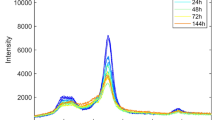
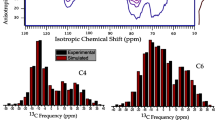
Abstract
The role of cellulose in various applications is remarkably going up in recent decades as it is a renewable material for wide areas of applications that can be delivered in huge quantities. This drives permanently growing interest to development of sensors for processing industry, that can give fast and reliable information about physicochemical structure of cellulosic materials. In this framework, Time-Domain NMR with its capability to be miniaturized may become very attractive to measure such structural parameters as crystallinity, domain sizes, and sorption properties. This contribution is aimed to summarize existing NMR relaxometric methods and update them with the techniques of signal processing and pulse sequences like Magic Sandwich Echo or multiple quantum transitions, that are well established for synthetic polymers, but still have not been extensively used for polysaccharides. Here, we also demonstrate our own results of cellulose study trying to contribute to the promotion of Time-Domain NMR applicability to biopolymers.
















Similar content being viewed by others
Data Availability
The datasets generated during and/or analyzed during the current study are available from the corresponding author on reasonable request.
References
Yu.B. Grunin, LYu. Grunin, VYu. Schiraya, M.S. Ivanova, D.S. Masas, Bioresour. Bioprocess. (2020). https://doi.org/10.1186/s40643-020-00332-8
D.J. Cosgrove, Nat. Rev. Mol. Cell Biol. (2005). https://doi.org/10.1038/nrm1746
S. Rongpipi, D. Ye, E.D. Gomez, E.W. Gomez, Front. Plant Sci. (2019). https://doi.org/10.3389/fpls.2018.01894
B.M. Jordan, J. Dumais, Biomechanics of plant cell growth, in encyclopedia of life sciences (ELS) (John Wiley & Sons Ltd, Toronto, 2010)
Yu.B. Grunin, LYu. Grunin, V.I. Talantcev, E.A. Nikolskaya, D.S. Masas, Biophysics (2015). https://doi.org/10.1134/S0006350915010133
R.A. Horn, Morphology of wood pulp fiber from softwoods and influence on paper strength (U.S. Department of Agriculture, Forest Service, 1974)
L. Viikari, A. Suurnäkki, S. Grönqvist, L. Raaska, A. Ragauskas, Forest products: biotechnology in pulp and paper processing, in Encyclopedia of microbiology (Academic Press, Cambridge, 2009)
E. Małachowska, M. Dubowik, A. Lipkiewicz, K. Przybysz, P. Przybysz, Sustainability (2020). https://doi.org/10.3390/su12177219
S.B. Stanković, D. Popović, G.B. Poparić, Polym. Test. (2008). https://doi.org/10.1016/j.polymertesting.2007.08.003
R. Mu, X. Hong, Y. Ni, Y. Li, J. Pang, Q. Wang, J. Xiao, Y. Zheng, Trends Food Sci. Technol. (2019). https://doi.org/10.1016/j.tifs.2019.09.013
Z. Shi, Y. Zhang, G.O. Phillips, G. Yang, Food Hydrocolloids (2014). https://doi.org/10.1016/j.foodhyd.2013.07.012
H.M.C. Azeredo, H. Barud, C.S. Farinas, V.M. Vasconcellos, A.M. Claro, Front. Sustain. Food Syst. (2019). https://doi.org/10.3389/fsufs.2019.00007
Y.P. Chauhan, R.S. Sapkal, V.S. Sapkal G.S. Zamre. Int. J. Chem. Sci. 7 (2009).
A. Fiorati, G. Grassi, A. Graziano, G. Liberatori, N. Pastori, L. Melone, L. Bonciani, L. Pontorno, C. Punta, I. Corsi, J. Cleaner Prod. (2020). https://doi.org/10.1016/j.jclepro.2019.119009
G.M. DeLoid, X. Cao, R.M. Molina, D.I. Silva, K. Bhattacharya, K.W. Ng, S.C.J. Loo, J.D. Brain, P. Demokritou, Nano. Environ. Sci. (2019). https://doi.org/10.1039/C9EN00184K
S. Das, J. Chakraborty, S. Chatterjee, H. Kumar, Nano. Environ. Sci (2018). https://doi.org/10.1039/C8EN00799C
S.H. Hassan, L.H. Voon, T.S. Velayutham, L. Zhai, H.C. Kim, J. Kim, J. Renewable Mater. (2018). https://doi.org/10.7569/JRM.2017.634173
A. Wolfberger, A. Petritz, A. Fian, J. Herka, V. Schmidt, B. Stadlober, R. Kargl, S. Spirk, T. Griesser, Cellulose (2015). https://doi.org/10.1007/s10570-014-0471-4
E. Kontturi, S. Spirk, Front. Chem. (2019). https://doi.org/10.3389/fchem.2019.00488
H. Seddiqi, E. Oliaei, H. Honarkar, J. Jin, L.C. Geonzon, R.G. Bacabac, J. Klein-Nulend, Cellulose (2021). https://doi.org/10.1007/s10570-020-03674-w
A.E. Zavadskii, Fibre Chem. (2007). https://doi.org/10.1007/s10692-007-0106-8
L. Segal, J.J. Creely, A.E. Martin, C.M. Conrad, Text. Res. J. (1959). https://doi.org/10.1177/004051755902901003
P. Penttilä, Structural characterization of cellulosic materials using X-ray and neutron scattering dissertation (University of Helsinki, Helsinki, 2013)
N. Abidi, M. Herath, Text. Res. J. (2017). https://doi.org/10.1177/0040517516688634
W.R. Kunusa, I. Isa, L.A.R. Laliyo, H. Iyabu, J. Phys. Conf. Ser. (2018). https://doi.org/10.1088/1742-6596/1028/1/012199
P. Krishnamachari, R. Hashaikeh, M. Tiner, Micron (2011). https://doi.org/10.1016/j.micron.2011.05.001
D. Capitani, F. Porro, A.L. Segre, Carbohydr. Polym. (2000). https://doi.org/10.1016/S0144-8617(99)00173-3
W. Zhao, A. Kirui, F. Deligey, F. Mentink-Vigier, Y. Zhou, B. Zhang, T. Wang, Biotechnol. Biofuels. (2021). https://doi.org/10.1186/s13068-020-01858-x
N.M. Khasanova, B.V. Sakharov, V.Y. Volkov, D.K. Nourgaliev, Low-field NMR method for analysis of heavy oils without extraction of asphaltenes. Conference Proceedings of 17th International Multidisciplinary Scientific GeoConference SGEM 2017, Vienna, Austria, 27–29 November 2017, 17, pp. 297–304.
T. Koso, D. Rico del Cerro, S. Heikkinen, T. Nypelö, J. Buffiere, J.E. Perea-Buceta, A. Potthast, T. Rosenau, H. Heikkinen, H. Maaheimo, A. Isogai, I. Kilpeläinen, A.W.T. King, Cellulose (2020). https://doi.org/10.1007/s10570-020-03317-0
L.Y. Grunin, Y.B. Grunin, E.A. Nikolskaya, N.N. Sheveleva, I.A. Nikolaev, Biophysics (2017). https://doi.org/10.1134/S0006350917020087
Y.-Q. Song, J. Magn. Reson. (2013). https://doi.org/10.1016/j.jmr.2012.11.010
R.F. Nogueira, M.I.B. Tavares, R.A.S. San Gil, A.G. Ferreira, Mater. Appl. Sci. (2011). https://doi.org/10.4236/msa.2011.25060
S.C. Chinn, A. Cook-Tendulkar, R. Maxwell, H. Wheeler, M. Wilson, Z. Harry Xie, Polym. Test (2007). https://doi.org/10.1016/j.polymertesting.2007.07.005
J. P. Patel, Shaw Ling Hsu. 2018. J. Polym. Sci. Part. Polym. Phys. https://doi.org/10.1002/polb.24583.
Y. Nishiyama, J. Wood Sci. (2009). https://doi.org/10.1007/s10086-009-1029-1
TD-NMR Analyzer Spin Track. http://www.nmr-design.com/products/nmr-analyzer-spin-track. Accessed 15 April 2023.
A. Maus, C. Hertlein, K. Saalwächter, Macromol. Chem. Phys. (2006). https://doi.org/10.1002/macp.200600169
R.R. Ernst, G. Bodenhausen, A. Wokaun, Principles of nuclear magnetic resonance in one and two dimensions (Clarendon Press, Oxford, 1987), p.610
J. Baum, A. Pines, J. Am. Chem. Soc. (1986). https://doi.org/10.1021/ja00284a001
K. Saalwächter, Prog. Nucl. Magn. Reson. Spectrosc. (2007). https://doi.org/10.1016/j.pnmrs.2007.01.001
M. Schneider, L. Gasper, D.E. Demco, B. Blümich, J. Chem. Phys. (1999). https://doi.org/10.1063/1.479291
M.P. Nicholas, E. Eryilmaz, F. Ferrage, D. Cowburn, R. Ghose, Prog. Nucl. Magn. Reson. Spectrosc. (2010). https://doi.org/10.1016/j.pnmrs.2010.04.003
A. Abragam, The principles of nuclear magnetism (Clarendon Press, Oxford, 1961), p.599
E.W. Hansen, P.E. Kristiansen, B. Pedersen, J. Phys. Chem. B. (1998). https://doi.org/10.1021/jp981753z
W. Derbyshire, M. van den Bosch, D. van Dusschoten, W. MacNaughtan, I.A. Farhat, M.A. Hemminga, J.R. Mitchell, J. Magn. Reson. (2004). https://doi.org/10.1016/j.jmr.2004.03.013
C. Hedesiu, D.E. Demco, R. Kleppinger, A.A. Buda, B. Blümich, K. Remerie, V.M. Litvinov, Polymer (2007). https://doi.org/10.1016/j.polymer.2006.12.019
T.M. Todoruk, I.D. Hartley, R. Teymoori, J. Liang, H. Peemoeller, Materials (2011). https://doi.org/10.3390/ma4010131
A.G. Redfield, J. IBM, J. Res. Dev. (1957). https://doi.org/10.1147/rd.11.0019
A.G. Redfield, Adv. Magn. Reson. 1, 32 (1966)
N. Bloembergen, E.M. Purcell, R.V. Pound, Phys. Rev. 73, 679 (1948)
V.I. Chizhik, Yu.S. Chernyshev, A.V. Donets, V.V. Frolov, A.V. Komolkin, M.G. Shelyapina, Magnetic resonance and its applications (Springer, Switzerland, 2014), p.788
J.H. Van Vleck, Phys. Rev. (1948). https://doi.org/10.1103/PhysRev.74.1168
E. Anoardo, G. Galli, G. Ferrante, Appl. Magn. Reson. (2001). https://doi.org/10.1007/BF03162287
M. Goldman, J. Magn. Reson. (2001). https://doi.org/10.1006/jmre.2000.2239
H.W. Spiess, Macromolecules (2017). https://doi.org/10.1021/acs.macromol.6b02736
V. Räntzsch, M. Haas, M.B. Özen, K.-F. Ratzsch, K. Riazi, S. Kauffmann-Weiss, J.K. Palacios, A.J. Müller, I. Vittorias, G. Guthausen, M. Wilhelm, Polymer (2018). https://doi.org/10.1016/j.polymer.2018.04.066
K. Saalwächter, in NMR Methods for Characterization of Synthetic and Natural Polymers (1st ed.), ed. by R. Zhang, T. Miyoshi, P. Sun (Royal Society of Chemistry, 2019), pp. 1–22.
D. Besghini, M. Mauri, R. Simonutti, Appl. Sci. (2019). https://doi.org/10.3390/app9091801
K. Schäler. Low-field NMR Studies of Structure and Dynamics in Semicrystalline Polymers. Dissertation Martin-Luther-Universität Halle-Wittenberg. Halle. 2012.
K. Van Putte, J. Van Den Enden, J. Am. Oil Chem. Soc. (1974). https://doi.org/10.1007/BF02633005
P. Mansfield, Phys. Rev. (1965). https://doi.org/10.1103/PhysRev.137.A961
A. Papon, K. Saalwächter, K. Schäler, L. Guy, F. Lequeux, H. Montes, Macromolecules (2011). https://doi.org/10.1021/ma102486x
M. Pieruccini, S. Sturniolo, M. Corti, A. Rigamonti, The. Eur. Phys. J. B. (2015). https://doi.org/10.1140/epjb/e2015-60417-6
A. Pines, W.K. Rhim, J.S. Waugh, J. Magn. Reson. (1972). https://doi.org/10.1016/0022-2364(72)90154-0
W.K. Rhim, A. Pines, J.S. Waugh, Phys. Rev. B. (1971). https://doi.org/10.1103/PhysRevB.3.684
K. Takegoshi, C.A. McDowell, Chem. Phys. Lett. (1985). https://doi.org/10.1016/0009-2614(85)80134-2
S. Matsui, J. Magn. Reson. (1992). https://doi.org/10.1016/0022-2364(92)90015-Y
R.H.S. Garcia, J.G. Filgueiras, E.R. deAzevedo, L.A. Colnago, Solid State. Nucl. Reson. Magn. (2019). https://doi.org/10.1016/j.ssnmr.2019.101619
W.K. Rhim, H. Kessemeier, Phys. Rev. B. (1971). https://doi.org/10.1103/PhysRevB.3.3655
J. van Duynhoven, I. Dubourg, G.J. Goudappel, E. Roijers, J. Amer. Oil Chem. Soc. (2002). https://doi.org/10.1007/s11746-002-0493-7
MM. Contreras, CR. Nascimento, RP. Cucinelli Neto, MF. Costa, CA. Costa. Data Brief. 2018. https://doi.org/10.1016/j.dib.2018.04.145.
Yu.B. Grunin, M.S. Ivanova, D.S. Masas, LYu. Grunin, Biophysics (2019). https://doi.org/10.1134/S0006350919060071
L. Grunin, M.H. Oztop, S. Guner, S.F. Baltaci, Magn. Reson. Chem. (2019). https://doi.org/10.1002/mrc.4866
ISO 8292: Animal and vegetable fats and oils—determination of solid fat content—pulsed nuclear magnetic resonance method—Part 1: Direct method. 2008. pp 27
E.A. Nikolskaya, L.Yu. Grunin, Yu.B. Grunin, Y. Hiltunen, Analit. Kontrol. Anal. Control. 2013.
M. Tyufekchiev, A. Kolodziejczak, P. Duan, M. Foston, K. Schmidt-Rohr, M.T. Timko, Green Chem. (2019). https://doi.org/10.1039/C9GC02466B
K.M. Kovalov, O.M. Alekseev, M.M. Lazarenko, Yu.F. Zabashta, Yu.E. Grabovskii, SYu. Tkachov, Nanoscale Res. Lett. (2017). https://doi.org/10.1186/s11671-017-2231-5
V.S. Fedotova, M.P. Sokolova, V.K. Vorobiov, E.V. Sivtsov, N.V. Lukasheva, M.A. Smirnov, Polymers (2023). https://doi.org/10.3390/polym15092156
S. Cichosz, A. Masek, Materials (2020). https://doi.org/10.3390/ma13204573
B. Medronho, B. Lindman, Curr. Opin. Colloid Interface Sci. (2014). https://doi.org/10.1016/j.cocis.2013.12.001
V. Ottesen, K. Syverud, Cellulose (2021). https://doi.org/10.1007/s10570-020-03517-8
A. Ottenhall, J. Henschen, J. Illergård, M. Ek, Environ. Sci.: Water Res. Technol. (2018). https://doi.org/10.1039/C8EW00514A
K. Ratajczak, M. Stobiecka, Carbohydr. Polym. (2020). https://doi.org/10.1016/j.carbpol.2019.115463
E. Vittadini, L.C. Dickinson, P. Chinachoti, Carbohydr. Polym. (2001). https://doi.org/10.1016/S0144-8617(00)00282-4
E.L. Lindh, C. Terenzi, L. Salmén, I. Furó, Phys. Chem. Chem. Phys. (2017). https://doi.org/10.1039/C6CP08219J
E.L. Hahn, Phys. Rev. (1950). https://doi.org/10.1103/PhysRev.80.580
S. Meiboom, D. Gill, Rev. Sci. Instrum. (1958). https://doi.org/10.1063/1.1716296
A.S. Traore, L. Foucat, J.P. Renou, Biopolymers (2000). https://doi.org/10.1002/(SICI)1097-0282(200005)53:6%3c476::AID-BIP4%3e3.0.CO;2-8
V.V. Rodin, Biophys. Rev. (2020). https://doi.org/10.1007/s12551-020-00694-5
J. Li, E. Ma, Forests (2021). https://doi.org/10.3390/f12070886
K.S.W. Sing, D.H. Everett, R.A.W. Haul, L. Moscou, R.A. Pierotti, J. Rouquérol, T. Siemieniewska, Pure Appl. Chem. (1985). https://doi.org/10.1351/pac198557040603
Yu.B. Grunin, LYu. Grunin, L.S. Gal’braikh, N.N. Sheveleva, D.S. Masas, Fibre Chem. (2018). https://doi.org/10.1007/s10692-018-9890-6
G.H. Sørland, K. Djurhuus, H.C. Widerøe, J.R. Lien, A. Skauge, Diffusion Fundamentals. 5 2007.
J. Arnold, Mobile NMR for rock porosity and permeability dissertation (RWTH Aachen University, Aachen, 2007)
E.D. Ostroff, J.S. Waugh, Phys. Rev. Lett. (1966). https://doi.org/10.1103/PhysRevLett.16.1097
J.S. Waugh, L.M. Huber, J. Chem. Phys. (1967). https://doi.org/10.1063/1.1712185
J.S. Waugh, L.M. Huber, U. Haeberlen, Phys. Rev. Lett. (1968). https://doi.org/10.1103/PhysRevLett.20.180
F. Yamaguchi, T.D. Ladd, C.P. Master, Y. Yamamoto, N. Khaneja. Efficient decoupling and recoupling in solid state NMR for quantum computation. ArXiv: Quantum Phys. 2004.
K. Adamić, Chem. Acta. 40(3), 111 (1968)
M. Foston, A.J. Ragauskas, Energy Fuels (2010). https://doi.org/10.1021/ef100882t
M. Sikora, M. Krystyjan, A. Dobosz, P. Tomasik, K. Walkowiak, Ł Masewicz, P.Ł Kowalczewski, H.M. Baranowska, Polymers (2019). https://doi.org/10.3390/polym11111764
A. Rachocki, J. Tritt-Goc, J. Polym. Res. (2006). https://doi.org/10.1007/s10965-005-9026-6
A.A. Baker, W. Helbert, J. Sugiyama, M.J. Miles, Biophys. J. (2000). https://doi.org/10.1016/S0006-3495(00)76367-3
R.E. Taylor, A.D. French, G.R. Gamble, D.S. Himmelsbach, R.D. Stipanovic, D.P. Thibodeaux, P.J. Wakelyn, C. Dybowski, J. Mol. Struct. (2008). https://doi.org/10.1016/j.molstruc.2007.08.006
V.J. McBrierty, NMR Spectroscopy of Polymers in the Solid State, in comprehensive polymer science and supplements (Elsevier, Londan, 1989)
J. Leisen, H.W. Beckham, M.A. Sharaf, Macromolecules (2004). https://doi.org/10.1021/ma0489974
F. Vaca Chávez, K. Saalwächter, Macromolecules (2011). https://doi.org/10.1021/ma102571u
R. Fechete, D.E. Demco, B. Blümich, J. Magn. Reson. (2004). https://doi.org/10.1016/j.jmr.2004.03.026
A. Wiesmath, NMR methods for strongly inhomogeneous magnetic fields application on elastomers using the NMR-MOUSE dissertation (RWTH Aachen University, Aachen, 2001)
F. Vaca Chávez, K. Saalwächter, Macromolecules (2011). https://doi.org/10.1021/ma1025708
F. Vaca Chávez, K. Saalwächter, Phys. Lett. Rev. (2010). https://doi.org/10.1103/PhysRevLett.104.198305
K. Saalwächter, A. Heuer, Macromolecules (2006). https://doi.org/10.1021/ma052567b
F. Furtado, J. Damron, M.-L. Trutschel, C. Franz, K. Schröter, R.C. Ball, K. Saalwächter, D. Panja, Macromolecules (2014). https://doi.org/10.1021/ma4021938
M. Mauri, Y. Thomann, H. Schneider, K. Saalwächter, Solid State Nucl. Magn. Reson. (2008). https://doi.org/10.1016/j.ssnmr.2008.07.001
Funding
This work was partially funded from the European Union's Horizon 2020 Research and Innovation program—MSCA-RISE under Grant Agreement No 101008228.
Author information
Authors and Affiliations
Contributions
LG: Conceptualization, Methodology, Validation, Investigation, Data curation, Writing—original draft, Writing—review and editing, Visualization, Supervision; MI: Methodology, Validation, Investigation, Data curation, Writing—original draft; VS: Investigation, Writing—original draft; TG: Investigation, Writing—original draft.
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Ethical Approval
Not applicable.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Grunin, L., Ivanova, M., Schiraya, V. et al. Time-Domain NMR Techniques in Cellulose Structure Analysis. Appl Magn Reson 54, 929–955 (2023). https://doi.org/10.1007/s00723-023-01600-4
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00723-023-01600-4