Abstract
Acid decomposition using concentrated H2SO4 at elevated temperature (>200 °C) is a common process to recover rare earth elements (REE) from refractory minerals where the reactions form REE sulfates, which are dissolved during a subsequent water leach. While the decomposition of REE orthophosphates and fluorocarbonates is well-documented, investigations focusing on REE silicates are more limited. The current study focuses on allanite-(Ce), since, in addition to the REEs, the presence of other sulfate-forming cations in the crystalline structure can help better define the decomposition of complex silicates.
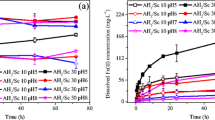
After treatment using concentrated H2SO4 at temperatures between 175 °C and 225 °C, important passivation effects from the formation of amorphous silica layers were observed. Decomposition occurs rather during the water leach if its temperature is maintained at near-boiling conditions for 20 h, suggesting that water helps attenuate passivation. Consequently, experiments were performed where water was premixed with H2SO4 during the initial acid treatment to lower concentration down to 55 wt%. The water addition drastically enhanced decomposition rate at acid treatment temperatures down to 100–125 °C, where REE recoveries >90 wt% were achieved after a room-temperature water leach. The extent of passivation may be linked to the solubility of produced sulfates. With concentrated acid, low sulfate solubility induces local saturation at the crystal dissolution front leading to diffusion-limited cation transfer across the growing silica layer. When the water content is increased, the sulfate-forming cations can enter in solution providing efficient transport, hence minimizing passivation. This decreases both acid treatment and water leach temperatures, significantly reducing energy consumption without additional reagent costs.












Similar content being viewed by others
References
Apostolidis CI, Distin PA (1978) The kinetics of the sulphuric acid leaching of nickel and magnesium from reduction roasted serpentine. Hydrometallurgy 3:181–196
Armbruster T, Bonazzi P, Akasaka M, Bermanec V, Chopin C, Gieré R, Heuss-Assbichler S, Liebscher X, Menchetti S, Pan Y, Pasero M (2006) Recommended nomenclature of epidote-group minerals. Eur J Mineral 18:551–567
Armstrong JT (1988) Quantitative analysis of silicate and oxide materials: comparison of Monte Carlo, ZAF, and ϕ(rz) procedures. In: Newbury DE (ed) Microbeam analysis. San Francisco Press, pp 239–246
Baillie MG, Hayton JD (1970) A process for the recovery of high grade rare earth concentrates from Mary Kathleen uranium tailings. In: 9th International Mineral Processing Congress (1970: Czechoslovakia). Institution of Mining and Metallurgy, London, pp. 334–345
Binnemans K, Jones PT, Müller T, Yurramendi L (2018) Rare earths and the balance problem: how to deal with changing markets? J Sustain Metall 4:126–146
Demol J, Ho E, Soldenhoff K, Senanayake G (2019) The sulfuric acid bake and leach route for processing rare earth ores and concentrates: a review. Hydrometallurgy 188:123–139
Dreisinger D, Verbaan N, Johnson M (2016) The search minerals direct extraction process for rare earth element recovery. In: Alam S, Kim H, Neelameggham NR, Ouchi T, Oosterhof H (eds) Rare metals technology 2016. Wiley-TMS, pp 3-16
European Commission, Directorate-General for Internal Market, Industry, Entrepreneurship, and SMEs (2017). Study on the review of the list of critical raw materials. Publications Office of the European Union. https://op.europa.eu/en/publication-detail/-/publication/08fdab5f-9766-11e7-b92d-01aa75ed71a1.
Hellmann R, Wirth R, Daval D, Barnes J-P, Penisson J-M, Tisserand D, Epicier T, Florin B, Hervig RL (2012) Unifying natural and laboratory chemical weathering with interfacial dissolution–reprecipitation: a study based on the nanometer-scale chemistry of fluid–silicate interfaces. Chem Geol 294-295:203–216
Hutton CO (1951) Allanite from Yosemite National Park, Tuolumne co., California. Am Mineral 36:233–248
Kobylin P, Kaskiala T, Salmien J (2007) Modeling of H2SO4-FeSO4-H2O and H2SO4-Fe2(SO4)3 systems for metallurgical applications. Ind Eng Chem Res 46:2601–2608
Linke WF, Seidell A (1965) Solubilities, inorganic and metal-organic compounds: a compilation of solubility data from the periodical literature. Van Nostrand, New York, 1914 pp
Nicol MJ, Akilan C (2018) The kinetics of the dissolution of chrysocolla in acid solutions. Hydrometallurgy 178:7–11
Notzl H, Khan S, Verbaan N (2013) Hydrometallurgical plant design parameters for the Avalon rare earth process. In: London IM, Goode JR, Moldoveanu G, Rayat MS (eds) Proceedings of the 52nd conference of metallurgists (COM 2013). Canadian Institute of Mining, Metallurgy and Petroleum, Montreal, Quebec, Canada, pp 201–214
Ruiz-Agudo E, Putnis CV, Putnis A (2014) Coupled dissolution and precipitation at mineral-fluid interfaces. Chem Geol 383:132–146
Sadri F, Nazari AM, Ghahreman A (2017) A review on the cracking, baking and leaching process of rare earth elements concentrates. J Rare Earths 35:739–752
Terry B (1983a) The acid decomposition of silicate minerals part I. Reactivities and modes of dissolution of silicates Hydrometallurgy 10:135–150
Terry B (1983b) The acid decomposition of silicate minerals part II. Hydrometallurgical applications Hydrometallurgy 10:151–171
Um N, Hirato T (2013) Dissolution behaviour of La2O3, Pr2O3, Nd2O3, CaO and Al2O3 in sulfuric acid solutions and study of cerium recovery from rare earth polishing powder waste via two-stage sulfuric acid leaching. Mater Trans 54:713–719
U.S. Department of Energy (2011) Critical materials strategy. U.S. Department of Energy, Office of International Affairs https://www.energy.gov/node/349057.
Van Gosen BS, Verplanck PL, Seal RR, Long KR, Gambogi J (2017) Rare earth elements. In: Schulz KJ, DeYoung JH, Seal RR, Bradley DC (eds) Critical mineral resources of the United States – economic geology and prospects for future supply, US Geol Surv prof paper, vol 1802, pp O1–O31
Verbaan N, Bradley K, Brown J, Mackie S (2015) A review of hydrometallurgical flowsheets considered in current REE projects. In: Simandl GJ, Neetz M (eds) Symposium on strategic and critical materials proceedings, November 13–14 2015, Victoria, British Columbia. British Columbia Geol Surv paper, vol 2015-3, pp 147–162
Wise ME, Brooks SD, Garland RM, Cziczo DJ, Martin ST, Tolbert MA (2003) Solubility and freezing effects of Fe2+ and Mg2+ in H2SO4 solutions representative of upper tropospheric and lower stratospheric sulfate particles. J Geophys Res 108:AAC15–1–AAC15–11
Acknowledgements
Technical support for the EPMA characterization by Dominique Duguay, the XRD analyses by Derek Smith, and the image processing by Rolando Lastra from CanmetMINING (Ottawa, ON) are gratefully acknowledged. We are grateful to three anonymous peers for their constructive reviews. Financial support for this project was provided by Natural Resources Canada through a special fund for the REE and Chromite Research and Development Initiative.
Author information
Authors and Affiliations
Corresponding author
Additional information
Editorial handling: L. Nasdala
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Thibault, Y., Gamage McEvoy, J. Minimizing the impact of passivation during allanite-(Ce) decomposition in sulfuric acid media for rare earth recovery. Miner Petrol 114, 559–571 (2020). https://doi.org/10.1007/s00710-020-00724-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00710-020-00724-w