Abstract
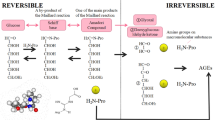
Advanced glycation end products (AGEs) are a group of proteins and lipids becoming glycated and oxidized after persistent contact with reducing sugars or short-chain aldehydes with amino group and/or high degree of oxidative stress. The accumulation of AGEs in the body is a natural process that occurs with senescence, when the turnover rate of proteins is reduced. However, increased circulating AGEs have been described to arise at early lifetime and are associated with adverse outcome and survival, in particular in settings of cardiovascular diseases. AGEs contribute to the development of cardiac dysfunction by two major mechanisms: cross-linking of proteins or binding to their cell surface receptor. Recently, growing evidence shows that high-molecular weight AGEs (HMW-AGEs) might be as important as the characterized low-molecular weight AGEs (LMW-AGEs). Here, we point out the targets of AGEs in the heart and the mechanisms that lead to heart failure with focus on the difference between LMW-AGEs and the less characterized HMW-AGEs. As such, this review is a compilation of relevant papers in the form of a useful resource tool for researchers who want to further investigate the role of HMW-AGEs on cardiac disorders and need a solid base to start on this specific topic.
Similar content being viewed by others
References
Acharya AS, Manning JM (1983) Reaction of glycolaldehyde with proteins: latent crosslinking potential of alpha-hydroxyaldehydes. Proc Natl Acad Sci USA 80(12):3590–3594
Ahmed N (2005) Advanced glycation endproducts—role in pathology of diabetic complications. Diabetes Res Clin Pract 67(1):3–21
Ahmed N, Thornalley PJ (2007) Advanced glycation endproducts: what is their relevance to diabetic complications? Diabetes Obes Metab 9(3):233–245
Ahmed N, Babaei-Jadidi R et al (2005) Degradation products of proteins damaged by glycation, oxidation and nitration in clinical type 1 diabetes. Diabetologia 48(8):1590–1603
Aleshin A, Ananthakrishnan R et al (2008) RAGE modulates myocardial injury consequent to LAD infarction via impact on JNK and STAT signaling in a murine model. Am J Physiol Heart Circ Physiol 294(4):H1823–H1832
Aronson D (2003) Cross-linking of glycated collagen in the pathogenesis of arterial and myocardial stiffening of aging and diabetes. J Hypertens 21(1):3–12
Berg TJ, Snorgaard O et al (1999) Serum levels of advanced glycation end products are associated with left ventricular diastolic function in patients with type 1 diabetes. Diabetes Care 22(7):1186–1190
Bidasee KR, Nallani K et al (2003a) Chronic diabetes increases advances glycation end products on cardiac ryanodine receptors calcium-release channels (primers). Diabetes 52:1825–1836
Bidasee KR, Xu L et al (2003b) Diketopyridylryanodine has three concentration-dependent effects on the cardiac calcium-release channel/ryanodine receptor. J Biol Chem 278(16):14237–14248
Bidasee KR, Zhang Y et al (2004) Diabetes increases formation of advanced glycation end products on Sarco(endo)plasmic reticulum Ca2+-ATPase. Diabetes 53(2):463–473
Brownlee M (1995) Advanced protein glycosylation in diabetes and aging. Annu Rev Med 46:223–234
Bucala R, Tracey KJ et al (1991) Advanced glycosylation products quench nitric oxide and mediate defective endothelium-dependent vasodilatation in experimental diabetes. J Clin Investig 87(2):432–438
Bucciarelli LG, Kaneko M et al (2006) Receptor for advanced-glycation end products: key modulator of myocardial ischemic injury. Circulation 113(9):1226–1234
Bucciarelli LG, Ananthakrishnan R et al (2008) RAGE and modulation of ischemic injury in the diabetic myocardium. Diabetes 57(7):1941–1951
Campbell DJ, Somaratne JB et al (2012) Diastolic dysfunction of aging is independent of myocardial structure but associated with plasma advanced glycation end-product levels. PLoS One 7(11):e49813
Corman B, Duriez M et al (1998) Aminoguanidine prevents age-related arterial stiffening and cardiac hypertrophy. Proc Natl Acad Sci USA 95(3):1301–1306
Deluyker D, Ferferieva V et al (2016) Cross-linking versus RAGE: how do high molecular weight advanced glycation products induce cardiac dysfunction? Int J Cardiol 210:100–108
Donaldson C, Taatjes DJ et al (2010) Combined immunoelectron microscopic and computer-assisted image analyses to detect advanced glycation end-products in human myocardium. Histochem Cell Biol 134(1):23–30
Esposito C, Gerlach H et al (1989) Endothelial receptor-mediated binding of glucose-modified albumin is associated with increased monolayer permeability and modulation of cell surface coagulant properties. J Exp Med 170(4):1387–1407
Finot PA, Magnenat E (1981) Metabolic transit of early and advanced Maillard products. Prog Food Nutr Sci 5(1–6):193–207
Foerster A, KüHNE Y, Henle T (2005) Studies on absorption and elimination of dietary maillard reaction products. Ann N Y Acad Sci 1043:474–481
Furth AJ (1997) Glycated proteins in diabetes. Br J Biomed Sci 54(3):192–200
Gerdemann A, Lemke HD et al (2000) Low-molecular but not high-molecular advanced glycation end products (AGEs) are removed by high-flux dialysis. Clin Nephrol 54(4):276–283
Glomb MA, Monnier VM (1995) Mechanism of protein modification by glyoxal and glycolaldehyde, reactive intermediates of the Maillard reaction. J Biol Chem 270(17):10017–10026
Goldberg T, Cai W et al (2004) Advanced glycoxidation end products in commonly consumed foods. J Am Diet Assoc 104(8):1287–1291
Goldin A, Beckman JA et al (2006) Advanced glycation end products: sparking the development of diabetic vascular injury. Circulation 114(6):597–605
Grzebyk E, Knapik-Kordecka M et al (2013) Advanced glycation end-products and cathepsin cysteine protease in type 2 diabetic patients. Pol Arch Med Wewn 123(7–8):364–370
Hartog JW, Voors AA et al (2007) Advanced glycation end-products (AGEs) and heart failure: pathophysiology and clinical implications. Eur J Heart Fail 9(12):1146–1155
He C, Sabol J et al (1999) Dietary glycotoxins: inhibition of reactive products by aminoguanidine facilitates renal clearance and reduces tissue sequestration. Diabetes 48(6):1308–1315
Hegab Z, Gibbons S et al (2012) Role of advanced glycation end products in cardiovascular disease. World J Cardiol 4(4):90–102
Heidland A, Sebekova K et al (2004) Paradox of circulating advanced glycation end product concentrations in patients with congestive heart failure and after heart transplantation. Heart 90(11):1269–1274
Hellwig M, Geissler S et al (2011) Transport of free and peptide-bound glycated amino acids: synthesis, transepithelial flux at Caco-2 cell monolayers, and interaction with apical membrane transport proteins. ChemBioChem 12(8):1270–1279
Henle T (2003) AGEs in foods: do they play a role in uremia? Kidney Int 63:S145–S147
Jara N, Leal MJ et al (2012) Dietary intake increases serum levels of carboxymethil-lysine (CML) in diabetic patients. Nutr Hosp 27(4):1272–1278
Kilhovd BK, Berg TJ et al (1999) Serum levels of advanced glycation end products are increased in patients with type 2 diabetes and coronary heart disease. Diabetes Care 22(9):1543–1548
Kisugi R, Kouzuma T et al (2007) Structural and glycation site changes of albumin in diabetic patient with very high glycated albumin. Clin Chim Acta 382(1–2):59–64
Koyama Y, Takeishi Y et al (2007) High serum level of pentosidine, an advanced glycation end product (AGE), is a risk factor of patients with heart failure. J Card Fail 13(3):199–206
Kranstuber AL, Del Rio C et al (2012) Advanced glycation end product cross-link breaker attenuates diabetes-induced cardiac dysfunction by improving sarcoplasmic reticulum calcium handling. Front Physiol 3:292
Li SY, Sigmon VK et al (2007) Advanced glycation endproduct induces ROS accumulation, apoptosis, MAP kinase activation and nuclear O-GlcNAcylation in human cardiac myocytes. Life Sci 80(11):1051–1056
Little WC, Zile MR et al (2005) The effect of alagebrium chloride (ALT-711), a novel glucose cross-link breaker, in the treatment of elderly patients with diastolic heart failure. J Card Fail 11(3):191–195
Lorenzi M (2007) The polyol pathway as a mechanism for diabetic retinopathy: attractive, elusive, and resilient. Exp Diabetes Res 2007:61038
Luevano-Contreras C, Chapman-Novakofski K (2010) Dietary advanced glycation end products and aging. Nutrients 2(12):1247–1265
Makita Z, Bucala R et al (1994) Reactive glycosylation endproducts in diabetic uraemia and treatment of renal failure. Lancet 343(8912):1519–1522
McNair ED, Wells CR et al (2009) Low levels of soluble receptor for advanced glycation end products in non-ST elevation myocardial infarction patients. Int J Angiol 18(4):187–192
Miura J, Yamagishi S et al (2003) Serum levels of non-carboxymethyllysine advanced glycation endproducts are correlated to severity of microvascular complications in patients with Type 1 diabetes. J Diabetes Complicat 17(1):16–21
Moheimani F, Morgan PE et al (2010) (2010) Deleterious effects of reactive aldehydes and glycated proteins on macrophage proteasomal function: possible links between diabetes and atherosclerosis. Biochim Biophys Acta 1802(6):561–571
Monnier VM, Sun W et al (2014) Glucosepane: a poorly understood advanced glycation end product of growing importance for diabetes and its complications. Clin Chem Lab Med 52(1):21–32
Nagai R, Matsumoto K et al (2000) Glycolaldehyde, a reactive intermediate for advanced glycation end products, plays an important role in the generation of an active ligand for the macrophage scavenger receptor. Diabetes 49(10):1714–1723
Nielsen JM, Kristiansen SB et al (2009) Blockage of receptor for advanced glycation end products prevents development of cardiac dysfunction in db/db type 2 diabetic mice. Eur J Heart Fail 11(7):638–647
Ott C, Jacobs K et al (2014) Role of advanced glycation end products in cellular signaling. Redox Biol 2:411–429
Penfold SA, Coughlan MT et al (2010) Circulating high-molecular-weight RAGE ligands activate pathways implicated in the development of diabetic nephropathy. Kidney Int 78(3):287–295
Petrova R, Yamamoto Y et al (2002) Advanced glycation endproduct-induced calcium handling impairment in mouse cardiac myocytes. J Mol Cell Cardiol 34(10):1425–1431
Peyroux J, Sternberg M (2006) Advanced glycation endproducts (AGEs): pharmacological inhibition in diabetes. Pathol Biol (Paris) 54(7):405–419
Poulsen MW, Hedegaard RV et al (2013) Advanced glycation endproducts in food and their effects on health. Food Chem Toxicol 60:10–37
Poulsen MW, Andersen JM et al (2016) Short-term effects of dietary advanced glycation end products in rats. Br J Nutr 115(4):629–636
Ramasamy R, Schmidt AM (2012) Receptor for advanced glycation end products (RAGE) and implications for the pathophysiology of heart failure. Curr Heart Fail Rep 9(2):107–116
Rojas A, Mercadal E et al (2008) Advanced Glycation and ROS: a link between diabetes and heart failure. Curr Vasc Pharmacol 6(1):44–51
Schalkwijk CG, Stehouwer CD et al (2004) Fructose-mediated non-enzymatic glycation: sweet coupling or bad modification. Diabetes Metab Res Rev 20(5):369–382
Seiquer I, Diaz-Alguacil J et al (2006) Diets rich in Maillard reaction products affect protein digestibility in adolescent males aged 11-14 y. Am J Clin Nutr 83(5):1082–1088
Sharp PS, Rainbow S et al (2003) Serum levels of low molecular weight advanced glycation end products in diabetic subjects. Diabet Med 20(7):575–579
Singh R, Barden A et al (2001) Advanced glycation end-products: a review. Diabetologia 44(2):129–146
Takeuchi M, Makita Z et al (1999) Detection of noncarboxymethyllysine and carboxymethyllysine advanced glycation end products (AGE) in serum of diabetic patients. Mol Med 5(6):393–405
Takeuchi M, Takino J et al (2015) Assessment of the concentrations of various advanced glycation end-products in beverages and foods that are commonly consumed in Japan. PLoS One 10(3):e0118652
Tang Y, Chen A (2014) Curcumin eliminates the effect of advanced glycation end-products (AGEs) on the divergent regulation of gene expression of receptors of AGEs by interrupting leptin signaling. Lab Investig 94(5):503–516
Thomas MC, Tsalamandris C et al (2004) Low-molecular-weight AGEs are associated with GFR and anemia in patients with type 2 diabetes. Kidney Int 66(3):1167–1172
Thomas MC, Forbes JM et al (2005) Low-molecular weight advanced glycation end products: markers of tissue AGE accumulation and more? Ann N Y Acad Sci 1043:644–654
Thornalley PJ (1999) Clinical significance of glycation. Clin Lab 45:263–273
Thornalley PJ, Langborg A et al (1999) Formation of glyoxal, methylglyoxal and 3-deoxyglucosone in the glycation of proteins by glucose. Biochem J 344(Pt 1):109–116
Uribarri J, Woodruff S et al (2010) Advanced glycation end products in foods and a practical guide to their reduction in the diet. J Am Diet Assoc 110(6):911–916 (e912)
Vazzana N, Santilli F et al (2009) Soluble forms of RAGE in internal medicine. Intern Emerg Med 4(5):389–401
Vlassara H, Striker GE (2011) AGE restriction in diabetes mellitus: a paradigm shift. Nat Rev Endocrinol 7(9):526–539
Vlassara H, Striker GE (2013) Advanced glycation endproducts in diabetes and diabetic complications. Endocrinol Metab Clin N Am 42(4):697–719
Wang Z, Jiang Y et al (2012) Advanced glycation end-product Nepsilon-carboxymethyl-Lysine accelerates progression of atherosclerotic calcification in diabetes. Atherosclerosis 221(2):387–396
Wautier JL, Zoukourian C et al (1996) Receptor-mediated endothelial cell dysfunction in diabetic vasculopathy. Soluble receptor for advanced glycation end products blocks hyperpermeability in diabetic rats. J Clin Investig 97(1):238–243
Willemsen S, Hartog JW et al (2012a) Advanced glycation end-products, a pathophysiological pathway in the cardiorenal syndrome. Heart Fail Rev 17(2):221–228
Willemsen S, Hartog JW et al (2012b) The role of advanced glycation end-products and their receptor on outcome in heart failure patients with preserved and reduced ejection fraction. Am Heart J 164(5):742–749 (e743)
Yan SF, Ramasamy R et al (2009) The receptor for advanced glycation endproducts (RAGE) and cardiovascular disease. Expert Rev Mol Med 11:e9
Zieman SJ (2004) Advanced glycation end products cross-linking pathophysiologic role and therapeutic target in cardiovascular disease. Drugs 64(5):459–470
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Research involving human participants and/or animals
This article does not contain any studies with human participants or animals performed by any of the authors.
Informed consent
None.
Additional information
Handling Editor: J. D. Wade.
Rights and permissions
About this article
Cite this article
Deluyker, D., Evens, L. & Bito, V. Advanced glycation end products (AGEs) and cardiovascular dysfunction: focus on high molecular weight AGEs. Amino Acids 49, 1535–1541 (2017). https://doi.org/10.1007/s00726-017-2464-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00726-017-2464-8