules and proteins. J Chem Theory Comput 12(1):281–296. https://doi.org/10.1021/acs.jctc.5b00864
Ikeguchi M (2004) Partial rigid-body dynamics in NPT, NPAT and NP gamma T ensembles for proteins and membranes. J Comput Chem 25(4):529–541. https://doi.org/10.1002/jcc.10402
Jain P, Pemmaraju N, Ravandi F (2014) Update on the biology and treatment options for hairy cell leukemia. Curr Treat Options Oncol 15(2):187–209. https://doi.org/10.1007/s11864-014-0285-5
Jorgensen WL, Maxwell DS, TiradoRives J (1996) Development and testing of the OPLS all-atom force field on conformational energetics and properties of organic liquids. J Am Chem Soc 118(45):11225–11236. https://doi.org/10.1021/ja9621760
Kevin J Bowers EC, Huafeng Xu, Ron O. Dror, Michael P. Eastwood,, Brent A. Gregersen JLK, Istvan Kolossvary, Mark A. Moraes, Federico D. Sacerdoti,, John K. Salmon YS, David E. Shaw (2006) Scalable Algorithms for Molecular Dynamics Simulations on Commodity Clusters
Koch HP, Aichinger A, Bohne B, Plank G (1994) In vitro inhibition of adenosine deaminase by a group of steroid and Triterpenoid compounds. Phytother Res 8(2):109–111. https://doi.org/10.1002/ptr.2650080213
Kutryb-Zajac B, Mateuszuk L, Zukowska P, Jasztal A, Zabielska MA, Toczek M, Jablonska P, Zakrzewska A, Sitek B, Rogowski J, Lango R, Slominska EM, Chlopicki S, Smolenski RT (2016) Increased activity of vascular adenosine deaminase in atherosclerosis and therapeutic potential of its inhibition. Cardiovasc Res 112(2):590–605. https://doi.org/10.1093/cvr/cvw203
Kutryb-Zajac B, Koszalka P, Mierzejewska P, Bulinska A, Zabielska MA, Brodzik K, Skrzypkowska A, Zelazek L, Pelikant-Malecka I, Slominska EM, Smolenski RT (2018) Adenosine deaminase inhibition suppresses progression of 4T1 murine breast cancer by adenosine receptor-dependent mechanisms. J Cell Mol Med 22(12):5939–5954. https://doi.org/10.1111/jcmm.13864
Kutryb-Zajac B, Mierzejewska P, Slominska EM, Smolenski RT (2020) Therapeutic perspectives of adenosine deaminase inhibition in cardiovascular diseases. Molecules. https://doi.org/10.3390/molecules25204652
La Motta C, Sartini S, Mugnaini L, Salerno S, Simorini F, Taliani S, Marini AM, Da Settimo F, Lavecchia A, Novellino E, Antonioli L, Fornai M, Blandizzi C, Del Tacca M (2009) Exploiting the pyrazolo[3,4-d]pyrimidin-4-one ring system as a useful template to obtain potent adenosine deaminase inhibitors. J Med Chem 52(6):1681–1692. https://doi.org/10.1021/jm801427r
Leist TP, Weissert R (2011) Cladribine. Clin Neuropharmacol 34(1):28–35. https://doi.org/10.1097/WNF.0b013e318204cd90
Li J, Abel R, Zhu K, Cao Y, Zhao S, Friesner RA (2011) The VSGB 20 model: a next generation energy model for high resolution protein structure modeling. Proteins 79(10):2794–2812. https://doi.org/10.1002/prot.23106
Mark P, Nilsson L (2001) Structure and dynamics of the TIP3P, SPC, and SPC/E water models at 298 K. J Phys Chem A 105(43):9954–9960. https://doi.org/10.1021/jp003020w
Marrone TJ, Straatsma TP, Briggs JM, Wilson DK, Quiocho FA, McCammon JA (1996) Theoretical study of inhibition of adenosine deaminase by (8R)-coformycin and (8R)-deoxycoformycin. J Med Chem 39(1):277–284. https://doi.org/10.1021/jm9505674
Martínez-Rosell G, Giorgino T, De Fabritiis G (2017) PlayMolecule proteinprepare: a web application for protein preparation for molecular dynamics simulations. J Chem Inf Model 57(7):1511–1516. https://doi.org/10.1021/acs.jcim.7b00190
Miteva MA, Guyon F, Tuffery P (2010) Frog2: Efficient 3D conformation ensemble generator for small compounds. Nucleic Acids Res 38:W622–W627. https://doi.org/10.1093/nar/gkq325
Morris GM, Huey R, Lindstrom W, Sanner MF, Belew RK, Goodsell DS, Olson AJ (2009) AutoDock4 and autodocktools4: automated docking with selective receptor flexibility. J Comput Chem 30(16):2785–2791. https://doi.org/10.1002/jcc.21256
Nakajima Y, Kanno T, Nagaya T, Kuribayashi K, Nakano T, Gotoh A, Nishizaki T (2015) Adenosine deaminase inhibitor EHNA exhibits a potent anticancer effect against malignant pleural mesothelioma. Cell Physiol Biochem 35(1):51–60. https://doi.org/10.1159/000369674
Shan YB, Klepeis JL, Eastwood MP, Dror RO, Shaw DE (2005) Gaussian split ewald: a fast ewald mesh method for molecular simulation. J Chem Phys. https://doi.org/10.1063/1.1839571
Singh W, Karabencheva-Christova TG, Black GW, Ainsley J, Dover L, Christov CZ (2016) Conformational dynamics ligand binding and effects of mutations in NirE an s-adenosyl-l-methionine dependent methyltransferase. Scient Rep. https://doi.org/10.1038/srep20107
Stuart SJ, Zhou RH, Berne BJ (1996) Molecular dynamics with multiple time scales: the selection of efficient reference system propagators. J Chem Phys 105(4):1426–1436. https://doi.org/10.1063/1.472005
Talhaoui N, Taamalli A, Gómez-Caravaca AM, Fernández-Gutiérrez A, Segura-Carretero A (2015) Phenolic compounds in olive leaves: analytical determination, biotic and abiotic influence, and health benefits. Food Res Int 77:92–108. https://doi.org/10.1016/j.foodres.2015.09.011
Trincavelli ML (2013) Unveiling the binding mode of adenosine deaminase inhibitors to the active site of the enzyme: implication for rational drug design : presented by Maria P. Abbracchio Purinergic Signal 9(1):1–3. https://doi.org/10.1007/s11302-013-9353-8
Uba AI, Wu C (2022) Identification of potential antagonists of CRF1R for possible treatment of stress and anxiety neuro-disorders using structure-based virtual screening and molecular dynamics simulation. Comput Biol Chem. https://doi.org/10.1016/j.compbiolchem.2022.107743
Uba AI, Yelekçi K (2019) Crystallographic structure versus homology model: a case study of molecular dynamics simulation of human and zebrafish histone deacetylase 10. J Biomol Struct Dyn 38(15):4397–4406. https://doi.org/10.1080/07391102.2019.1691658
Uba AI, Radicella C, Readmond C, Scorese N, Liao S, Liu H, Wu C (2020) Binding of agonist WAY-267,464 and antagonist WAY-methylated to oxytocin receptor probed by all-atom molecular dynamics simulations. Life Sci. https://doi.org/10.1016/j.lfs.2020.117643
Uba AI, Scorese N, Dean E, Liu H, Wu C (2021) Activation mechanism of corticotrophin releasing factor receptor type 1 elucidated using molecular dynamics simulations. ACS Chem Neurosci 12(9):1674–1687. https://doi.org/10.1021/acschemneuro.1c00126
Wilson DK, Rudolph FB, Quiocho FA (1991) Atomic structure of adenosine deaminase complexed with a transition-state analog: understanding catalysis and immunodeficiency mutations. Science 252(5010):1278–1284. https://doi.org/10.1126/science.1925539
Yu C, Zhuang L, Xu F, Zhao L-h, Wang X-h, Wang C-h, Ning L-y, Zhang X-l, Zhang D-m, Wang X-q, Su J-b (2022) Increased levels of serum adenosine deaminase and increased risk of diabetic peripheral neuropathy in type 2 diabetes. Front Endocrinol. https://doi.org/10.3389/fendo.2022.997672
Author information
Authors and Affiliations
Contributions
Conceptualization, AIU and GZ; methodology, AIU, NJP, CW; validation, AIU, CW; Validation and visualization, AIU, CW, investigation, AIU, NJP, CW, GZ; Funding; CW, writing—original draft preparation, AIU, NJP; writing—review and editing, CW, G.Z All authors have read and agreed to the published version of the manuscript.
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare no competing interests.
Additional information
Handling editor: F. Polticelli.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Uba, A.I., Paradis, N.J., Wu, C. et al. Phenolic compounds as potential adenosine deaminase inhibitors: molecular docking and dynamics simulation coupled with MM-GBSA calculations. Amino Acids 55, 1729–1743 (2023). https://doi.org/10.1007/s00726-023-03310-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00726-023-03310-4